Eukaryotic initiation factor EIF-3.G augments mRNA translation efficiency to regulate neuronal activity
Abstract
The translation initiation complex eIF3 imparts specialized functions to regulate protein expression. However, understanding of eIF3 activities in neurons remains limited despite widespread dysregulation of eIF3 subunits in neurological disorders. Here, we report a selective role of the C. elegans RNA-binding subunit EIF-3.G in shaping the neuronal protein landscape. We identify a missense mutation in the conserved Zinc-Finger (ZF) of EIF-3.G that acts in a gain-of-function manner to dampen neuronal hyperexcitation. Using neuron-type-specific seCLIP, we systematically mapped EIF-3.G-mRNA interactions and identified EIF-3.G occupancy on GC-rich 5′UTRs of a select set of mRNAs enriched in activity-dependent functions. We demonstrate that the ZF mutation in EIF-3.G alters translation in a 5′UTR-dependent manner. Our study reveals an in vivo mechanism for eIF3 in governing neuronal protein levels to control neuronal activity states and offers insights into how eIF3 dysregulation contributes to neurological disorders.
Introduction
Protein synthesis is principally regulated by variations in the translation initiation mechanism, whereby multiple eukaryotic initiation factors (eIF1 through 6) engage elongation-competent ribosomes with the mRNA open reading frame (Sonenberg and Hinnebusch, 2009). eIF3 is the largest translation initiation complex, composed of 13 subunits in metazoans, with versatile functions throughout the general translation initiation pathway (Valášek et al., 2017). Extensive biochemical and structural studies have shown that eIF3 promotes translation initiation by orchestrating effective interactions between the ribosome, target mRNA, and other eIFs (Smith et al., 2016; Cate, 2017). Mutations and misexpression of various subunits of eIF3 are associated with human diseases, such as cancers and neurological disorders (Gomes-Duarte et al., 2018), raising the importance to advance mechanistic understanding of eIF3’s function in vivo.
Recent work has begun to reveal that different eIF3 subunits can selectively regulate translation in a manner depending on cell type, mRNA targets, and post-translational modification. Interaction of eIF3 RNA-binding subunits with specific 5′UTR stem-loop structures of mRNAs can trigger a translational switch for cell proliferation in human 293 T cells (Lee et al., 2015), and can also act as a translational repressor, such as the case for human Ferritin mRNA (Pulos-Holmes et al., 2019). Under cellular stress, such as heat shock, the eIF3 complex circumvents cap-dependent protein translation initiation and recruits ribosomes directly to m6A marks within the 5′UTR of mRNAs encoding stress response proteins (Meyer et al., 2015). Other specialized translation mechanisms appear to involve activities of particular eIF3 subunits that were previously hidden from view. For example, human eIF3d possesses a cryptic mRNA cap-binding function that is activated by phosphorylation and stimulates pre-initiation complex assembly on specific transcripts (Lee et al., 2016; Lamper et al., 2020), while eIF3e specifically regulates metabolic mRNA translation (Shah et al., 2016). These findings hint that many other eIF3-guided mechanisms of cell-specific translational control await discovery.
In the nervous system, emerging evidence suggests that eIF3 subunits may have critical functions. Knockdown of multiple eIF3 subunits impairs expression of dendrite pruning factors in developing sensory neurons of Drosophila (Rode et al., 2018). In mouse brain, eIF3h directly interacts with collybistin, a conserved neuronal Rho-GEF protein underlying X-linked intellectual disability with epilepsy (Sertie et al., 2010; Machado et al., 2016). In humans, altered expression of the eIF3 complex in the substantia nigra and frontal cortex correlates with Parkinson’s Disease progression (Garcia-Esparcia et al., 2015). Downregulation of mRNAs encoding eIF3 subunits is observed in a subset of motor neurons in amyotrophic lateral sclerosis patients (Cox et al., 2010). Furthermore, a single-nucleotide polymorphism located in the intron of human eIF3g elevates its mRNA levels and is associated with narcolepsy (Holm et al., 2015). While these data suggest that eIF3 function in neurons is crucial, mechanistic understanding will require experimental models enabling in vivo investigation of how eIF3 affects protein translation with neuron-type specificity.
Protein translation in C. elegans employs all conserved translation initiation factors. We have investigated the mechanisms of protein translation in response to neuronal overexcitation using a gain-of-function (gf) ion channel that arises from a missense mutation in the pore-lining domain of the acetylcholine receptor subunit ACR-2 (Jospin et al., 2009). The cholinergic motor neurons (ACh-MNs) in the ventral cord of acr-2(gf) mutants experience constitutive excitatory inputs, which gradually diminish pre-synaptic strength and cause animals to display spontaneous seizure-like convulsions and uncoordinated locomotion (Jospin et al., 2009; Zhou et al., 2017). acr-2(gf) induces activity-dependent transcriptome changes (McCulloch et al., 2020). However, it is unclear how protein translation conducts the activity-dependent proteome changes that sustain function of these neurons.
Here, we demonstrate that C. elegans EIF-3.G/eIF3g regulates the translation efficiency of select mRNAs in ACh-MNs. We characterized a mutation (C130Y) in the zinc-finger of EIF-3.G that suppresses behavioral deficits of acr-2(gf) without disrupting general protein translation. By systematic profiling of EIF-3.G and mRNA interactions in ACh-MNs, we identified preferential binding of EIF-3.G to long and GC-rich 5'UTRs of mRNAs, many of which encode modulators of ACh-MN activity. We further provided in vivo evidence that EIF-3.G regulates the expression of two of its mRNA targets dependent on their 5′UTRs. Our findings illustrate the selectivity of EIF-3.G in augmenting mRNA translation to mediate neuronal activity changes.
Results
A missense mutation in EIF-3.G ameliorates convulsion behaviors caused by cholinergic hyperexcitation
We previously characterized numerous mutations that suppress convulsion and locomotion behaviors of acr-2(gf) animals (McCulloch et al., 2017). One such suppressor mutation, ju807, was found to contain a single nucleotide alteration in eif-3.G, encoding subunit G of the EIF-3 complex (Figure 1A; see Materials and methods). C. elegans EIF-3.G is composed of 262 amino acids, sharing overall 35% or 32% sequence identity with human eIF3g and S. cerevisiae TIF35 orthologs, respectively (Figure 1—figure supplement 1A). Both biochemical and structural data show that eIF3g/TIF35 proteins bind eIF3i/TIF34 through a domain in the N-terminus (Figure 1B; Valášek et al., 2017). eIF3g/EIF-3.G also has a predicted CCHC zinc finger followed by an RNA recognition motif (RRM) at the C-terminus (Figure 1B and Figure 1—figure supplement 1A). The ju807 mutation changes the second cysteine of the CCHC motif (Cys130, corresponding to Cys160 in human eIF3g) to tyrosine (Figure 1B). Hereafter, we designate eif-3.G(ju807) as eif-3.G(C130Y).
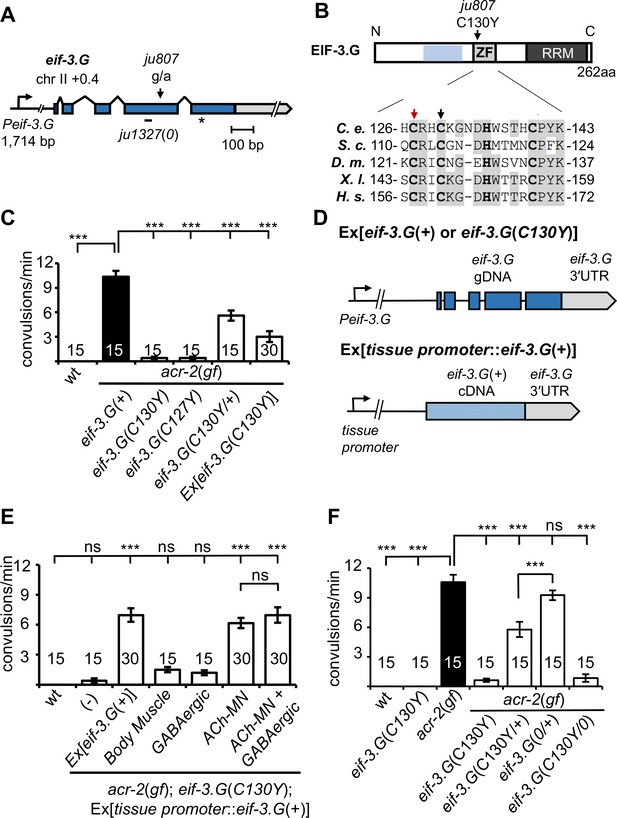
eif-3.G(C130Y) suppresses acr-2(gf) convulsion behavior in the cholinergic motor neurons.
(A) Illustration of the genomic locus of eif-3.G: Peif-3.G denotes the promoter, blue boxes are exons for coding sequences and gray for 3′UTR. Arrowhead indicates guanine to adenine change in ju807; and short line below represents a 19 bp deletion in ju1327, designated eif-3.G(0), that would shift the reading frame at aa109, resulting in a premature stop (asterisk) after addition of 84aa of no known homology. (B) Illustration of EIF-3.G: shaded blue represents EIF-3.I binding region, ZF for Zinc Finger, RRM for RNA Recognition Motif. Below is a multi-species alignment of the zinc finger domain with bold residues as the CCHC motif and gray for conserved residues. ju807 causes a C130Y substitution (black arrow). C127Y (red arrow, ju1840) was generated with CRISPR editing. C. elegans (C. e.; NP_001263666.1), S. cerevisiae (S. c.; NP_010717.1), D. melanogaster (D. m.; NP_570011.1), X. laevis (X. l.; NP_001087888.1), and H. sapiens (H.s.; AAC78728.1). (C) Quantification of convulsion frequencies of animals of indicated genotypes, with the strains (left to right) as: N2, MT6241, CZ21759, CZ28495, CZ21759, CZ22977. Ex[eif-3.G(C130Y)] transgenes (juEx7015/juEx7016) expressed full-length genomic DNA cloned from eif-3.G(ju807). (D) Illustration of eif-3.G expression constructs: top shows the transgene expressing genomic eif-3.G(+ for wild type and C130Y for ju807) with the endogenous eif-3.G promoter and 3′UTR, and coding exons in blue; bottom shows cell-type expression of eif-3.G cDNA driven by tissue-specific promoters (Pmyo-3- body muscle, Punc-25- GABAergic motor neurons, Punc-17β - cholinergic motor neurons). (E) Quantification of convulsion frequencies shows that convulsion behavior of eif-3.G(C130Y); acr-2(gf) double mutants is rescued by transgenes that overexpress eif-3.G(+) genomic DNA or an eif-3.G(+) cDNA in the ACh-MNs, but not in the GABAergic motor neurons or body muscle. Strains (left to right)- N2, CZ21759, CZ23125/ CZ23126, CZ22980/ CZ22981, CZ23791/ CZ23880, CZ22982/ CZ22983, CZ27881/ CZ27882. (F) Quantification of convulsion frequencies in animals of the indicated genotypes (left to right)- N2, CZ22917, MT6241, CZ21759, CZ28495, CZ21759, CZ21759, CZ23310, CZ26828. Data in (D-F) are shown as mean ± SEM and sample size is indicated within or above each bar. Statistics: (***) p<0.001, (ns) not significant by one-way ANOVA with Bonferroni’s post hoc test.
-
Figure 1—source data 1
Source data for Figure 1C.
Quantification of convulsions per 60 s in strains of the indicated genotypes.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig1-data1-v2.xlsx
-
Figure 1—source data 2
Source data for Figure 1E.
Quantification of convulsions per 60 s in the indicated strains.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig1-data2-v2.xlsx
-
Figure 1—source data 3
Source data for Figure 1F.
Quantification of convulsions per 60 s in strains. Strain name or genotype is indicated in top row.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig1-data3-v2.xlsx
Compared to acr-2(gf) single mutants, eif-3.G(C130Y); acr-2(gf) animals exhibited nearly wild-type movement and strongly attenuated convulsion behavior (Figure 1C; Videos 1–3). acr-2(gf) animals carrying heterozygous eif-3.G(C130Y/+) showed partial suppression of convulsions (Figure 1C). Overexpression of wild type eif-3.G full-length genomic DNA in eif-3.G(C130Y); acr-2(gf) double mutants restored convulsions to levels similar to eif-3.G(C130Y/+); acr-2(gf) (Figure 1D–E; Materials and methods). Overexpression of eif-3.G(C130Y) full-length genomic DNA in acr-2(gf) single mutants also partially suppressed convulsions (Figure 1C–D). In wild-type animals, overexpression of eif-3.G(+) or eif-3.G(C130Y) caused no observable effects on locomotion. These data show that eif-3.G(C130Y) acts in a semi-dominant manner to ameliorate convulsion and uncoordinated locomotion behaviors of acr-2(gf). To further test that altering the EIF-3.G zinc finger motif accounts for the observed suppression of acr-2(gf), we edited the first cysteine of the CCHC motif (Cys127) to tyrosine using CRISPR-Cas9, and found that eif-3.G(C127Y) suppressed acr-2(gf) convulsions to levels identical to eif-3.G(C130Y) (Figure 1B–C). This data provides support for the importance of the EIF-3.G zinc finger in regulation of ACh-MN activity. Hereafter, we focused our analysis on eif-3.G(C130Y).
