Tadr is an axonal histidine transporter required for visual neurotransmission in Drosophila
Figures
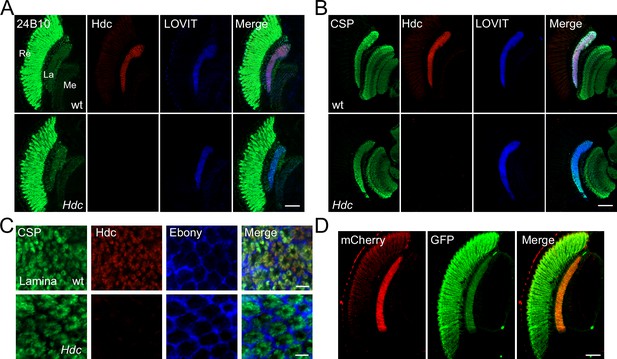
Histidine decarboxylase (Hdc) localizes to neuronal terminals.
(A–B) Cryosections of w1118 and Hdc mutant heads were labeled with antibodies against Hdc (red), 24B10 (A) (green, photoreceptor cell marker), CSP (cysteine string protein) (B) (green, localized to synaptic vesicles), and LOVIT (blue, labeling photoreceptor terminals). Scale bars, 50 μm. (C) Cross sections of the lamina layer showing overlapping patterns of Hdc (red) and CSP (green) localization, and a complementary pattern of Hdc (red) and Ebony (blue, expressed in lamina epithelial glia). Scale bars, 5 μm. (D) Cryosections of heads from trp-Hdc-mCherry/trp-GFP flies were labeled with antibodies against mCherry (red) and GFP (green). Scale bar, 50 μm. La, lamina; Me, medulla; Re, retina.
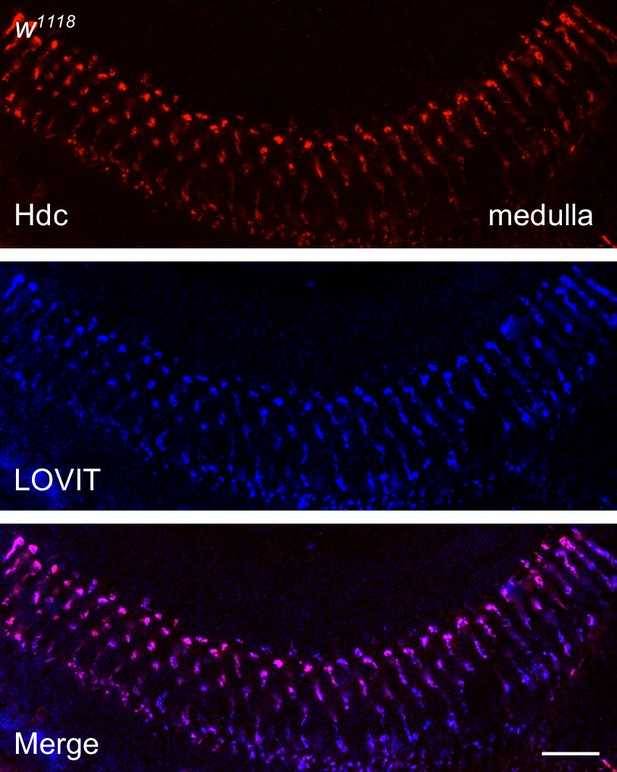
Histidine decarboxylase (Hdc) co-localizes with LOVIT in the medulla.
A longitudinal section of the distal medulla neuropil from 3-day-old w1118 fly was labeled with antibodies against Hdc (red) and LOVIT (blue, labeling photoreceptor terminals). Scale bars, 20 μm.

TADR (torn and diminished rhabdomeres) is required in photoreceptors for normal visual transmission.
(A–D) Electroretinograms (ERGs) recorded from flies expressing various UAS-tadr RNAi transgenes (tadrRNAi1 and tadrRNAi2) under the control of (A) GMR-Gal4, a driver specific for compound eyes (GMR-Gal4/ UAS-tadrRNAi1 or GMR-Gal4/+;UAS-tadrRNAi2/+) and (C) the glial-specific driver repo-Gal4 (repo-Gal4/ UAS-tadrRNAi1 or repo-Gal4/+;UAS-tadrRNAi2/+). (B) Quantitative analysis of the amplitudes of ERG OFF transients shown in A compared with control flies (GMR> GFPRNAi, GMR-Gal4/+;UAS-GFPRNAi/+) (one-way ANOVA; n = 5; ***p < 0.001). (D) Quantitative analysis of the amplitudes of ERG OFF transients shown in C compared with control flies (repo> GFPRNAi, repo-Gal4/UAS-GFPRNAi) (one-way ANOVA; n = 10; ns, not significant). Arrowheads indicate ON and OFF transients. One-day-old flies were dark adapted for 1 min and subsequently exposed to a 5 s pulse of orange light. (E–F) ERG recordings (E) and quantitative analysis of the amplitudes of ERG OFF transients (F) from wild-type (w1118), tadr2, tadr2;trp-GFP, and tadr2;trp-tadr flies. Displayed are comparisons to wild-type (w1118) flies (one-way ANOVA; n = 10; ***p < 0.001; ns, not significant). (G) Phototactic behavior of flies corresponding to those in (A) and (C) compared with control flies (GMR> GFPRNAi or repo-> GFPRNAi flies). (H) Phototactic behavior of flies corresponding to those in (E). Each group is comprised of at least twenty 3-day-old flies. Five repeats were quantified for each group (one-way ANOVA, ***p < 0.001; ns, not significant).
-
Figure 2—source data 1
Source data for quantitative of electroretinogram (ERG) transients and phototaxis behaviors.
- https://cdn.elifesciences.org/articles/75821/elife-75821-fig2-data1-v2.xlsx
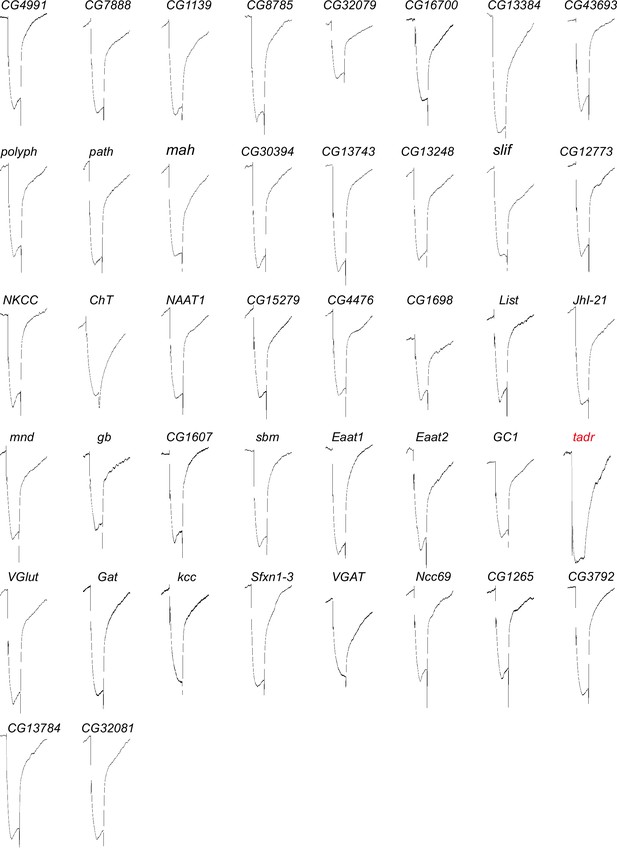
Electroretinogram (ERG) recordings for putative amino acid transporters functioned in visual transmission.
ERGs recorded from flies knocking down 42 putative amino acid transporter genes under GMR-Gal4. One-day-old flies were dark adapted for 1 min and subsequently exposed to a 5 s pulse of orange light.
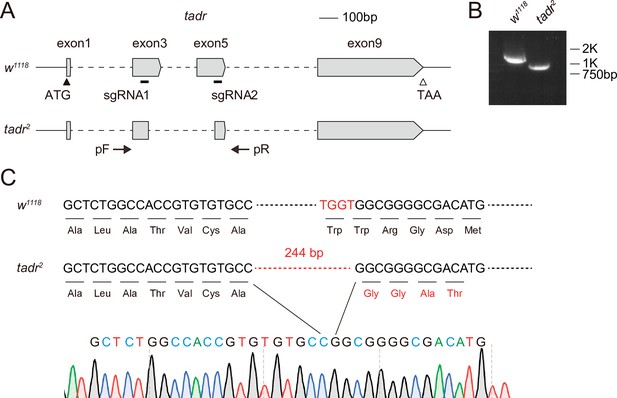
Generation of tadr2 flies.
(A) Schematic for tadr knock-out by sgRNA targeting. Organization of the tadr (torn and diminished rhabdomeres) locus and the expected structure of the tadr2 allele are shown. Boxes represent exons with the coding region between ATG and TAA. The sgRNA1 and sgRNA2 primer pairs were used to generate the tadr2 allele. Arrows indicate the primers used for genomic PCR. (B) PCR products obtained from tadr2 mutants show successful gene deletions of ~600 bp. (C) Verification of the tadr2 locus by DNA sequencing. The tadr2 mutation eliminates 665 bp within the tadr locus including all of exon 4 and parts of exons 3 and 5. This leads to a deletion for 244 nt in the spliced tadr2 mRNA, resulting in a frame shift at the truncated site, as shown.