N2 [Wild type] C. elegans movement on solid nematode growth media.
MT6241 [acr-2(gf)] C. elegans movement on solid nematode growth media.
CZ21759 [eif-3.G(C130Y); acr-2(gf)] C. elegans movement on solid nematode growth media.
We next determined in which cell types eif-3.G(C130Y) functions using cell-specific expression analysis (Figure 1D; also see Materials and methods). acr-2(gf) phenotypes arise from a hyperactive ACR-2-containing ion channel expressed in the ventral cord cholinergic motor neurons (ACh-MNs) (Jospin et al., 2009). We found that overexpressing eif-3.G(+) cDNA in ACh-MNs (Punc-17β) restored convulsions of eif-3.G(C130Y); acr-2(gf) animals to a similar degree as those expressing full-length eif-3.G(+) under the endogenous promoter (Peif-3.G) (Figure 1E). In contrast, overexpression of eif-3.G(+) cDNA in either ventral cord GABAergic neurons (GABA-MNs, Punc-25) or body muscle (Pmyo-3) in eif-3.G(C130Y); acr-2(gf) animals caused no detectable effects (Figure 1E). Co-expression of eif-3.G(+) in both ACh-MNs and GABA-MNs showed similar effects on eif-3.G(C130Y); acr-2(gf) animals to that from expressing eif-3.G(+) in ACh-MNs alone (Figure 1E). Thus, eif-3.G(C130Y) functions in ACh-MN to modulate acr-2(gf) behaviors.
EIF-3.G(C130Y) selectively affects translation in ACh-MNs
eif-3.G(C130Y) single mutants exhibit normal development, locomotion, and other behaviors (such as male mating and egg-laying) indistinguishably from wild-type animals (Figure 1F, Video 4). Axon morphology and synapse number of ACh-MNs were also normal in eif-3.G(C130Y) animals (Figure 1—figure supplement 2A–B). To dissect how the C130Y mutation affects EIF-3.G function, we next generated a genetic null mutation (ju1327) using CRISPR editing (Figure 1A and Figure 1—figure supplement 1A; designated eif-3.G(0), see Materials and methods). Homozygous eif-3.G(0) animals arrested development at L1 stage, consistent with EIF-3 complex members being required for C. elegans development (Kamath et al., 2003). eif-3.G(0/+); acr-2(gf) animals were indistinguishable from acr-2(gf) single mutants (Figure 1F). We additionally tested null mutations in EIF-3.E and EIF-3.H, two essential subunits of EIF-3 complex, and found that acr-2(gf) animals carrying heterozygous null mutations in either eif-3 subunit gene showed convulsions similar to eif-3.G(0/+); acr-2(gf) (Figure 1—figure supplement 3A). Moreover, hemizygous eif-3.G(C130Y/0) animals are healthy at all stages and suppress behaviors of acr-2(gf) to levels comparable to eif-3.G(C130Y) (Figure 1F). Reducing one copy of eif-3.H(+) or eif-3.E(+) in eif-3.G(C130Y); acr-2(gf) animals also did not modify the suppression effect of eif-3.G(C130Y) (Figure 1—figure supplement 3A). These observations suggest that eif-3.G(C130Y) retains sufficient function of wild-type eif-3.G, and likely affects a regulatory activity that is not dependent on EIF-3 subunit dosage.
CZ22197 [eif-3.G(C130Y)] C. elegans movement on solid nematode growth media.
We considered that EIF-3.G(C130Y) could alter EIF-3.G protein levels in ACh-MNs. To test this, we generated single-copy chromosomal integrated transgenes expressing EIF-3.G(WT) or EIF-3.G(C130Y) tagged with GFP at the N-terminus under the control of the endogenous eif-3.G promoter (Materials and methods and Supplementary file 1). Fluorescence from both GFP::EIF-3.G(WT) and GFP::EIF-3.G(C130Y) was observed in all somatic cells (Figure 1—figure supplement 1B). In ACh-MNs, both proteins showed cytoplasmic localization (Figure 2B). The GFP::EIF-3.G(WT) transgene rescued eif-3.G(0) to adults (Supplementary file 1) and also restored convulsion behavior in the eif-3.G(C130Y); acr-2(gf) background (Figure 2A). In contrast, the GFP::EIF-3.G(C130Y) transgene reduced convulsion behavior in the acr-2(gf) background. Furthermore, we introduced the GFP::EIF-3.G(C130Y) transgene into the eif-3.G(0); acr-2(gf) background and observed that this transgene rescued the arrested larvae to adults and nearly abolished convulsion behavior (Figure 2A). This analysis shows that GFP::EIF-3.G(WT) and GFP::EIF-3.G(C130Y) retain function and lends further support that eif-3.g(C130Y) is responsible for the suppression of acr-2(gf). Quantification of GFP levels in the ACh-MNs showed equivalent intensity and localization of GFP::EIF-3.G (WT and C130Y) between wild type and acr-2(gf) animals (Figure 2B), indicating that EIF-3.G(C130Y) does not increase EIF-3.G protein stability.
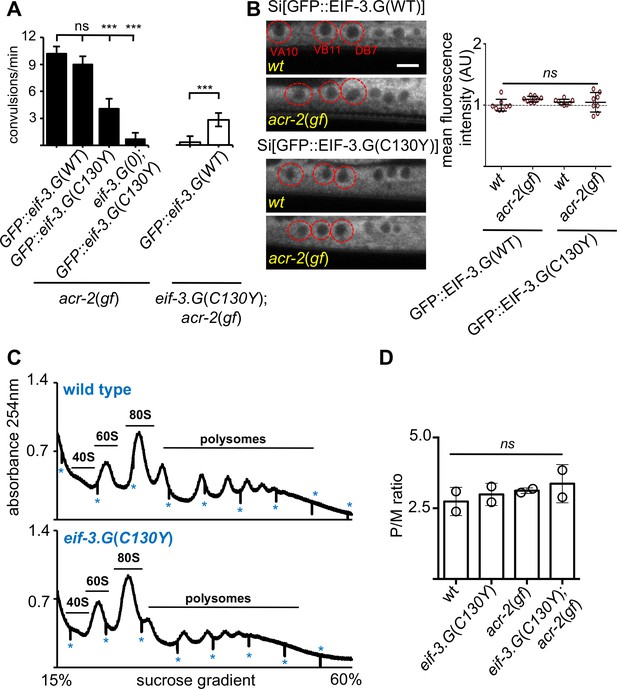
eif-3.G(C130Y) involves a selective function of EIF-3.G on translational control.
(A) Quantification of convulsion frequency in animals expressing GFP::EIF-3.G(WT) or GFP::EIF-3.G(C130Y) under Peif-3.G in the indicated genetic backgrounds; and the strains (left to right) are: MT6241, CZ24729, CZ24652, CZ28497, CZ21759, CZ28107. Error bars represent ± SEM with n = 15 per sample. (***) P< 0.001, (ns) not significant, by one-way ANOVA with Bonferroni’s post-hoc test. (B) EIF-3.G(WT) and EIF-3.G(C130Y) show comparable expression in ACh-MNs. Left are representative single-plane confocal images of EIF-3.G(WT)::GFP or EIF-3.G(C130Y)::GFP driven by the Peif-3.G promoter as single-copy transgenes in L4 animals (head to the left). Red circles mark the soma of VA10, VB11, and DB7 ACh-MN, based on co-expressing a Pacr-2-mcherry marker. Scale bar = 4 µm. Right: Mean GFP fluorescence intensities (AU) in ACh-MN soma in animals of the indicated genotypes (n = 8). Each data point represents the mean intensity from VA10, VB11, and DB7 neurons in the same animal and normalized to the mean GFP::EIF-3.G intensity in a wildtype background. Error bars represent ± SEM; (ns) not significant by one-way ANOVA with Sidak’s multiple comparisons test. (C) Representative polysome profile traces from total mRNA-protein extracts of wild type and eif-3.G(C130Y) single mutant animals. Vertical lines (marked by *) within traces indicate the boundaries of fraction collection. (D) Polysome::monosome (P/M) ratios calculated based on the area under the respective curves for polysomal and monosome (80S) fractions using two replicates of polysome profiles from total extracts of indicated genotypes. (ns) not significant by one-way ANOVA with Bonferroni’s post-hoc test.
-
Figure 2—source data 1
Source data for Figure 2A.
Quantification of convulsions per 60 s in the indicated strains.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig2-data1-v2.xlsx
-
Figure 2—source data 2
Source data for Figure 2B.
Quantification of relative fluorescence intensity in the indicated strains.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig2-data2-v2.xlsx
-
Figure 2—source data 3
Source data for Figure 2C.
Quantification of polysome to monosome ratios in wildtype(N2) and strains of the indicated genotypes.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig2-data3-v2.xlsx
We further assessed whether eif-3.G(C130Y) alters global translation by performing polysome profile analysis using whole C. elegans lysates of L4 stage animals. Both the distribution and ratio of monosomes and polysomes were similar among wild type, eif-3.G(C130Y), acr-2(gf) and eif-3.G(C130Y); acr-2(gf) animals (Figure 2C–D), indicating that eif-3.G(C130Y) possesses normal function in the majority of tissues. It is possible that eif-3.G(C130Y) suppresses acr-2(gf) by simply reducing ACR-2 translation. We tested this by examining a functional GFP-tagged ACR-2 single-copy insertion transgene (oxSi39). We observed both the levels of ACR-2::GFP fluorescence and post-synaptic localization in ACh-MNs were comparable between wild type and eif-3.G(C130Y) animals (Figure 1—figure supplement 3B). These data support the conclusion that eif-3.G(C130Y) preferentially affects EIF-3’s function in ACh-MNs.
The activity of EIF-3.G(C130Y) requires its RRM
The RRM located at the C-terminus of eIF3g has been shown to bind RNA in a non-specific manner (Hanachi et al., 1999). To address the role of the RRM in EIF-3.G’s function, we generated a transgene expressing EIF-3.G(∆RRM) (Figure 3; Supplementary file 1). Expressing EIF-3.G(∆RRM) under the endogenous promoter Peif-3.G in a wild-type background did not alter development or locomotion, and also did not rescue eif-3.G(0) developmental arrest, supporting the essentiality of the EIF-3.G RRM. We then generated a transgene expressing EIF-3.G(C130Y) lacking the RRM domain (C130Y ∆RRM) in neurons of the acr-2(gf) background (Figure 3). In contrast to full-length eif-3.G(C130Y), eif-3.G(C130Y ∆RRM) did not alter convulsion behavior of acr-2(gf) mutants (Figure 3B), indicating that eif-3.G(C130Y) function requires its RRM.
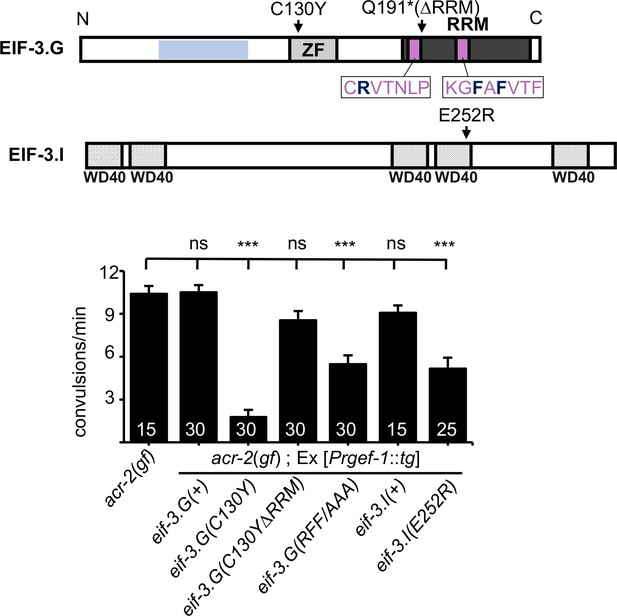
eif-3.G(C130Y) requires the RNA-binding domain (RRM) to suppress acr-2(gf) behaviors.
Top illustration of the EIF-3.G protein showing the EIF-3.I binding region (blue), zinc finger (ZF), RRM (dark grey), Q191* mutation in the EIF-3.G(∆RRM) transgene, RNP motifs (purple), and the RFF residues (bold dark blue) changed to alanine in the eif-3.G(RFF/AAA) construct. Below is an illustration of C. elegans EIF-3.I pointing to the position of E252R within the fourth WD40 domain. Bottom graph is quantification of convulsion frequency in acr-2(gf) animals expressing eif-3.G and eif-3.I variants in the nervous system (Prgef-1). The strains (left to right) are: MT6241, CZ23203/ CZ23204, CZ28152/ CZ28153, CZ23304/ CZ23305, CZ28152/ CZ28153, CZ28057/ CZ28058, CZ28064/ CZ28065. Bars represent mean convulsion frequency ± SEM and sample sizes are indicated within or above bars. (***) p< 0.001, (ns) not significant, by one-way ANOVA with Bonferroni’s post-hoc test.