-
Figure 2—figure supplement 2—source data 1
The full raw unedited gels for PCR products obtained from c mutants.
- https://cdn.elifesciences.org/articles/75821/elife-75821-fig2-figsupp2-data1-v2.pdf
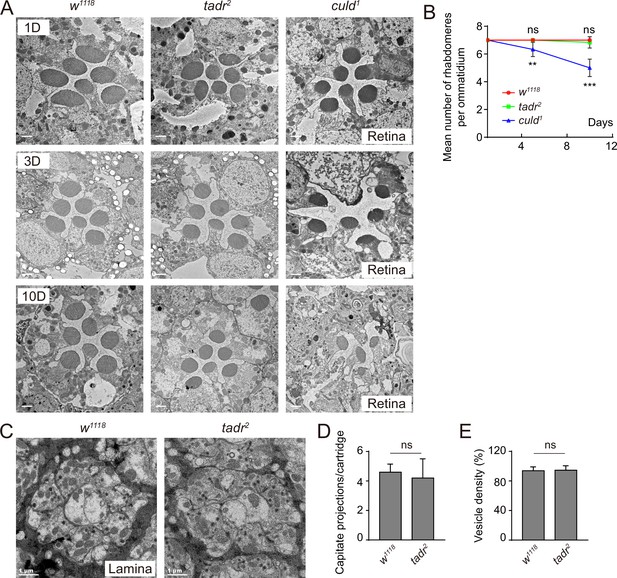
Ommatidia and cartridges are normal in tadr2 mutants.
(A) Cross-view of transmission electron microscopy (TEM) sections of the retinal layers from wild-type (w1118), tadr2, and culd1 flies at indicated days. Scale bars: 2 µm. (B) Quantification of the average number of rhabdomeres per ommatidium at indicated days for wild-type (w1118), tadr2, and culd1 flies. (C) Cross-view of TEM sections of lamina neuropil from 3-day-old w1118 and tadr2 flies. (D–E) Quantification of capitate projection (C) and synaptic vesicle density (D) from B. Sections from four different flies were quantified for each genotype.
-
Figure 2—figure supplement 3—source data 1
Source data for quantifying the average number of rhabdomeres per ommatidium at indicated days for wild-type (w1118), tadr2, and culd1 flies, as well as for quantifying capitate projection and synaptic vesicle density in tadr2 mutants.
- https://cdn.elifesciences.org/articles/75821/elife-75821-fig2-figsupp3-data1-v2.xlsx
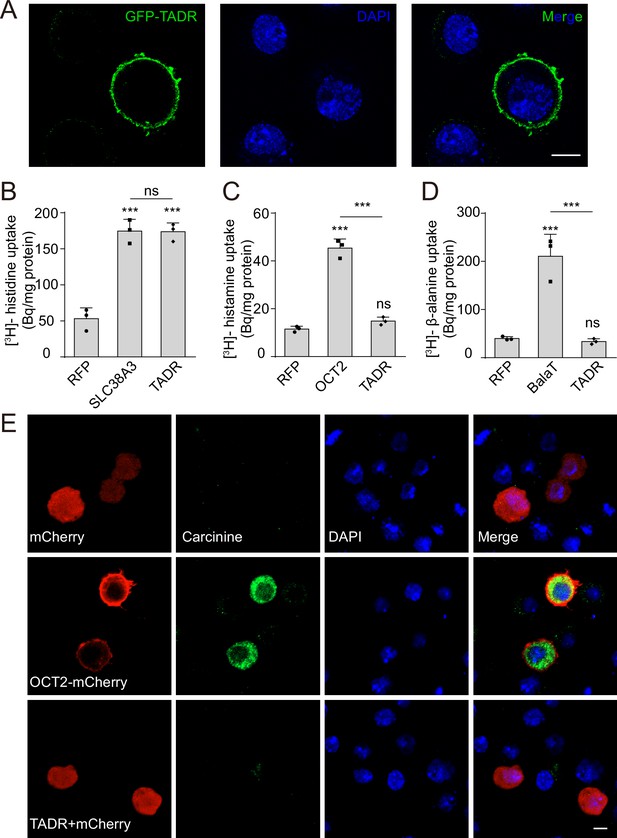
TADR (torn and diminished rhabdomeres) is a plasma membrane histidine transporter.