-
Figure 3—source data 1
Source data for Figure 3.
Quantification of convulsions per 60 s in the indicated strains.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig3-data1-v2.xlsx
Studies on S. cerevisiae TIF35/EIF3.G have shown that its RRM promotes scanning of the translation pre-initiation complex through structured 5′UTRs (Cuchalová et al., 2010). Specifically, alanine substitution of three residues in the two ribonucleoprotein (RNP) motifs (K194 in RNP2 and L235 and F237 in RNP1) in TIF35 reduced translation of mRNA reporters carrying 5′UTRs with hairpin structures, without altering the biochemical RNA-binding activity of EIF-3.G/TIF35. Equivalent amino acid residues in C. elegans EIF-3.G correspond to R185, F225, F227, which are conserved in human (R242, F282, F284) (Figure 3; Figure 1—figure supplement 1A). To determine whether these residues affect EIF-3.G’s function, we expressed C. elegans eif-3.G cDNA with the corresponding amino acids mutated to alanine, designated eif-3.G(RFF/AAA), in acr-2(gf) animals. We detected partial suppression of convulsion behavior in acr-2(gf) animals (Figure 3).
It was also reported that a missense mutation (Q258R) in yeast EIF-3.I/TIF34, located in the sixth WD40 repeat, reduced the rate of pre-initiation complex scanning through 5′UTRs (Cuchalová et al., 2010). To test if C. elegans eif-3.I shares similar activities, we made a mutant EIF-3.I(E252R), equivalent to yeast TIF34 (Q258R) (Figure 3). In acr-2(gf) animals, overexpressing eif-3.I(E252R), but not wild-type eif-3.I(+), caused suppression of convulsions to a similar degree as that by the eif-3.G(RFF/AAA) transgene (Figure 3). These analyses suggest that attenuation of acr-2(gf)-induced neuronal overexcitation may involve regulation of protein translation through modification of 5′UTR scanning rates during translation initiation.
Both EIF-3.G(WT) and EIF-3.G(C130Y) associate with mRNA 5′UTRs in the cholinergic motor neurons
EIF-3.G may interact with specific mRNAs in the nervous system to regulate cholinergic activity. Therefore, we next searched for mRNAs that are associated with EIF-3.G(WT) and EIF-3.G(C130Y) in the ACh-MNs using single-end enhanced crosslinking and immunoprecipitation (Van Nostrand et al., 2017). We generated single-copy transgenes expressing 3xFLAG-tagged EIF-3.G(WT), EIF-3.G(C130Y), or EIF-3.G(∆RRM) in the ACh-MNs of acr-2(gf) animals, with EIF-3.G(∆RRM) serving to detect indirect crosslinking events. We confirmed that the truncated EIF-3.G(∆RRM) transgene was expressed, but at reduced levels compared to the EIF-3.G(WT) and EIF-3.G(C130Y) transgenes (Figure 4—figure supplement 1A). Following cross-linking and immunoprecipitation using anti-FLAG antibodies, we obtained a comparable amount of immunoprecipitated GFP::EIF-3.G proteins and obtained more reads from seCLIP on animals expressing each GFP::EIF-3.G transgene than on control animals lacking any transgene (IgG(-); see Supplementary file 4). There was a strong correlation between read clusters detected among sets of two biological replicates (Figure 4—figure supplement 1B). We defined EIF-3.G-RNA crosslink sites as clusters of at least 20 high-quality reads with at least 1.5 fold change enrichment over the input control (see Materials and methods and Supplementary file 5). We further defined specific footprints of EIF-3.G(WT) and EIF-3.G(C130Y) by subtracting clusters detected with EIF-3.G(∆RRM) (Supplementary file 6, also see Materials and methods). The EIF-3.G-specific footprints were primarily located within or near the 5′UTRs of protein-coding genes (5′UTR proximal) (Figure 4A–B). In total, we detected 231 5′UTR proximal footprints of EIF-3.G(WT) or EIF-3.G(C130Y), which mapped to 225 different genes (Figure 4C). The number of reads comprising EIF-3.G(WT) or EIF-3.G(C130Y) footprints was similar (e.g. egl-30; Figure 4B) for most of these genes. While some footprints were differentially detected between EIF-3.G(WT) and EIF-3.G(C130Y), this was almost invariably due to small differences in seCLIP signal intensity (read cluster size) between samples close to the 20 reads threshold (Figure 4C), and we therefore did not further pursue its significance.
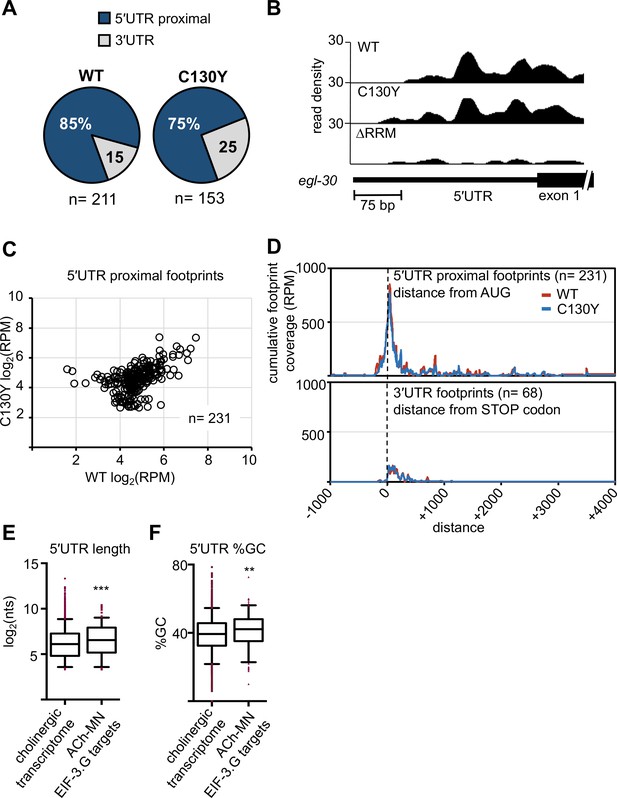
Both EIF-3.G(WT) and EIF-3.G(C130Y) associate with mRNA 5′UTRs in the cholinergic motor neurons.
(A) Pie charts displaying the proportion of EIF-3.G(WT) and EIF-3.G(C130Y) footprints located within each gene feature. (B) seCLIP read density track of EIF-3.G(WT) and EIF-3.G(C130Y) footprints on the 5′UTR of egl-30, compared to the EIF-3.G(∆RRM) control. (C) Scatter plot comparing the signal intensity, in reads per million (RPM), of all 231 5′UTR proximal footprints detected in EIF-3.G(WT) or EIF-3.G(C130Y). (D) Plots show the cumulative coverage of all 5′UTR proximal (top) or 3′UTR (bottom) footprints of EIF-3.G(WT) or EIF-3.G(C130Y) relative to the start codon (top) or stop codon (bottom) position. Coverage is presented as reads per million (RPM). (E–F) Box plots comparing length and GC-content of all 5′UTR sequences of EIF-3.G target mRNAs with annotations (n = 179) to all 5′UTRs in the acr-2(gf) cholinergic neuronal transcriptome (n = 4573). Boxes are 5–95 percentile with outliers aligned in red. Statistics: (***) p< 0.001, (**) p< 0.01 by two-tailed Mann-Whitney test.
-
Figure 4—source data 1
Source data for Figure 4A.
Number of read clusters representing footprints of EIF-3.G(WT) or EIF-3.G(C130Y) mapping to 5′UTR proximal or 3′UTR regions.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig4-data1-v2.xlsx
-
Figure 4—source data 2
Source data for Figure 4C.
seCLIP reads for EIF-3.G(WT) and EIF-3.G(C130Y) 5’UTR proximal footprints represented as log2(reads per million).
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig4-data2-v2.xlsx
-
Figure 4—source data 3
Source data for Figure 4D.
Cumulative EIF-3.G(WT) and EIF-3.G(C130Y) footprint coverage per base distance from the start codon (5’UTR proximal footprints) or stop codon (3’UTR footprints) represented as reads per million.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig4-data3-v2.xlsx
-
Figure 4—source data 4
Source data for Figure 4E.
Length of 5’UTRs in mRNAs expressed in the ACh-MN transcriptome and EIF-3.G targets.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig4-data4-v2.xlsx
-
Figure 4—source data 5
Source data for Figure 4F.
Percent GC of 5’UTRs in mRNAs expressed in the ACh-MN transcriptome and EIF-3.G targets.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig4-data5-v2.xlsx
In line with a recent report that the human eIF3 complex remains attached to 80S ribosomes in early elongation (Wagner et al., 2020), we observed the bulk of read clusters comprising EIF-3.G(WT) and EIF-3.G(C130Y) footprints mapping between (-)150 to (+)200 nucleotides of the start codon (Figure 4D). In contrast, the majority of signals comprising 3’UTR footprints of EIF-3.G(WT) and EIF-3.G(C130Y) were dispersed along the first 200 nucleotides downstream of the stop codon (Figure 4D). Overall, the footprint map shows that both EIF-3.G(WT) and EIF-3.G(C130Y) predominantly bind to similar locations within or near the 5′UTRs of 225 genes in the ACh-MNs, hereafter named EIF-3.G targets. Taken together with our finding that eif-3.G(C130Y) requires its RRM to suppress acr-2(gf), the seCLIP analysis suggests that the C130Y mutation does not dramatically alter the ability of EIF-3.G to associate with these mRNAs in the ACh-MNs.
EIF-3.G preferentially interacts with long and GC-rich 5′UTR sequences
5′UTR sequences are widely involved in gene-specific regulation of translation (Pelletier and Sonenberg, 1985; Leppek et al., 2018). We next assessed whether the selective role of EIF-3.G in protein translation might correlate with specific sequence features in the mRNA targets expressed in ACh-MNs by examining the length and GC-content of their 5′UTRs. In C. elegans, about 70% of mRNAs are known to undergo trans-splicing, and 5′UTRs of mRNAs with trans-splice leaders are usually short, with a median length of 29nt. We compared the EIF-3.G target gene list with a database containing a compilation of C. elegans trans-splice events from ENCODE analyses (Allen et al., 2011). We found that 133 of the 225 (59%) EIF-3.G targets are annotated to undergo trans-splicing, which is comparable to that of transcriptome-wide (Allen et al., 2011; Figure 4—figure supplement 2A), suggesting that trans-splicing events may not contribute to EIF-3.G’s selectivity on mRNA targets. Interestingly, we found that the trans-spliced 5′UTRs of these 133 transcripts are significantly longer (median length = 43nt), compared with all trans-spliced 5′UTRs in the C. elegans transcriptome (median length = 29nt; n = 6,674) (Figure 4—figure supplement 2B). To assess the GC content for EIF-3.G mRNA targets, we then applied a threshold to the cholinergic neuronal transcriptome of acr-2(gf) (McCulloch et al., 2020) defining a 5′UTR as at least 10 nucleotides upstream of ATG, and also selected the longest 5′UTR isoform per gene to avoid redundant analysis of target genes (see Materials and methods). Using this criterion, we identified a 5′UTR for 4573 different genes in the cholinergic transcriptome and for 179 of the 232 EIF-3.G targets in the ACh-MNs. The median 5′UTR among the 179 EIF-3.G target mRNAs was significantly longer (93 nt) and GC-enriched (42%), compared to the cholinergic transcriptome median (69 nt and 39% GC; n = 10,962; Figure 4E–F). We further analyzed the distribution of GC sequences in 5′UTRs, and observed non-random positioning such that some genes were relatively GC-rich near the start codon (e.g. zip-2 and sec-61) and others had enrichment closer to the distal 5' end (e.g. pdf-1 and kin-10), suggesting that discrete sequence elements in EIF-3.G associated transcripts may regulate translation (Figure 4—figure supplement 2C).
The incidence of long and GC-enriched 5′UTRs among EIF-3.G associated transcripts led us to speculate a major function of EIF-3.G, in addition to its necessity in general translation initiation, is in the selective regulation of translation. To extend our findings beyond C. elegans, we asked if the preferential association of EIF-3.G with these complex 5′UTRs could be conserved in mammals. We analyzed the published eIF3g PAR-CLIP sequencing data from HEK293 cells (Lee et al., 2015) by comparing the 5′UTR lengths of human eIF3g target genes to all genes with 5′UTRs annotated in the hg38 genome. We found that human transcripts associated with eIF3g contained significantly longer and GC-enriched 5′UTRs than average (Figure 4—figure supplement 2D–E). This analysis lends support for a conserved, specialized role of eIF3g in the translation of transcripts harboring complex 5′UTRs.