(A) TADR localized to the plasma membrane when transiently transfected into S2 cells. GFP-tagged TADR was labeled with GFP antibody (green) and DAPI (blue), which stained the nucleus. Scale bar, 2 μm. (B) TADR transported histidine into S2 cells. Human SLC38A3 and RFP were used as positive and negative controls, respectively. [3H]-histidine was added to the DMEM solution (final concentration 2.5 μM). (C–D) TADR did not transport histamine (C) or β-alanine (D) into S2 cells. [3H]-histamine or [3H]-β-alanine was added to the ECF buffer (final concentration 3.7 × 104 Bq), and organic cation transporter (OCT2) and BalaT served as positive transporter controls for histamine and β-alanine, respectively. Results are the mean ± SD of three experiments (one-way ANOVA, ***p < 0.001; ns, not significant). (E) TADR did not transport carcinine. S2 cells transiently expressing mCherry, OCT2-mCherry, or TADR/mCherry. Carcinine was added to the culture medium at a final concentration of 20 μM. Cells were labeled with rabbit anti-carcinine (green) antibody and DAPI (blue). The mCherry (red) signal was observed directly. Scale bar, 5 μm.
-
Figure 3—source data 1
Source data for histidine, histamine, and β-alanine uptake assay.
- https://cdn.elifesciences.org/articles/75821/elife-75821-fig3-data1-v2.xlsx
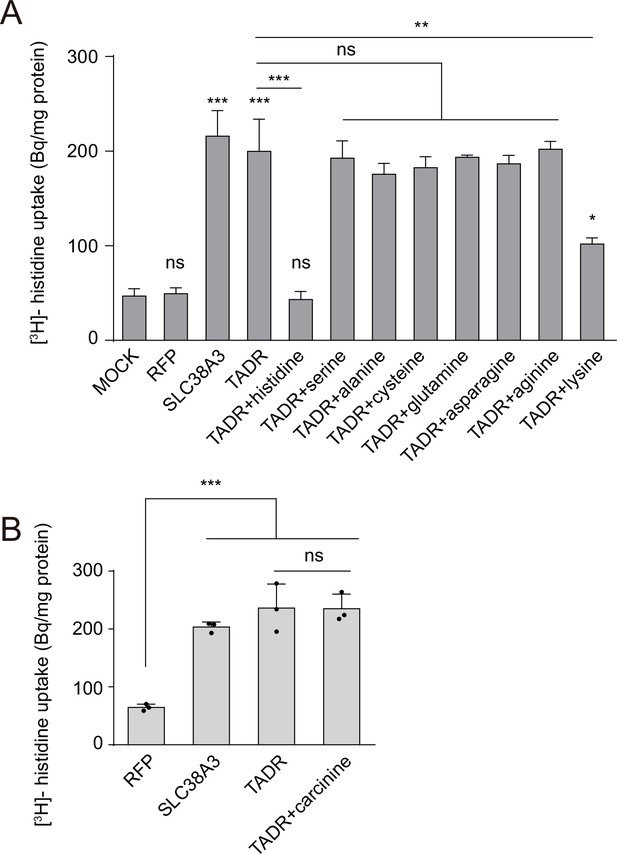
TADR (torn and diminished rhabdomeres) is a specific histidine transporter.
(A) Competition assays using [3H]-histidine in combination with high concentrations of different L-amino acids (0.5 mM for each L-amino acid vs. 2.5 µM [3H]-histidine). (B) Competition assays using [3H]-histidine in combination with a high concentration of carcinine (0.5 mM for carcinine vs. 2.5 µM [3H]-histidine). Results are the mean ± SD of three experiments (one-way ANOVA; ***p < 0.001; **p < 0.01; *p < 0.05; ns, not significant).
-
Figure 3—figure supplement 1—source data 1
Source data for competition assays using histidine in combination with different L-amino acid or carcinine.
- https://cdn.elifesciences.org/articles/75821/elife-75821-fig3-figsupp1-data1-v2.xlsx
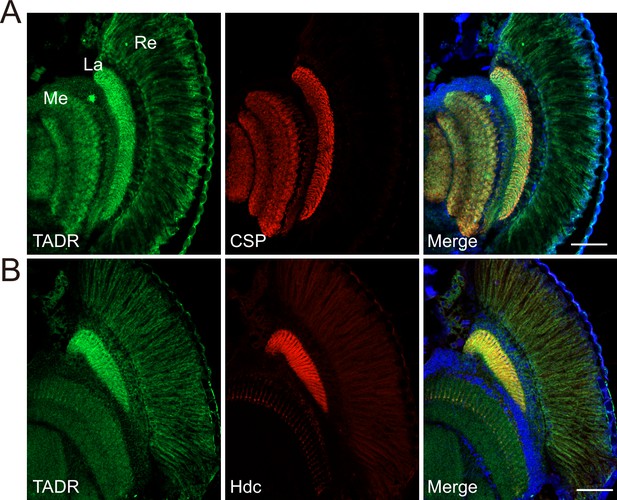
TADR (torn and diminished rhabdomeres) predominantly localizes to photoreceptor terminals.
(A–B) Cryosections from GFP-tadr knock-in flies. Expression of an N-terminal GFP-tagged version of TADR was driven by the native tadr promoter. Sections were labeled for GFP-TADR with CSP (cysteine string protein) (red) (A) or Hdc (red) (B) and DAPI (blue). Scale bars, 50 μm. La, lamina; Me, medulla; Re, retina.
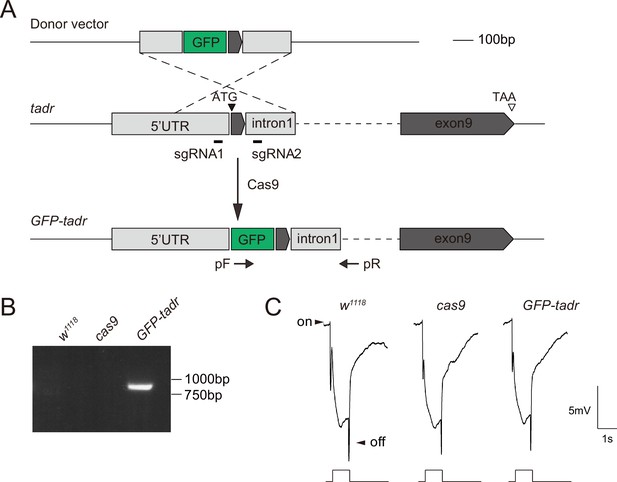
Generation of GFP-tadr (torn and diminished rhabdomeres) flies.
(A) Schematic for generating GFP-tadr flies. GFP was inserted into the tadr locus (tagging the N-terminal) using CRISPR/Cas9-mediated homologous recombination. PCR primers (arrows, pF and pR) were used to verify the GFP-tadr knock-in flies. (B) Genomic PCR products from wild-type (w1118), attP2, and GFP-tadr flies show successful gene targeting. (C) Electroretinogram(ERG) recordings from wild-type (w1118), nos-cas9, and GFP-tadr flies. Homozygous GFP-tadr flies showed intact ON and OFF transients.
-
Figure 4—figure supplement 1—source data 1
The full raw unedited gels for PCR products obtained from GFP-tadr flies.
- https://cdn.elifesciences.org/articles/75821/elife-75821-fig4-figsupp1-data1-v2.pdf
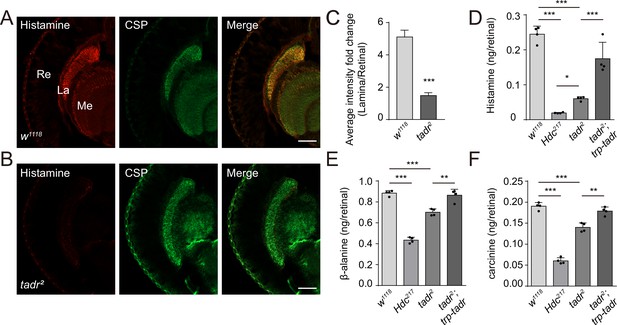
Loss of TADR (torn and diminished rhabdomeres) reduces histamine levels in vivo.
(A–C) Histamine signaling in photoreceptor terminals was disrupted. Head cryosections were stained for histamine together with CSP (cysteine string protein) (synaptic vesicle marker) in control (w1118) (A) and tadr2 (B) flies. Sections are parallel to photoreceptor axons. Scale bars, 50 μm. (C) Average red fluorescence intensity ratio between the entire lamina and the retina immunolabeled layers. (D–F) Histamine (D), β-alanine (E), and carcinine (F) levels in compound eyes of 3-day-old control (w1118), HdcP217, tadr2, and tadr2;trp-tadr flies. Each sample included dissected compound eyes from 40 flies (one-way ANOVA; n = 4; ***p < 0.001; **p < 0.01; *p < 0.05). La, lamina; Me, medulla; Re, retina.
-
Figure 5—source data 1
Source data for the levels of histamine, β-alanine, and carcinine in w1118, HdcP217, tadr2, and tadr2;trp-tadr mutant fly compound eyes.