EIF-3.G target mRNAs encode proteins that exhibit activity-dependent expression
To address whether EIF-3.G target mRNAs may preferentially affect specific biological processes, we performed Gene Ontology and KEGG pathway analysis. Significant GO term (Ashburner et al., 2000) enrichment was identified in neuropeptide signaling genes (GO:0050793; 15 genes), which are known to affect acr-2(gf) behavior (Stawicki et al., 2013; McCulloch et al., 2020), and in stress response genes (GO: 0006950; 28 genes), which could modulate neuronal homeostasis or function under circuit activity changes (Figure 5A). We also found many EIF-3.G target genes involved in protein translation and protein metabolism processes (GO:0019538; 29 genes; Figure 5A). Additional enrichment was associated with metabolic components, kinase signaling, and calcium and synaptic signaling pathways (Figure 5A). Calcium and synaptic signaling genes included the CAMKII unc-43, and the G-proteins egl-30 and goa-1, which are all known to regulate ACh-MN synaptic activity (Miller et al., 1999; Richmond, 2005; Treinin and Jin, 2020).
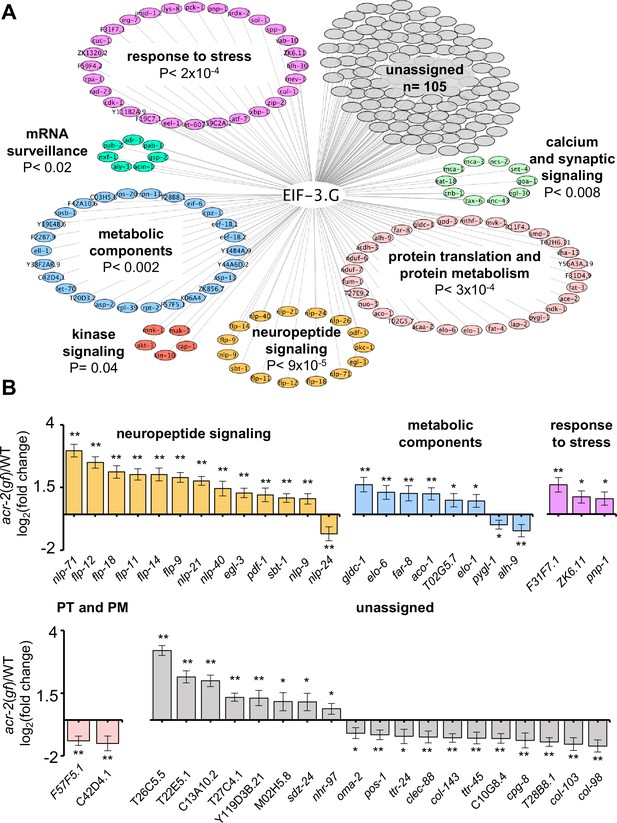
Gene network analyses of EIF-3.G target mRNAs show enrichment in activity-dependent expression.
(A) Cytoscape network of EIF-3.G target genes with enriched GO terms (neuropeptide signaling, response to stress, and protein translation and protein metabolism) or KEGG pathways (calcium and synaptic signaling, metabolic components, MAPK-signaling, and mRNA surveillance). Enrichment p-values are derived from statistical analysis of our EIF-3.G targets (n = 225) in the PANTHER database (Mi et al., 2019). (B) EIF-3.G target genes exhibiting significant transcript level changes in acr-2(gf) versus wild-type animals as determined from transcriptome sequencing of cholinergic neurons by McCulloch et al. PT and PM refers to protein translation and protein metabolism. Differential expression was assessed using DeSeq2 (Love et al., 2014) with significance thresholds of (*) p<0.05 and (**) p<0.01.
-
Figure 5—source data 1
Source data for Figure 5B.
Foldchange in transcript expression in the cholinergic neuronal transcriptome of acr-2(gf) and wild-type animals.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig5-data1-v2.xlsx
To determine if expression of EIF-3.G target mRNAs is regulated in an activity-dependent manner, we next incorporated differential transcript expression data between wild type and acr-2(gf) from a cholinergic neuron transcriptome dataset (McCulloch et al., 2020). We found that 83% of EIF-3.G target mRNAs in the ACh-MNs are present in the cholinergic neuron transcriptome. Among the 45 genes exhibiting significant expression changes dependent on acr-2(gf) (Figure 5B), nearly all neuropeptide signaling transcripts (12 of 15) as well as three stress response genes were upregulated in acr-2(gf) (Figure 5B). Genes encoding metabolic components were variably upregulated (e.g. Glycine decarboxylate/gldc-1, aconitase/aco-1) and downregulated (e.g. glycogen phosphorylase/pygl-1, aldehyde dehydrogenase/alh-9) (Figure 5B). These data support the idea that wild type EIF-3.G imparts translational control to activity-dependent expression changes and that EIF-3.G(C130Y) may exert specific regulation to alter their protein expression in ACh-MNs of acr-2(gf).
EIF-3.G modulates translation of HLH-30 and NCS-2 in hyperactive ACh-MNs
To experimentally validate that EIF-3.G regulates protein expression from its target mRNAs in the ACh-MNs, we next surveyed a number of candidate genes, chosen mainly based on the availability of transgenic reporters that contain endogenous 5′UTRs (Supplementary file 1). We identified two genes (hlh-30 and ncs-2) whose expression in ACh-MNs of acr-2(gf) animals shows dependency on EIF-3.G. hlh-30 produces multiple mRNA isoforms (Figure 6A), which encode the C. elegans ortholog of the TFEB stress response transcription factor with broad neuroprotective roles (Decressac et al., 2013; Polito et al., 2014; Lin et al., 2018). We observed strong seCLIP signals corresponding to EIF-3.G(WT) and EIF-3.G(C130Y) footprints in the 5′UTR of long isoform d, but not in isoform a (Figure 6B). The hlh-30d mRNA isoform has a 5′UTR of 190nt with 43% GC. Using computational RNA structure prediction (RNAfold), we found that the long hlh-30d 5′UTR forms strong stem-loop structures (∆G = −40.78 kcal/mol) that could affect HLH-30 translation. We examined expression of an HLH-30::EGFP fosmid reporter wgIs433, which encompasses the entire hlh-30 genomic region with cis-regulatory elements for all mRNA isoforms (Sarov et al., 2006; Figure 6C). HLH-30::GFP was observed throughout the nervous system and primarily localized to cytoplasm in all genetic backgrounds tested. We observed significantly enhanced HLH-30::GFP signals in the ACh-MNs of acr-2(gf) animals, compared to those in wild type (Figure 6C). While eif-3.G(C130Y) did not alter HLH-30::GFP, it reduced fluorescence intensity in acr-2(gf) to wild type levels (Figure 6C). As hlh-30 transcripts were detected at similar levels in ACh-MNs of wild type and acr-2(gf) animals (McCulloch et al., 2020), the enhanced HLH-30::GFP signal in acr-2(gf) likely reflects elevated translation upon neuronal activity changes, which is augmented by EIF-3.G. To strengthen this idea, we introduced an unc-13 null allele, which blocks presynaptic release (Richmond et al., 1999) to the above analyzed compound genetic mutants. We found that unc-13(0) abolished the enhanced HLH-30::GFP expression caused by acr-2(gf) (Figure 6C). Additionally, we tested a transgenic HLH-30a::GFP reporter expressing hlh-30a cDNA driven by the 2 kb sequence upstream of that isoform (Figure 6—figure supplement 1).
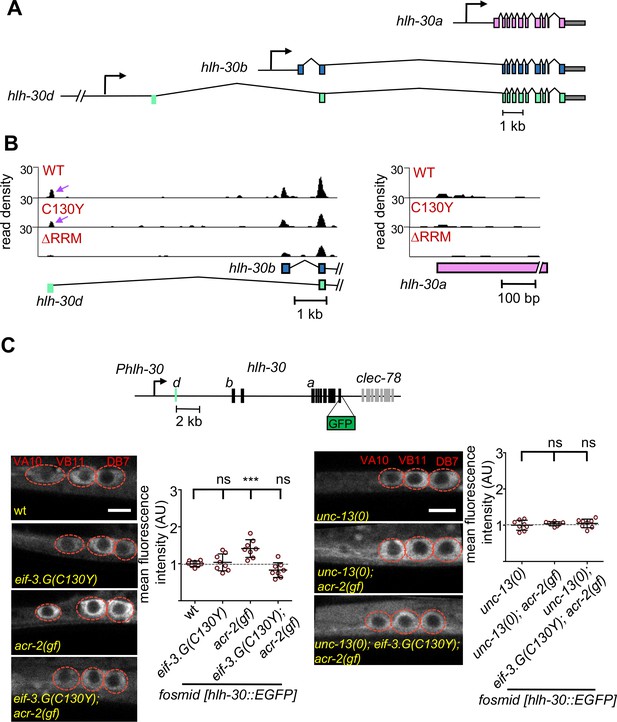
EIF-3.G(C130Y) impairs HLH-30 expression in ACh-MNs of acr-2(gf) animals.
(A) Gene models of hlh-30 isoforms a (pink), b (blue), and d (green), with presumptive promoters for each isoform depicted as right-pointing arrows and the 5′UTR of isoform d in green to the right of its promoter. (B) seCLIP read density tracks of footprints on the 5′ end of hlh-30 isoform b and d (left) and the 5′ end of hlh-30 isoform a (right) in each indicated EIF-3.G dataset. Purple arrows show footprints on the 5′UTR of hlh-30 isoform d. (C) Top: Illustration of the wgIs433 fosmid locus with hlh-30 coding exons in black and 5′UTR of isoform d in green to the right of the promoter. Bottom: Representative single-plane confocal images of the fosmid translational reporter wgIs433[hlh-30::EGFP::3xFLAG] in ACh-MNs in animals of indicated genotypes. Quantification of GFP intensity is shown on the right (n = eight for each genotype). Animals are oriented with anterior to the left. Scale bar = 4 µm. Red dashes indicate labeled ACh-MN soma. Each data point is the average fluorescence intensity quantified from the three ACh-MN soma per animal and normalized to the mean intensity obtained from wgIs433 in the wild type background. Statistics: (***) p< 0.001, (ns) not significant, one-way Anova with Bonferroni’s post hoc test.
-
Figure 6—source data 1
Source data for Figure 6C.
Quantification of relative fluorescence intensity in strains of the indicated genotypes.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig6-data1-v2.xlsx
We found that HLH-30a::GFP intensity was comparable between acr-2(gf) and eif-3.G(C130Y); acr-2(gf) (Figure 6D). These data strengthen the conclusion that enhanced HLH-30 translation in acr-2(gf) partly involves the complex 5′UTR of hlh-30d.
The Neuronal Calcium Sensor protein encoded by ncs-2 promotes calcium-dependent signaling in ACh-MNs (Zhou et al., 2017). We identified strong and specific association of EIF-3.G(WT) and EIF-3.G(C130Y) overlapping the 5′UTR of ncs-2 (Figure 7A). To evaluate NCS-2 expression, we examined a single-copy translational reporter (juSi260) expressing NCS-2::GFP under its endogenous promoter (Zhou et al., 2017; Figure 7B). NCS-2::GFP localized primarily to the neuronal processes in ventral nerve cord, because of the N-terminal myristoylation motif. Quantification of NCS-2::GFP showed that the fluorescence intensity in eif-3.G(C130Y); acr-2(gf) double mutants was significantly reduced, compared to those in wild type, eif-3.G(C130Y), and acr-2(gf) (Figure 7B). ncs-2 mRNA is SL1 trans-spliced, and the mature 5′UTR has 37 nt that is especially abundant in GC nucleotides (47% GC) (Figure 7B). Moreover, the ncs-2 5′UTR sequence is highly conserved with other nematode species (Figure 7—figure supplement 1A). By RNAfold prediction, we found this sequence could form a strong stem-loop structure (∆G = −5.10 kcal/mol). To test if NCS-2::GFP expression was regulated specifically through its 5′UTR, we replaced it with the 5′UTR of eif-3.G, which is comparatively reduced in GC-content (37% GC) and with much less folding stability (∆ = −1.95 kcal/mol) (Figure 7C). The eif-3.G 5′UTR is also less conserved across nematodes compared to that of ncs-2 (Figure 7—figure supplement 1A). We found that the NCS-2::GFP reporter with the 5′UTR of eif-3.G was expressed at similar levels in all genetic backgrounds (Figure 7C).
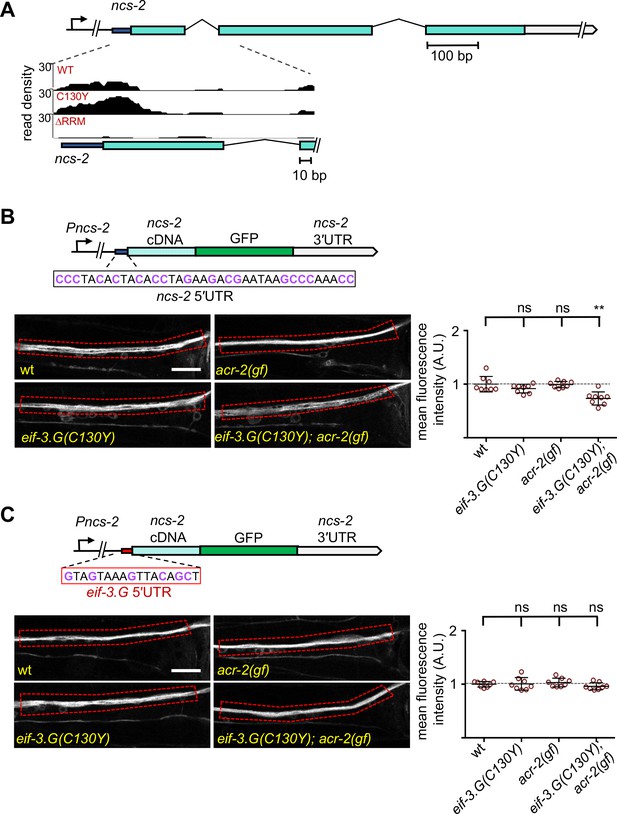
Regulation of NCS-2 expression by EIF-3.G depends on its GC-rich 5′UTR.