- https://cdn.elifesciences.org/articles/75821/elife-75821-fig5-data1-v2.xlsx
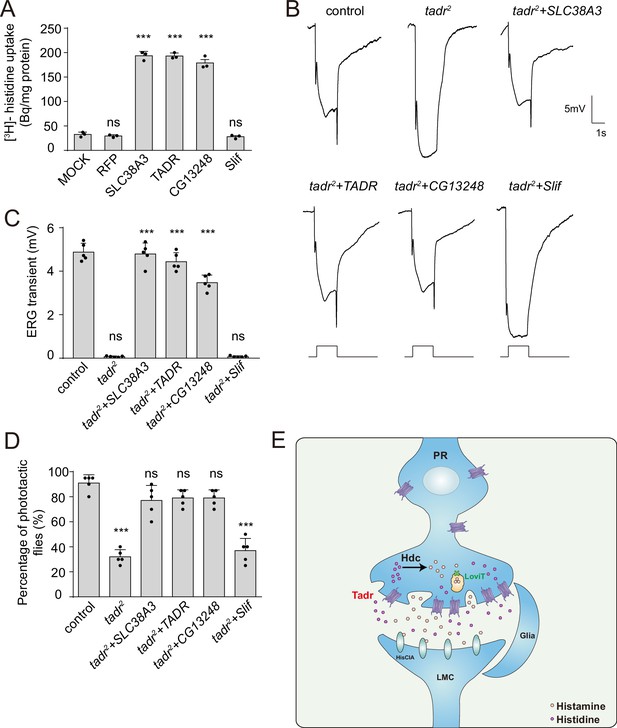
Rescue of defective visual transmission in tadr2 mutants by expressing other histidine transporters.
(A) SLC38A3 and CG13248 transported histidine into S2 cells, whereas the previously identified amino acid transporter, Slif, did not transport histidine. [3H]-histidine was added to the DMEM solution to a final concentration 2.5 μM. (B) Electroretinogram (ERG) recordings from control (w1118), tadr2, tadr2+ tadr (tadr2;longGMR-Gal4/UAS-tadr), tadr2+ CG13248 (tadr2;longGMR-Gal4/UAS-CG13248), tadr2+ Slif (tadr2;longGMR-Gal4/UAS-Slif), and tadr2+ SLC38 A3 (tadr2;longGMR-Gal4/UAS-SLC38A3) are shown. Young flies (<3 days after eclosion) were dark adapted for 1 min and subsequently exposed to a 5 s pulse of orange light. (C) Quantitative analysis of the amplitude of ERG OFF transients shown in B. Displayed are comparisons to control (w1118) flies (one-way ANOVA; n = 10; ***p < 0.001; ns, not significant). (D) Phototactic behaviors of 3-day-old control, tadr2, tadr2+ tadr, tadr2+ CG13248, tadr2+ Slif, and tadr2+ SLC38 A3 flies. Five repeats were made for each group, and each group had at least 20 flies (one-way ANOVA, ***p < 0.001; ns, not significant). (E) Model of the pathway for local histamine biosynthesis. Histidine is directly transported into photoreceptor cells at neuronal terminals by TADR, where it is used as a substrate to synthesize histamine by the decarboxylase, Hdc. Newly generated histamine is then loaded into synaptic vesicles by a LOVIT-dependent mechanism. Histamine, serving as a neurotransmitter, is released by photoreceptor cells (PR) to activate histamine-gated chloride channels (HisClA) on postsynaptic neurons (large monopolar cell [LMC]) to start visual transmission.
-
Figure 6—source data 1
Source data for histidine uptake assay and source data for quantitative of electroretinogram (ERG) transients and phototaxis behaviors.
- https://cdn.elifesciences.org/articles/75821/elife-75821-fig6-data1-v2.xlsx
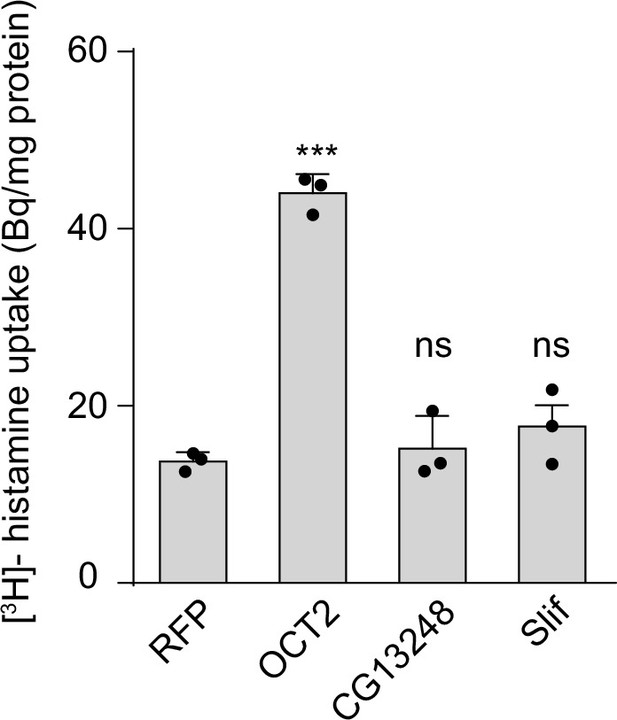
Both CG13248 and Slif do not transport histamine.
CG13248 and Slif did not transport histamine into S2 cells. [3H]-histamine was added to the ECF buffer (final concentration 3.7 × 104 Bq). Organic cation transporter (OCT2) served as a positive transporter control for histamine. Results are the mean ± SD of three experiments (one-way ANOVA, ***p < 0.001; ns, not significant).
-
Figure 6—figure supplement 1—source data 1
Source data for the histamine uptake assay.
- https://cdn.elifesciences.org/articles/75821/elife-75821-fig6-figsupp1-data1-v2.xlsx
Tables
Description of 42 putative amino acid transporters.
| CG number | On/OFF | Description |
|---|---|---|
| CG4991 | Yes | Amino acid transmembrane transporter activity/SLC36A1 or A2 |
| CG7888 | Yes | Amino acid transmembrane transporter activity/SLC36A1 or A2 |
| CG1139 | Yes | Amino acid transmembrane transporter activity/SLC36A1 or A2 |
| CG8785 | Yes | Amino acid transmembrane transporter activity/SLC36A1 or A2 |
| CG32079 | Yes | Amino acid transmembrane transporter activity/SLC36A1 or A2 |
| CG32081 | Yes | Amino acid transmembrane transporter activity/SLC36A1 or A2 |
| CG16700 | Yes | Amino acid transmembrane transporter activity/SLC36A1 or A4 |
| CG13384 | Yes | Amino acid transmembrane transporter activity/SLC36A1 or A4 |
| CG43693 | Yes | Amino acid transmembrane transporter activity/SLC36A1 or A4 |
| polyph | Yes | Amino acid transmembrane transporter activity/SLC36A1 or A2 |
| path | Yes | Amino acid transmembrane transporter activity/SLC36A1 or A2 |
| mah | Yes | Amino acid transmembrane transporter activity/SLC36A1 or A2 |
| CG30394 | Yes | Amino acid transmembrane transporter activity/SLC38A10 |
| CG13743 | Yes | Amino acid transmembrane transporter activity/SLC38A11 |
| CG13248 | Yes | Amino acid transmembrane transporter activity/SLC7A4 |
| slif | Yes | Amino acid transmembrane transporter activity/SLC7A1 or A2 |
| CG12773 | Yes | Amino acid transmembrane transporter activity/SLC12A8 |
| NKCC | Yes | Amino acid transmembrane transporter activity/SLC12A3 |
| ChT | Yes | Amino acid transmembrane transporter activity/SLC5A7 |
| NAAT1 | Yes | Amino acid transmembrane transporter activity/SLC6A7 or A9 |
| CG15279 | Yes | L-amino acid transmembrane transporter activity/SLC6A7 or A9 |
| CG4476 | Yes | L-amino acid transmembrane transporter activity/SLC6A7 or A9 |
| CG1698 | Yes | L-amino acid transmembrane transporter activity/SLC6A7 or A9 |
| List | Yes | L-amino acid transmembrane transporter activity/SLC6A7 |
| JhI-21 | Yes | L-amino acid transmembrane transporter activity/SLC6A5 |
| mnd | Yes | L-amino acid transmembrane transporter activity/SLC7A6 or A7 |
| gb | Yes | L-amino acid transmembrane transporter activity/SLC7A6 or A7 |
| CG1607 | Yes | L-amino acid transmembrane transporter activity/SLC7A8 |
| sbm | Yes | L-amino acid transmembrane transporter activity/SLC7A9 |
| Eaat1 | Yes | L-amino acid transmembrane transporter activity/SLC7A3 |
| Eaat2 | Yes | L-amino acid transmembrane transporter activity/SLC7A2 |
| GC1 | Yes | L-glutamate transmembrane transport/SLC25A18 |
| tadr | No | Cationic amino acid transporter/SLC7A4 or SLC7A1 |
| VGlut | Yes | Vesicular glutamate transporter/SLC17A7 |
| Gat | Yes | GABA transporter activity/SLC6A1 |
| kcc | Yes | Potassium:chloride symporter activity/SLC12A4 |
| Sfxn1-3 | Yes | Serine transmembrane transporter activity/SFXN1 |
| VGAT | Yes | Vesicular GABA transporter activity/SLC32A1 |