(A) Illustration of the ncs-2 genomic region. Dark blue represents 5′UTR, green boxes are coding exons, and gray is the 3′UTR. The inset below shows the read density track of seCLIP footprints on the 5′ region of ncs-2 mRNA. (B) Top: Schematic of the NCS-2(cDNA)::GFP translation reporter, including its 5′UTR (dark blue), driven by the 4 kb promoter Pncs-2. The 5′UTR sequences are GC rich (purple). Bottom: Representative single-plane confocal images of NCS-2::GFP in ventral nerve chord processes in young adult animals of the indicated genotypes. GFP intensity quantification is shown to the right. (C) Top: The ncs-2(5′UTR mutant)::GFP translational reporter has the 5′UTR of eif-3.G (red boxed sequence) replacing the ncs-2 5′UTR, driven by Pncs-2. Bottom: Representative single-plane confocal images of ventral nerve chord processes expressing the NCS-2(5′UTR mutant)::GFP translation reporter in young adult animals of the indicated genotypes. GFP intensity quantification is shown to the right. For (B) and (C), data points are normalized to the average fluorescence intensity of the respective translation reporter in the wild-type background. ROIs used for fluorescence quantification are boxed. Scale bar = 15 µm. Statistics: (**) P< 0.01, (ns) not significant by one-way Anova with Bonferroni’s post hoc test.
-
Figure 7—source data 1
Source data for Figure 7B.
Quantification of relative fluorescence intensity in strains of the indicated genotypes.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig7-data1-v2.xlsx
-
Figure 7—source data 2
Source data for Figure 7C.
Quantification of relative fluorescence intensity in the indicated strains.
- https://cdn.elifesciences.org/articles/68336/elife-68336-fig7-data2-v2.xlsx
To further determine the effects of the ncs-2 5′UTR in protein translation with neuronal type resolution, we generated a reporter in which the GFP coding sequence was fused in-frame after the first four amino acids of NCS-2, which retains the ncs-2 5′UTR but disrupts the myristoylation motif, thereby enabling visualization of NCS-2 in ACh-MNs (Figure 7—figure supplement 1B). Quantification of GFP fluorescence in the cell bodies of VA10, VB11, and DB7 ACh-MN showed significantly reduced expression in eif-3.G(C130Y); acr-2(gf) animals (Figure 7—figure supplement 1B). In contrast, a similar reporter but with the 5′UTR of eif-3.G displayed similar GFP levels in all genetic backgrounds (Figure 7—figure supplement 1C). Therefore, we conclude that eif-3.G regulates NCS-2 expression in the ACh-MNs through a mechanism involving its 5′UTR sequence.
Discussion
The eIF3 complex has been extensively studied for its essential roles in general translation initiation (Cate, 2017; Valášek et al., 2017). However, recent work gives support to the idea that eIF3 is also key to many of the specialized translational control mechanisms needed for tissue plasticity in vivo (Lee et al., 2015; Shah et al., 2016; Rode et al., 2018; Lamper et al., 2020). Our work expands the landscape of eIF3’s regulatory functions, revealing an in vivo role of the eIF3g subunit in stimulating the translation of proteins that mediate neuronal activity changes.
EIF-3.G ensures the efficient translation of mRNAs with GC-rich 5′UTRs
Our study is the first application of seCLIP-seq to map transcriptome-wide protein binding sites in a specific neuronal subtype (ACh-MNs) in C. elegans. With stringent thresholding, we identified 225 genes with strong EIF-3.G occupancy at mRNA 5′ ends. We find that EIF-3.G generally associates with mRNAs harboring long and GC-rich 5′UTRs, implying its RNA-binding function is selective for stimulating translation initiation on 5′ leaders prone to secondary structure or other forms of translation regulation. Our data provide in vivo support to the finding that yeast eIF3g/TIF35 promotes scanning through 5′UTRs with stem-loop structures (Cuchalová et al., 2010). The RRM of yeast eIF3g/TIF35 also promotes re-initiation of 40S ribosomes upon terminating at uORF stop codons on GCN4, thereby allowing efficient induction of genes whose translation is regulated by uORFs (Cuchalová et al., 2010). We did not observe uORFs in the 5′UTRs of ncs-2 or hlh-30, suggesting that at least for these mRNAs, eif-3.G(C130Y) involves reduced scanning through secondary structures or other yet undefined regulatory sequence elements.
It is worth noting that we also found EIF-3.G footprints in 3′UTRs, which could reflect molecular crosstalk between translation initiation and 3′UTR factors, given their proximity in the closed loop translation model (Imataka et al., 1998; Wells et al., 1998). EIF-3.G might anchor the closed-loop mRNA form that stimulates multiple rounds of translation, as was shown to be the case with eIF3h (Choe et al., 2018). It is also possible that EIF-3.G cooperates with 3′UTR interacting factors that regulate gene expression, as several C. elegans translation initiation factors co-immunoprecipitated with the miRISC complex (Zhang et al., 2007) and accumulating evidence supports interplay between various translation factors and RISC proteins that mediate translational repression by microRNAs (Ricci et al., 2013; Fukaya et al., 2014; Gu et al., 2014). Thus, further analysis is needed to examine the biological meaning of EIF-3.G association with 3′UTRs.
The EIF-3.G zinc finger conveys a selective function to translation initiation
The function of the zinc finger of eIF3g remains undefined. Through analysis of EIF-3.G(C130Y), our data provides in vivo insights that the zinc finger contributes to translation efficiency of mRNAs harboring complex 5′UTRs. We establish that EIF-3.G(C130Y) behaves as a genetic gain-of-function mutation without disrupting EIF-3 assembly or otherwise impairing general translation, measured by both polysome levels and the health of cells, tissues, and animals. Additionally, mutating a different cysteine within the zinc finger (C127Y) causes equivalent effects, further strengthening the important role of the entire zinc finger. The effect of EIF-3.G(C130Y) on acr-2(gf) behaviors depends on the RRM, suggesting that association with mRNA after assembly of the pre-initiation complex is required for EIF-3.G(C130Y) function. While we did not observe significant mis-positioning of EIF-3.G-mRNA interactions by EIF-3.G(C130Y), we acknowledge that seCLIP may not have the resolution required to reveal subtle differences in crosslinking sites caused by the C130Y alteration. Together, our data is consistent with a model where EIF-3.G(C130Y) imposes a translational stall after EIF-3 complex assembly and mRNA recruitment. In this view, we speculate that the zinc finger of EIF-3.G mediates interactions with other proteins, such as the ribosome, that critically regulate translation events after mRNA binding. In support of this model, yeast eIF3g/TIF35 was found to directly bind to small ribosomal protein RPS-3, though the molecular basis for mediating this interaction is not identified (Cuchalová et al., 2010). Further studies are required to address the precise molecular mechanism by which the EIF-3.G zinc finger imparts regulatory control over translation initiation.
EIF-3.G targets the translation of mRNAs that modulate neuronal function
Our study was driven by the genetic evidence that eif-3.G(C130Y) ameliorates convulsion behavior caused by the hyperactive ion channel ACR-2(GF). We show that EIF-3.G(C130Y) retains essential EIF-3.G function, yet it alters protein translation on select mRNAs in hyperactive ACh-MNs, as evidenced by its effects on NCS-2 and HLH-30 expression. We previously reported that complete loss-of-function of ncs-2 strongly suppresses acr-2(gf) behaviors to a similar degree as eif-3.G(C130Y) (Zhou et al., 2017). However, 50% reduction of ncs-2 expression does not cause detectable consequences and complete loss-of-function in hlh-30 also has no effects in either wild type or acr-2(gf). Thus, the small reduction of NCS-2 and HLH-30 waged by eif-3.G(C130Y) is unlikely to account for the full extent of phenotypic suppression of acr-2(gf). Our seCLIP data also revealed EIF-3.G interactions with many other genes that differentially impact acr-2(gf) behavior (e.g. neuropeptide flp-18, endopeptidase egl-3) and cholinergic activity (e.g. G proteins goa-1, egl-30). Interestingly, many of the pre-synaptic genes that regulate acr-2(gf) behavior, such as unc-13/Munc13, unc-17/VAChT (Zhou et al., 2013; Takayanagi-Kiya et al., 2016; McCulloch et al., 2017), do not have EIF-3.G footprints. Thus, our data is consistent with a model where eif-3.G(C130Y) ameliorates behaviors of acr-2(gf) through the cumulative changes of select ACh-MN activity regulators.
eif-3.G function may be specialized for activity-dependent gene expression
The eIF3 complex is widely implicated in brain disorders, and deregulated eIF3g is specifically linked to narcolepsy (Gomes-Duarte et al., 2018). However, given the essential role of eIF3 in protein translation in all tissues, investigation of its functions in the nervous system remains limited. Our results reveal that EIF-3.G permits normal activity-dependent protein expression changes, and suggest that dysregulated EIF-3.G might potentiate aberrant neuronal behavior in disorders such as epilepsy by altering the neuronal protein landscape. It is worth noting that pore-lining mutations in human nicotinic receptors that occur at similar positions as acr-2(gf) are causally linked to epilepsy (Xu et al., 2011). We speculate that EIF-3.G may be a potential target for intervention of disorders involving abnormal neurological activity.
In summary, our findings echo the general notion that fine-tuning the activity of essential cellular machinery, such as ribosomes and translation complexes holds the key to balance cellular proteome under dynamic environmental challenges or disease conditions. Emerging studies from cell lines show that stress conditions can induce post-translational modification of eIF3 subunits (Lamper et al., 2020) or cap-independent interactions with mRNAs to modify proteomes (Meyer et al., 2015). Through characterization of the G subunit of eIF3, we reveal the first mechanistic insights into how the eIF3 complex regulates neuronal activity. It is likely that individual eIF3 subunits could each possess unique functions relevant in certain contexts, altogether providing the eIF3 complex with extensive utility to remodel the proteome in response to changing cellular environments.
Materials and methods
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Antibody | anti-FLAG (Rabbit) | Sigma-Aldrich | Cat# F7425, RRID:AB_439687 | WB (1:2000) |
| Antibody | anti-Actin clone C4 (Mouse monoclonal) | MP Biomedicals | Cat# 08691002, RRID:AB_2335304 | WB (1:2000) |
| Antibody | Anti-FLAG M2 Magnetic Beads | Sigma-Aldrich | Cat# M8823, RRID:AB_2637089 | IP |
| Recombinant protein reagent | Cas9-NLS (purified protein) | UC Berkely QB3 | ||
| Genetic reagent (C. elegans) | + | CGC | RRID:CGC_N2 | |
| Genetic reagent (C. elegans) | acr-2(n2420) X | Jospin et al., 2009 | MT6241 | |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II | This work | CZ22197 | Figure 1F |
| Genetic reagent (C. elegans) | eif-3.G(ju1840) II | This work | CZ28494 | Figure 1C |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X | This work | CZ21759 | Figure 1C |
| Genetic reagent (C. elegans) | eif-3.G(ju1840) II; acr-2(n2420) X | This work | CZ28495 | Figure 1C |
| Genetic reagent (C. elegans) | eif-3.G(ju1327) / mnC1 II | This work | CZ22974 | Figure 1F |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx7015 | This work | CZ22976 | Figure 1C |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx7016 | This work | CZ22977 | Figure 1C |
| Genetic reagent (C. elegans) | eif-3.G(ju807) I;acr-2(n2420) X; juEx7045 | This work | CZ23125 | Figure 1E |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; juEx7046 | This work | CZ23126 | Figure 1E |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; juEx7019 | This work | CZ22980 | Figure 1E |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; juEx7020 | This work | CZ22981 | Figure 1E |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; juEx7439 | This work | CZ23791 | Figure 1E |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; juEx7440 | This work | CZ23880 | Figure 1E |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; juEx7021 | This work | CZ22982 | Figure 1E |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; juEx7022 | This work | CZ22983 | Figure 1E |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; juEx8062 | This work | CZ27881 | Figure 1E |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; juEx8063 | This work | CZ27882 | Figure 1E |
| Genetic reagent (C. elegans) | eif-3.G(ju1327) /mnC1 II; acr-2(n2420) X | This work | CZ23310 | Figure 1F |
| Genetic reagent (C. elegans) | eif-3.G(ju807) / eif-3.G(ju1327) II | This work | CZ25714 | Figure 1F |
| Genetic reagent (C. elegans) | eif-3.G(ju807) / eif-3.G(ju1327) II; acr-2(n2420) X | This work | CZ26828 | Figure 1F |
| Genetic reagent (C. elegans) | juSi320 IV | This work | CZ24063 | Figure 2B; Figure 1—figure supplement 1B |
| Genetic reagent (C. elegans) | eif-3.G(ju1327) /mnC1 II; juSi320 IV | This work | CZ24079 | Figure 2A |
| Genetic reagent (C. elegans) | juSi320 IV; acr-2(n2420) X | This work | CZ24729 | Figure 2A |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; juSi320 IV; acr-2(n2420) X | This work | CZ28107 | Figure 2A |
| Genetic reagent (C. elegans) | juSi331 IV | This work | CZ24651 | Figure 2B; Figure 1—figure supplement 1B |
| Genetic reagent (C. elegans) | juSi331 IV; acr-2(n2420) X | This work | CZ24652 | Figure 2A |
| Genetic reagent (C. elegans) | eif-3.G(ju1327) / mnC1 II; juSi331 IV; acr-2(n2420) X | This work | CZ28497 | Figure 2A |
| Genetic reagent (C. elegans) | juIs14 IV | Wang et al., 2017 | CZ631 | |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; juIs14 IV | This work | CZ24161 | Figure 1—figure supplement 2 |
| Genetic reagent (C. elegans) | juIs14 IV; acr-2(n2420) X | McCulloch et al., 2020 | CZ5808 | |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; juIs14 IV; acr-2(n2420) X | This work | CZ8905 | Figure 1—figure supplement 2A |
| Genetic reagent (C. elegans) | nuIs94 | Hallam et al., 2000 | KP2229 | |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; nuIs94 | This work | CZ24021 | Figure 1—figure supplement 2B |
| Genetic reagent (C. elegans) | acr-2(n2420) X; nuIs94 | This work | CZ5815 | Figure 1—figure supplement 2B |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420)X; nuIs94 | This work | CZ24021 | Figure 1—figure supplement 2B |
| Genetic reagent (C. elegans) | eif-3.E(ok2607) I / hT2 I,III; acr-2(n2420) X | This work | CZ27434 | Figure 1—figure supplement 3A |
| Genetic reagent (C. elegans) | eif-3.E(ok2607) I / hT2 I, III; eif-3.G(ju807) II; acr-2(n2420) X | This work | CZ27433 | Figure 1—figure supplement 3A |
| Genetic reagent (C. elegans) | eif-3.H(ok1353) I / hT2 I, III; acr-2(n2420) X | This work | CZ27435 | Figure 1—figure supplement 3A |
| Genetic reagent (C. elegans) | eif-3.H(ok1353) I / hT2 I, III; eif-3.G(ju807) II; acr-2(n2420) X | This work | CZ27436 | Figure 1—figure supplement 3A |
| Genetic reagent (C. elegans) | oxSi39 IV | Qi et al., 2013 | CZ12338 | |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; oxSi39 IV | This work | CZ23854 | Figure 1—figure supplement 3B |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx7056 | This work | CZ23203 | Figure 3 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx7057 | This work | CZ23204 | Figure 3 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx8100 | This work | CZ28152 | Figure 3 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx8101 | This work | CZ28153 | Figure 3 |
| Genetic reagent (C. elegans) | juEx7113 | This work | CZ26777 | Figure 3 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx7114 | This work | CZ23304 | Figure 3 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx7115 | This work | CZ23305 | Figure 3 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx8095 | This work | CZ28066 | Figure 3 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx8096 | This work | CZ28067 | Figure 3 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx8087 | This work | CZ28057 | Figure 3 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx8088 | This work | CZ28058 | Figure 3 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx8089 | This work | CZ28064 | Figure 3 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx8090 | This work | CZ28065 | Figure 3 |
| Genetic reagent (C. elegans) | unc-119(tm4063) III; wgIs433 | Sarov et al., 2006 | OP433 | |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; unc-119(tm4063)III; wgIs433 | This work | CZ28145 | Figure 6C |
| Genetic reagent (C. elegans) | acr-2(n2420) X; unc-119(tm4063) III; wgIs433 | This work | CZ27913 | Figure 6C |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; unc-119(tm4063) III; acr-2(n2420) X; wgIs433 | This work | CZ27914 | Figure 6C |
| Genetic reagent (C. elegans) | sqIs17 | Dittman and Kaplan, 2006 | MAH240 | |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; sqIs17 | This work | CZ28334 | Figure 6—figure supplement 1 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; sqIs17 | This work | CZ28212 | Figure 6—figure supplement 1 |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; sqIs17 | This work | CZ28218 | Figure 6—figure supplement 1 |
| Genetic reagent (C. elegans) | unc-13(s69) I; wgIs433 | This work | CZ28491 | Figure 6C |
| Genetic reagent (C. elegans) | unc-13(s69) I; acr-2(n2420) X; wgIs433 | This work | CZ28492 | Figure 6C |
| Genetic reagent (C. elegans) | unc-13(s69) I; eif-3.G(ju807); acr-2(n2420) X; wgIs433 | This work | CZ28493 | Figure 6C |
| Genetic reagent (C. elegans) | juSi260 ncs-2(tm1943) I | Zhou et al., 2017 | CZ22459 | |
| Genetic reagent (C. elegans) | juSi260 ncs-2(tm1943) I; eif-3.G(ju807) II | This work | CZ23225 | Figure 7B |
| Genetic reagent (C. elegans) | juSi260 ncs-2(tm1943) I; acr-2(n2420) X | This work | CZ22345 | Figure 7B |
| Genetic reagent (C. elegans) | juSi260 ncs-2(tm1943) I; eif-3.G(ju807) II; acr-2(n2420) X | This work | CZ28110 | Figure 7B |
| Genetic reagent (C. elegans) | juSi391 ncs-2(tm1943) I | This work | CZ28213 | Figure 7C |
| Genetic reagent (C. elegans) | juSi391 ncs-2(tm1943) I; eif-3.G(ju807) II | This work | CZ28340 | Figure 7C |
| Genetic reagent (C. elegans) | juSi391 ncs-2(tm1943) I;acr-2(n2420) X | This work | CZ28252 | Figure 7C |
| Genetic reagent (C. elegans) | juSi391 ncs-2(tm1943) I;eif-3.G(ju807) II; acr-2(n2420) X | This work | CZ28253 | Figure 7C |
| Genetic reagent (C. elegans) | juSi392 ncs-2(tm1943) I | This work | CZ28277 | Figure 7—figure supplement 1B |
| Genetic reagent (C. elegans) | juSi392 ncs-2(tm1943) I;eif-3.G(ju807) II | This work | CZ28312 | Figure 7—figure supplement 1B |
| Genetic reagent (C. elegans) | juSi392 ncs-2(tm1943) I; acr-2(n2420) X | This work | CZ28291 | Figure 7—figure supplement 1B |
| Genetic reagent (C. elegans) | juSi392 ncs-2(tm1943) I; eif-3.G(ju807) II; acr-2(n2420) X | This work | CZ28292 | Figure 7—figure supplement 1B |
| Genetic reagent (C. elegans) | juSi393 ncs-2(tm1943) I | This work | CZ28278 | Figure 7—figure supplement 1C |
| Genetic reagent (C. elegans) | juSi393 ncs-2(tm1943) I; eif-3.G(ju807) II | This work | CZ28311 | Figure 7—figure supplement 1C |
| Genetic reagent (C. elegans) | juSi393 ncs-2(tm1943) I; acr-2(n2420) X | This work | CZ28293 | Figure 7—figure supplement 1C |
| Genetic reagent (C. elegans) | juSi393 ncs-2(tm1943) I; eif-3.G(ju807) II; acr-2(n2420) X | This work | CZ28294 | Figure 7—figure supplement 1C |
| Genetic reagent (C. elegans) | juEx2045 | --- | CZ9635 | |
| Genetic reagent (C. elegans) | hlh-30(tm1978) IV | CGC | CZ23321 | |
| Genetic reagent (C. elegans) | hlh-30(tm1978) IV; acr-2(n2420) X | This work | CZ28174 | Related to Figure 6C |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; hlh-30(tm1978) IV; acr-2(n2420) X | This work | CZ28175 | Related to Figure 6C |
| Genetic reagent (C. elegans) | eif-3.G(ju1327) II /mnC1; juSi363 IV; acr-2(n2420) X | This work | CZ26759 | Related to Figure 4—figure supplement 1A |
| Genetic reagent (C. elegans) | eif-3.G(ju1327) II / mnC1 II; juSi366 IV; acr-2(n2420) X | This work | CZ26760 | Related to Figure 4—figure supplement 1A |
| Genetic reagent (C. elegans) | juSi364 IV; acr-2(n2420) X | This work | CZ26494 | Figure 4—figure supplement 1A |
| Genetic reagent (C. elegans) | eif-3.G(ju807)II juSi364 IV; acr-2(n2420) X | This work | CZ26243 | Related to Figure 4—figure supplement 1A |
| Genetic reagent (C. elegans) | juSi365 IV | This work | CZ26588 | Related to Figure 4—figure supplement 1A |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; juSi365 IV; acr-2(n2420) X | This work | CZ26565 | Figure 4—figure supplement 1A |
| Genetic reagent (C. elegans) | juSi365 IV; acr-2(n2420) X | This work | CZ26566 | Related to Figure 4—figure supplement 1A |
| Genetic reagent (C. elegans) | juSi368 IV | This work | CZ26656 | Related to Figure 4—figure supplement 1A |
| Genetic reagent (C. elegans) | juSi368 IV; acr-2(n2420) X | This work | CZ26623 | Figure 4—figure supplement 1A |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; juSi368 IV; acr-2(n2420) X | This work | CZ26480 | Related to Figure 4—figure supplement 1A |
| Genetic reagent (C. elegans) | wgIs506 | Sarov et al., 2006 | OP506 | Supplementary file 1 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; wgIs506 | This work | CZ27926 | Supplementary file 1 |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; wgIs506 | This work | CZ27927 | Supplementary file 1 |
| Genetic reagent (C. elegans) | dhc-1::GFP(it45) I | Lapierre et al., 2013 | OD2955 | Supplementary file 1 |
| Genetic reagent (C. elegans) | dhc-1::GFP(it45) I; acr-2(n2420) X | This work | CZ27858 | Supplementary file 1 |
| Genetic reagent (C. elegans) | dhc-1::GFP(it45) I; eif-3.G(ju807) II; acr-2(n2420) X | This work | CZ27859 | Supplementary file 1 |
| Genetic reagent (C. elegans) | wgIs432 | Sarov et al., 2006 | OP432 | Supplementary file 1 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; wgIs432 | This work | CZ27915 | Supplementary file 1 |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; wgIs432 | This work | CZ28021 | Supplementary file 1 |
| Genetic reagent (C. elegans) | wgIs638 | Sarov et al., 2006 | OP638 | Supplementary file 1 |
| Genetic reagent (C. elegans) | unc-119(tm4063) III;acr-2(n2420) X; wgIs638 | This work | CZ28108 | Supplementary file 1 |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; wgIs638 | This work | CZ27916 | Supplementary file 1 |
| Genetic reagent (C. elegans) | let-607(tm1423) I; unc-119(ed3) III; vrIs121 | Sarov et al., 2006 | YL651 | Supplementary file 1 |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; let-607(tm1423) I; unc-119(ed3) III; vrIs121 | This work | CZ28143 | Supplementary file 1 |
| Genetic reagent (C. elegans) | acr-2(n2420) X; let-607(tm1423) I;unc-119(ed3) III; vrIs121 | This work | CZ28119 | Supplementary file 1 |
| Genetic reagent (C. elegans) | eif-3.G(ju807) II; acr-2(n2420) X; let-607(tm1423) I; unc-119(ed3) III; vrIs121 | This work | CZ28111 | Supplementary file 1 |
| Genetic reagent (C. elegans) | juIs172 | CGC | EE86 | |
| Genetic reagent (C. elegans) | egl-30(md186) I;dpy-20(e1282ts) IV; syIs105 | CGC | PS4263 | |
| Genetic reagent (C. elegans) | juEx7964 | McCulloch et al., 2020 | CZ27420 | |
| Genetic reagent (C. elegans) | acr-2(n2420) X; juEx7964 | McCulloch et al., 2020 | CZ27217 | |
| Genetic reagent (C. elegans) | eif-3.G(C130Y) II; acr-2(n2420) X; juEx7964 | This work | CZ28109 | Supplementary file 1 |
| Recombinant DNA reagent (plasmid) | pCZGY2729 | Andrusiak et al., 2019 | RRID:Addgene_135096 | Site-specific insertion using CRISPR/Cas9 editing of C. elegans ChrIV |
| Recombinant DNA reagent (plasmid) | pCZGY2750 | Andrusiak et al., 2019 | RRID:Addgene_135094 | Expresses Cas9 and sgRNA for editing of C. elegans ChrIV |
| Recombinant DNA reagent (plasmid) | pCZGY2727 | This work | Site-specific insertion using CRISPR/Cas9 editing of C. elegans ChrI | |
| Recombinant DNA reagent (plasmid) | pCZGY2748 | This work | Expresses Cas9 and sgRNA for editing of C. elegans ChrI |
C. elegans genetics
Request a detailed protocolAll C. elegans strains were maintained at 20°C on nematode growth media (NGM) plates seeded with OP50 bacteria (Brenner, 1974). Compound mutants were generated using standard C. elegans genetic procedures and strain genotypes are listed in key resource table and Supplementary file 1. Primers for genotyping are in Supplementary file 2.