| Ncc69 | Yes | Sodium:potassium:chloride symporter activity/SLC12A1 or A2 |
| CG1265 | Yes | Lysosomal amino acid transporter/SLC66A3 |
| CG3792 | Yes | Lysosomal amino acid transporter/SLC66 |
| CG13784 | Yes | Lysosomal amino acid transporter/SLC66A2 |
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (Drosophila melanogaste) | tadr (cDNA) | Drosophila Genomics Resource Center | FLYB:FBcl0168145 | FlyBase symbol: LD25644 |
| Gene (Drosophila melanogaste) | slif (cDNA) | Drosophila Genomics Resource Center | FLYB:FBcl0167622 | FlyBase symbol: LD37241 |
| Gene (Drosophila melanogaste) | CG13248 (cDNA) | Drosophila Genomics Resource Center | FLYB:FBcl0718746 | FlyBase symbol: FI04531 |
| Gene (Homo sapiens) | SLC38A3 | NCBI database | Gene ID: 10,991 | Synthesized from GENEWIZ, China. |
| Genetic reagent (Drosophila melanogaster) | tadrRNAi1 | Vienna Drosophila Resource Center | BDSC:v330472RRID:FlyBase_FBst0492187 | P{VSH330472}attP40 |
| Cell line (Drosophila melanogaste) | S2 | This paper | FLYB:FBtc0000181; RRID:CVCL_Z992 | Cell line maintained in N. Perrimon lab; FlyBase symbol: S2-DRSC. |
| Antibody | Anti-24B10(Mouse monoclonal) | DHSB | RRID:AB_528161 | IF(1:100) |
| Antibody | Anti-CSP(Mouse monoclonal) | DHSB | RRID:AB_528183 | IF(1:100) |
| Antibody | Anti-RFP(Ratmonoclonal) | Chromotek | RRID:AB_2336064 | IF(1:200) |
| Antibody | Anti-GFP(Rabbit polyclonal) | Invitrogen | RRID:AB_221569 | IF(1:200) |
| Antibody | Anti-LOVIT(Rat polyclonal) | Xu and Wang, 2019 | IF(1:100) | |
| Antibody | Anti-Hdc(Rabbit polyclonal) | This paper | IF(1:50) | |
| Antibody | Anti-Ebony(Rabbit polyclonal) | University of Wisconsin | IF(1:200) | |
| Antibody | Anti-Histamine(Rabbit polyclonal) | ImmunoStar | RRID:AB_572245 | IF(1:100) |
| Recombinant DNA reagent | pTrp-GFP | This paper | Used for generation of transgenic flies (maintained in T. Wang lab) | |
| Recombinant DNA reagent | pTrp-GFP-tadr | This paper | Used for generation of transgenic flies (maintained in T. Wang lab) | |
| Recombinant DNA reagent | pTrp-tadr-GFP | This paper | Used for generation of transgenic flies (maintained in T. Wang lab) | |
| Recombinant DNA reagent | pTrp-tadr | This paper | Used for generation of transgenic flies (maintained in T. Wang lab) | |
| Sequence-based reagent | sgRNA1 (for tadr2 mutant) | This paper | sgRNAs | GTGCCTGCGCTGCCCTGGCG |
| Sequence-based reagent | sgRNA2 (for tadr2 mutant) | This paper | sgRNAs | TTTTAAGCGCCGTCGGCTGG |
| Sequence-based reagent | forward primer (for tadr2 mutant) | This paper | PCR primers | CAATGGCAGGTGGGAGTTAGG |
| Sequence-based reagent | reverse primer (for tadr2 mutant) | This paper | PCR primers | TTAGAGTCGCCGTGAATCGTC |
| Sequence-based reagent | sgRNA (for GFP-tadr knock-in) | This paper | sgRNAs | ACAACAACGACAATGTCGAG |
| Peptide, recombinant protein | Hdc peptide (D. melanogaste) | This paper | Synthesized by ChinaPeptides (Soochow, China) | CDFKEYRQRGKEMVDY |
| Chemical compound, drug | [3H]-Histidine | American radiolabeled chemicals | ART 0234 | 30–60 Ci/mM |
| Chemical compound, drug | [3H]-Histamine | American radiolabeled chemicals | ART 1432 | 10–40 Ci/mM |
| Chemical compound, drug | [3H]-β-alanine | American radiolabeled chemicals | ART 0205 | 30–60 Ci/mM |
| Software, algorithm | GraphPad Prism software | GraphPad Prism (https://graphpad.com) | RRID:SCR_015807 | Version 7.0.0 |
| Software, algorithm | ImageJ software | ImageJ (http://imagej.nih.gov/ij/) | RRID:SCR_003070 |