Identification of eif-3.G(ju807)
Request a detailed protocolWe employed a custom workflow on the GALAXY platform to identify SNPs unique to strains containing suppressor mutations of acr-2(gf), compared to the N2 reference strain (McCulloch et al., 2017). Following SNP mapping using genetic recombinants, we located ju807 to eif-3.G on chromosome II. We then performed transgenic expression experiments and found that both over-expression and single-copy expression of eif-3.G(+) in ju807; acr-2(gf) animals restored convulsions.
Quantification of convulsion behavior
Request a detailed protocolConvulsions were defined as contractions that briefly shorten animal body length, as previously reported (Jospin et al., 2009; Video 2). L4 larvae were cultured overnight on fresh NGM plates seeded with OP50 bacteria at 20°C. The following day, each young adult was moved to a fresh seeded plate, and after climatized for 90 s, convulsions were counted over a subsequent 90 s. The average convulsion frequency represented data over 60 s. All statistical tests were performed using GraphPad Prism6 software and p-values <0.05 were considered significant.
CRISPR-mediated genome editing
Request a detailed protocolWe used a previously described method (Dickinson et al., 2013) with minor modifications to generate eif-3.G(ju1327) deletion allele. Briefly, we designed sgRNA target sequence CAATTCACAAGAAATCGCGC, and cloned it into a Cas9-sgRNA expression construct pSK136 (derived from pDD162, with site-directed mutagenesis). A DNA mixture containing 50 ng/µl pSK136, 1 ng/µl Pmyo-2::mCherry (pCFJ90), and 50 ng/µl 100 bp ladder (Invitrogen, Carlsbad, CA) was microinjected into N2 adults. We screened F2 progenies from F1 animals carrying the co-injection marker for deletions in eif-3.G and identified a 19 bp deletion, designated ju1327. Heterozygous ju1327 was twice outcrossed to N2 and then crossed to the mnC1 balancer for stable strain maintenance (CZ22974).
The eif-3.G(ju1840) allele, which causes a C127Y mutation in EIF-3.G, was generated using a co-CRISPR genome editing method with unc-58(gf) as a selection marker (Paix et al., 2017). We microinjected a Cas9 complex containing the sgRNA sequence GGTCGTTTCCTTTGCAATGA, a DNA repair template incorporating TAT (encoding Y127) in place of TGC (C127), and a previously described sgRNA and repair template for unc-58(gf) into N2 adult hermaphrodites. We genotyped for eif-3.G(ju1840) among heterozygous unc-58(gf) F1 progeny and subsequently identified F2 animals homozygous for eif-3.G(ju1840) and unc-58(+).
Molecular biology and transgenesis
Request a detailed protocolAll transgene constructs were cloned using the Gateway cloning system (Invitrogen, Carlsbad, CA) or Gibson Assembly (NEB, Ipswich, MA), unless otherwise noted. Primers used in their construction are detailed in Supplementary file 3. For single-copy insertion transgenes, we used a previously described CRISPR/Cas9 method to integrate a single genomic copy on chromosome IV (Andrusiak et al., 2019). For extrachromosomal transgenes, we microinjected a DNA mixture containing 2 ng/µl transgene plasmid, 2.5 ng/µl pCFJ90(Pmyo-2::mCherry), and 50 ng/µl 100 bp ladder (Invitrogen, Carlsbad, CA) into young adults, following standard procedure (Mello et al., 1991).
To generate the eif-3.G(+) or eif-3.G(C130Y) genomic constructs (pCZGY3006 or pCZGY3007), we amplified a 2223 bp region from genomic DNA of N2 or CZ21759 eif-3.G(C130Y); acr-2(gf), respectively, which includes 1714 bp upstream of the start codon of isoform A (F22B5.2a.1) and 331 bp downstream of the stop codon, and cloned the amplicon into the PCR8 vector (Invitrogen, CA).
To generate all eif-3.G cDNA expression clones, we made mixed-stage cDNA libraries with poly-dT primer for N2 or CZ21759 using Superscript III (ThermoFisher Scientific, San Diego, CA). We then amplified and eif-3.G cDNA using primers for the SL1 trans-splice leader (YJ74) and eif-3.G isoform A 3′UTR (YJ11560) and Phusion polymerase (Thermo Fisher Scientific, San Diego, CA). The cDNA clones in PCR8 vector were then used to generate tissue-specific expression constructs using Gateway cloning destination vectors (pCZGY1091 for Punc-17β, pCZGY925 for Pmyo-3, pCZGY66 for Prgef-1, and pCZGY80 for Punc-25).
We used PCR site-directed mutagenesis, in which the nucleotide changes are introduced by the primers to generate the Prgef-1::eif-3.G(∆RRM) and Prgef-1::eif-3.G(C130Y ∆RRM) constructs (pCZGY3026 and pCZGY3027, respectively) with primers YJ11561 and YJ11562 on the templates pCZGY2715 and pCZGY2716, respectively. The Prgef-1::eif-3.G(RFF/AAA) construct (pCZGY3512) was generated by two rounds site directed PCR mutagenesis on pCZGY3010, first using primers YJ12463 and YJ12464, then primers YJ12465 and YJ12466. To generate Pref-1::eif-3.I(+) (pCZGY3508), we amplified eif-3.I cDNA from N2 cDNA libraries using primers YJ12453 and YJ12454, and used Gibson Assembly to clone into the pCZGY66 backbone containing Prgef-1. We then performed site-directed mutagenesis on pCZGY3508 using primers YJ12457 and YJ12458 to generate the Prgef-1::eif-3.I(Q252R) construct (pCZGY3509).
We generated the GFP::EIF-3.G clones pCZGY3018 and pCZGY3019 via Gibson assembly, using eif-3.G(+) or eif-3.G(C130Y) cDNA amplified using primers YJ12604 and YJ12605, and the GFP-coding DNA amplified using primers YJ12602 and YJ12603.
To generate Punc-17β::EIF-3.G::3xFLAG::SL2::GFP constructs (pCZGY3538 for WT, pCZGY3539 for C130Y, and pCZGY3540 for ∆RRM) used in seCLIP experiments, Punc-17β promoter was amplified from pCZGY1091 using primers YJ12164 and YJ12418, each eif-3.G cDNA (wild type, C130Y, or ∆RRM) was amplified with an N-terminal 3xFLAG sequence from subclones using the primers YJ12419 and YJ12420, SL2 trans-splice sequence was amplified from N2 genomic DNA using primers YJ12421 and YJ12422, and GFP was amplified from pCZGY3018 using primers YJ12423 and YJ12424. These fragments were then Gibson Assembled into the pCZGY2729 backbone (RRID:Addgene_135096), which facilitates CRISPR/Cas9 single copy insertion on chromosome IV (Andrusiak et al., 2019).
All ncs-2 transgenes were similarly cloned using primers for Gibson assembly into pCZGY2727. To generate the Pncs-2::5′UTR mutant::ncs-2 cDNA construct (pCZGY3526), we amplified Pncs-2 from N2 gDNA using primers YJ12554 and YJ12555. A fragment containing SL1 trans-spliced eif-3.G 5′UTR incorporated in the forward primer, ncs-2 cDNA, GFP, and the ncs-2 3′UTR was amplified from CZ22459 gDNA using primers YJ12556 and YJ12557. The Pncs-2::GFP(+) construct (pCZGY3533) was cloned by amplifying Pncs-2 through the first four codons of ncs-2 CDS from N2 gDNA using primers YJ12554 and YJ12579, and GFP and the ncs-2 3′UTR from CZ22459 gDNA using YJ12580 and YJ12557. The Pncs-2::5′UTR mutant::GFP construct (pCZGY3534) was cloned by amplifying Pncs-2 from N2 gDNA using primers YJ12554 and YJ12555, and the eif-3.G 5′UTR, the first four codons of the ncs-2 CDS, GFP, and the ncs-2 3′UTR from CZ22459 gDNA using primers YJ12581 and YJ12557.
Fluorescence microscopy and GFP intensity quantification
Request a detailed protocolL4 or young adult animals were immobilized in 1 mM levamisole in M9 and mounted on microscope slides with 2% agar. All images were collected on a Zeiss LSM800 confocal microscope, unless specified, with identical image acquisition settings: 1.25 µm pixel size with 0.76 µs pixel time, 50 µm pinhole, with genotype-blinding to observer when possible. The positions of VA10, VB11, and DB7 cholinergic motor neurons were identified using juEx2045(Pacr-2-mCherry), based on their stereotypical patterning in the posterior ventral nerve cord. These neurons were chosen for quantification because they were consistently visible in single focal plane images. All quantification of GFP intensity in these neurons was performed using the Integrated Density function in ImageJ (Schindelin et al., 2012). We acquired the mean integrated density from the VA10, VB11, and DB7 cell bodies, subtracted background intensity from an equivalent area, and the resulting values were then normalized to the mean area of the cell bodies of the same animal. We similarly quantified fluorescence intensities in the ventral nerve cord of animals expressing GFP-tagged full-length ncs-2 cDNA, except integrated densities were obtained from one ROI per image (red boxes in Figure 7B and C). All data was normalized to the mean fluorescence intensity of the transgene in the wildtype background. All statistical analysis was performed with GraphPad Prism6 software.
Axon commissures, observed as fluorescent structures extending from the ventrally located neuron cell body to the dorsal body wall, shown in Figure 1—figure supplement 2A were visualized with juIs14[Pacr-2::GFP] and manually quantified. Imaging shown in Figure 1—figure supplement 2B was performed using a Zeiss Axioplan two microscope installed with Chroma HQ filters and a 63x objective lens. Synaptic puncta labeled by nuIs94[SNB-1::GFP], were manually quantified in the region anterior to the ventral nerve chord between VD6 and VD7.
Polysome profiling
Request a detailed protocolWe prepared C. elegans lysates and sucrose gradients using the protocol described in Ding and Grosshans, 2009. To synchronize animals, gravitated adults were treated with 20% Alkaline Hypochlorite Solution and embryos were plated on four 30 cm NGM plates seeded with OP50, and grown to the L4 stage at 20°C. Approximately 200 µl packed L4 C. elegans were harvested by centrifugation in M9 media at 1500 RPM, washed three times in ice-cold M9 media supplemented with 1 mM cycloheximide, then once more in lysis buffer base solution (140 mM KCl, 20 mM Tris-HCl (pH 8.5), 1.5 mM MgCl2, 0.5% NP-40, 1 mM DTT, 1 mM cycloheximide) followed by snap freezing in liquid nitrogen. The frozen pellets were resuspended in 450 µl lysis buffer (140 mM KCl, 20 mM Tris-HCl (pH 8.5), 1.5 mM MgCl2, 0.5% NP-40, 2% PTE, 1% sodium deoxycholate, 1 mM DTT, 1 mM cycloheximide, 0.4 units/µl RNAsin) and crushed to a fine powder with a mortar and pestle pre-cooled with liquid nitrogen. Protein lysate concentrations were then determined using a Bradford assay (Bio-Rad, Hercules, CA). Fifteen to 60% sucrose gradients were prepared in 89 mm polypropylene centrifuge tubes (Beckman Coulter) using standard settings on a Foxy Jr. density gradient fractionation system (Teledyne ISCO, Lincoln, NE) and lysate volumes corresponding to equal protein amounts between samples were loaded on top of the gradients. Loaded gradients were then spun in an Optima L-80 ultracentrifuge (Beckman Coulter) at 36,000 rpm at 4°C for 3 hr. Fractions were then collected and RNA absorbance was continuously acquired using a UA-6 detector (Teledyne ISCO, Lincoln, NE) with a 70% sucrose chase solution. We calculated the area under the curve (AUC) for monosome (80S) and polysome absorbance traces using the Simpson’s rule method in SciPy (Virtanen et al., 2020) and used the AUC values to calculate the polysome to monosome ratios.
Western blot analysis
Request a detailed protocolA total of 500 µl of mixed staged worms were resuspended in lysis buffer (140 mM KCl, 20 mM Tris-HCl (pH 8.5), 1.5 mM MgCl2, 0.5% NP-40, 1% sodium deoxycholate, 1 mM DTT) supplemented with protease inhibitors (Complete Ultra Tablets, Roche), frozen in liquid nitrogen, and crushed to a fine powder. The lysates were clarified by centrifugation at max speed in a tabletop centrifuge and protein levels were quantified using a Bradford assay (Bio-Rad, Hercules, CA). The resulting protein lysates were then boiled in Laemmli buffer with 10% 2-mercaptoethanol, run on SDS-PAGE gels, and transferred to PVDF blots, which were probed with anti-FLAG (F7425, RRID:AB_439687) or anti-Actin (clone C4, RRID:AB_2335304) antibodies.
seCLIP library preparation and sequencing
Request a detailed protocolWe performed single-end enhanced CrossLink and ImmunoPrecipitation (seCLIP) experiments according to the published protocol in Van Nostrand et al., 2017, with the following adjustments to ensure efficient immunoprecipitation yield from C. elegans lysates. Mixed stage animals were grown on ~12 NGM plates (30 cm) and washed twice with M9, spinning at 1500 rpm between washes. Animals were then resuspended in 5 ml M9 media and rocked on a rotator for 10 min to remove gut bacteria, followed by one more wash with M9 at 1500 rpm. The animals were spread on one NGM plate (30 cm) and then UV-crosslinked with a Spectrolinker XL-1000 (Spectronics, New Cassel, NY) using energy setting 3 kJ/m2 according to Broughton and Pasquinelli, 2013. Afterwards, animals were resuspended in 4 ml lysis buffer [150 mM NaCl, 1 M HEPES, 100 mM DTT, 6.25 µl RNAsin (Promega) per 10 ml, 10% glycerol, 10% Triton X-100, one protease inhibitor tablet per 10 ml] and split into two tubes for each replicate. The resuspension was disrupted on an XL-2000 Sonicator (QSonica, Newtown, CT) with seven pulses (powersetting = 11, 10 s each, 50 s on ice in between) and immediately spun at 4750 RPM for 5 min at 4°C. All subsequent steps, beginning with RNAse A treatment of the supernatant, was performed according to the seCLIP protocol (Van Nostrand et al., 2017), except that high-salt and low-salt wash buffers were replaced with a single buffer (2M NaCl, 1M HEPES, 30% glycerol, 1% Triton X-100, one protease inhibitor tablet per 10 mL) optimized for anti-FLAG RNA IP from C. elegans lysates (Blazie et al., 2015). Immunoprecipitation was performed with anti-FLAG beads (Sigma, RRID:AB_2637089). cDNA libraries were prepared from both the immunoprecipitated mRNA (CLIP) as well as the sample before immunoprecipitation (INPUT), such that crosslink sites can be defined by read enrichment in the CLIP sample over input as described (Van Nostrand et al., 2017). seCLIP libraries were validated using the D1000 high sensitivity screen tape system (Agilent, La Jolla, CA) and quantified using a Qubit instrument (Thermo Fisher, San Diego, CA) before pooling and sequencing on HiSeq4000 (Illumina, San Diego, CA) at the IGM Genomics Center, University of California San Diego.
seCLIP sequence mapping
Request a detailed protocolAfter demultiplexing barcoded reads, we used the CLIPPER software pipeline (Lovci et al., 2013) to trim barcodes, remove PCR duplicate reads, filter reads mapping to repetitive elements, and map the remaining reads to the C. elegans reference genome (ce10). The total number of uniquely mapped reads obtained after filtering is in Supplementary file 4. A large proportion of reads obtained from the ∆RRM and IgG samples mapped to repetitive elements and were discarded, explaining the smaller number of uniquely mapped reads in these samples. In seCLIP, RNA-binding sites are defined as read clusters enriched in the crosslink immunoprecipitated sample (CLIP) over the input control (INPUT) (Van Nostrand et al., 2017), which are comprehensively identified across each dataset using CLIPPER. Read clusters were reproducibly identified from independent biological replicates of seCLIP, except in the ∆RRM control reflecting background, supporting the specificity of our data (Figure 4—figure supplement 1).
EIF-3.G footprint identification from mapped seCLIP reads
Request a detailed protocolWe defined EIF-3.G footprints as seCLIP read clusters appearing in both replicates with 20 reads and 1.5 fold-change enrichment over the INPUT control in at least one replicate. Footprints matching these criteria in the IgG (no transgene) and the EIF-3.G(∆RRM) control samples were considered background and subtracted from the EIF-3.G(WT) and EIF-3.G(C130Y) datasets (Supplementary files 5 and 6). We annotated footprints to their gene features (eg. 5′UTR, CDS) using a script (Yee, 2021; https://github.com/byee4/annotator) that overlaps read clusters with the C. elegans genome annotation WS235. We grouped all clusters annotated in the CDS and 5'UTR into one category (5'UTR proximal), since clusters mapping in CDS were almost always located within 200nts of a 5'UTR (Figure 4D).
Gene ontology and KEGG pathway analysis
Request a detailed protocolGO analysis was performed using 225 EIF-3.G target genes as input to the biological process annotation set within the Gene Ontology Resource tool (Ashburner et al., 2000). A total 211 gene names were recognized by the database and GO term enrichment was defined using a threshold of p< 0.05. Pathway analysis of EIF-3.G target genes was performed using the Kyoto Encyclopedia of Genes and Genomes (KEGG) annotation tool within the DAVID bioinformatics resource (Jiao et al., 2012) using default settings.
Analysis of activity-dependent expression changes among EIF-3.G target mRNAs in cholinergic neurons
Request a detailed protocolWe studied activity-dependent transcript expression changes among the EIF-3.G target genes (n = 225) by re-analyzing the cholinergic neuron-specific transcriptomes reported in McCulloch et al., 2020 using the Galaxy platform (Afgan et al., 2018). We downloaded raw FASTA reads from transcriptome sequencing of wild type and acr-2(gf) animals (n = two replicates each; accession #’s SRR10320705, SRR10320706, SRR10320707, SRR10320707) and mapped them to the C. elegans reference genome (ce10) using BWA (Li and Durbin, 2009). Differential expression among the EIF-3.G target genes was quantified using Feature Counts (Liao et al., 2014) and DeSeq2 (Love et al., 2014).
in silico analysis of 5′UTR sequence features, secondary structure, and conservation
Request a detailed protocolWe downloaded all C. elegans transcript 5'UTRs (WS271) from Parasite Biomart (Howe et al., 2016), and calculated 5′UTR lengths as the sequence between the 5' distal end and the start codon of each transcript. To have meaningful length calculation, we only considered 5′UTRs annotated with at least 10nt and restricted our analysis to the longest 5′UTR isoform for each gene to avoid considering multiple transcripts of the same gene. By these criteria we identified 5′UTRs for 10,962 WS271 protein coding transcripts and 179 transcripts with EIF-3.G footprints. We used the same criteria to determine features of the acr-2(gf) cholinergic transcriptome 5′UTRs (McCulloch et al., 2020) for the analysis shown in Figure 4E–F.
For the analysis shown in Figure 4—figure supplement 2D–E, the genomic coordinates of human gene 5′UTRs were downloaded from Ensembl and used to obtain 5′UTR sequences from the human genome reference sequence (hg38). eIF3g footprints from HEK293 cells were previously described (Lee et al., 2015). We defined our analysis of 5′UTR sequences using the same criteria described for C. elegans and the data show the comparison between 5′UTRs of 255 genes with human eIF3g footprints and 19,914 total genes in the human genome annotation (hg38).
To calculate GC-enrichment, we used BEDTools (Quinlan and Hall, 2010) to generate a FASTA of 5′UTR sequences from their genomic coordinates and used Biopython (Cock et al., 2009; https://github.com/biopython/biopython) to calculate the total %GC in their sequences (Figure 4F) as well as %GC within 10nt bins incremented from the start codon ATG for the analysis shown in Figure 4—figure supplement 2C.
To predict secondary structures of the 5′ ends of hlh-30d, ncs-2, and eif-3.G mRNAs, we used the RNAfold Web Server (Gruber et al., 2008) with default settings. To better understand the contribution of gene-specific 5'UTR sequences, we excluded the SL1 sequences of ncs-2 and eif-3.G from folding predictions. The free energies (∆G) for each sequence reported in our results were derived from the reported thermodynamic ensemble. Data showing conservation of eif-3.G and ncs-2 5′UTR sequences compared with 135 nematode species (phyloP135way scores) was obtained from the UCSC Genome Browser with the genomic position along the sequence of each 5′UTR as input.
Data availability
Raw and processed seCLIP datasets from this study have been uploaded to the Gene Expression Omnibus (GEO) under accession number GSE152704.
-
NCBI Gene Expression OmnibusID GSE152704. seCLIP of C. elegans EIF-3.G in the cholinergic motor neurons.
-
NCBI Gene Expression OmnibusID GSE139212. Neuronal transcriptome analyses reveal novel neuropeptide modulators of excitation and inhibition imbalance in C. elegans.
References
-
The galaxy platform for accessible, reproducible and collaborative biomedical analyses: 2018 updateNucleic Acids Research 46:W537–W544.https://doi.org/10.1093/nar/gky379
-
A global analysis of C. elegans trans-splicingGenome Research 21:255–264.https://doi.org/10.1101/gr.113811.110
-
Human eIF3: from 'blobology' to biological insightPhilosophical Transactions of the Royal Society B: Biological Sciences 372:20160176.https://doi.org/10.1098/rstb.2016.0176
-
The RNA recognition motif of eukaryotic translation initiation factor 3g (eIF3g) is required for resumption of scanning of posttermination ribosomes for reinitiation on GCN4 and together with eIF3i stimulates linear scanningMolecular and Cellular Biology 30:4671–4686.https://doi.org/10.1128/MCB.00430-10
-
Characterization of the p33 subunit of eukaryotic translation initiation factor-3 from Saccharomyces cerevisiaeThe Journal of biological chemistry 274:8546–8553.https://doi.org/10.1074/jbc.274.13.8546
-
EIF3G is associated with narcolepsy across ethnicitiesEuropean journal of human genetics : EJHG 23:1573–1580.https://doi.org/10.1038/ejhg.2015.4
-
WormBase 2016: expanding to enable helminth genomic researchNucleic acids research 44:D774–D780.https://doi.org/10.1093/nar/gkv1217
-
Functional 5' UTR mRNA structures in eukaryotic translation regulation and how to find themNature reviews. Molecular cell biology 19:158–174.https://doi.org/10.1038/nrm.2017.103
-
Rbfox proteins regulate alternative mRNA splicing through evolutionarily conserved RNA bridgesNature structural & molecular biology 20:1434–1442.https://doi.org/10.1038/nsmb.2699
-
Collybistin binds and inhibits mTORC1 signaling: a potential novel mechanism contributing to intellectual disability and autismEuropean journal of human genetics : EJHG 24:59–65.https://doi.org/10.1038/ejhg.2015.69
-
Novel Mutations in Synaptic Transmission Genes Suppress Neuronal Hyperexcitation in Caenorhabditis elegansG3: Genes, Genomes, Genetics 7:2055–2063.https://doi.org/10.1534/g3.117.042598
-
Selective clearance of aberrant tau proteins and rescue of neurotoxicity by transcription factor EBEMBO molecular medicine 6:1142–1160.https://doi.org/10.15252/emmm.201303671
-
Hyperactivation of B-type motor neurons results in aberrant synchrony of the Caenorhabditis elegans motor circuitThe Journal of Neuroscience 33:5319–5325.https://doi.org/10.1523/JNEUROSCI.4017-12.2013
-
miRNA repression of translation in vitro takes place during 43S ribosomal scanningNucleic acids research 41:586–598.https://doi.org/10.1093/nar/gks1076
-
UNC-13 is required for synaptic vesicle fusion in C. elegansNature neuroscience 2:959–964.https://doi.org/10.1038/14755
-
Synaptic functionWormBook : The Online Review of C. elegans Biology 1:14.https://doi.org/10.1895/wormbook.1.69.1
-
Fiji: an open-source platform for biological-image analysisNature methods 9:676–682.https://doi.org/10.1038/nmeth.2019
-
Embraced by eIF3: structural and functional insights into the roles of eIF3 across the translation cycleNucleic Acids Research 45:10948–10968.https://doi.org/10.1093/nar/gkx805
Article and author information
Author details
Funding
National Institutes of Health (NS R37 035546)
- Yishi Jin
University of California, San Diego (NS007220)
- Stephen M Blazie
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Acknowledgements
We thank Brian Yee, Eric Van Nostrand, Gabriel Pratt, and Gene Yeo for advice on adapting the seCLIP protocol to C. elegans and technical support with seCLIP data analysis; Timothy Shaw and Jens Lykke-Andersen for sharing equipment and invaluable assistance with polysome profile analysis; Yan Zhao, Ann Zhou and Ippei Ozaki for assistance in constructing expression clones and strains; Yunbo Li for advice on protein analysis; Malena Hansen, Josh Kaplan, and Keming Zhou for transgenes, Kenneth Miller for Punc-17β plasmid. Some strains used in this study were provided by the Caenorhabditis Genetics Center, which is funded by NIH Office of Research Infrastructure Programs (P40 OD010440). We acknowledge WormBase and UCSC Genome databases for genomic information resource. We thank Andrew D Chisholm and members of our laboratory for critical reading of the manuscript. S B was a trainee on the UCSD T32 training grant (NS007220). This work is funded by NIH grant (NS R37 035546 to Y J).
Copyright
© 2021, Blazie et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 2,006
- views
-
- 283
- downloads
-
- 24
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Citations by DOI
-
- 24
- citations for umbrella DOI https://doi.org/10.7554/eLife.68336






