Large domains of heterochromatin direct the formation of short mitotic chromosome loops
Figures
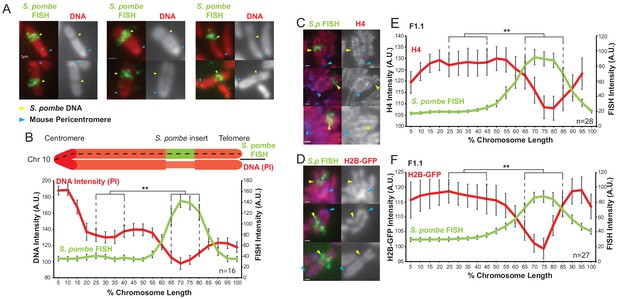
S. pombe DNA inserted into a mouse chromosome adopts a distinct structure on mitotic chromosomes.
(A) Metaphase spreads of mouse F1.1 chromosomes showing the distinct structure of the S. pombe DNA insert. Propidium iodide stained DNA (PI, red), S. pombe DNA FISH using probes from total S. pombe DNA (green – yellow arrows), centromeres (regions of brighter DNA staining - blue arrows). Scale bars: 1 μm. (B) Schematic representation and average chromosome profile of the F1.1 insert-bearing chromosome across several images (n = 16, Figure 1—source data 1). Signal intensities of PI DNA stain (red) and FISH signal (green) were measured along the length of the chromosomes and binned according to their position, from the centromere (0–5%) to the telomere (95–100%). Error bars represent ± standard error from the mean (SEM). Average DNA stain intensity was compared between the regions of 25–40% (endogenous mouse DNA) and 65–80% (S. pombe DNA corresponding to the highest FISH signal) by the KS test (**p<0.001). (C and D) Metaphase spreads of F1.1 cells either stained by immunofluorescence for histone H4 (C) or expressing tagged histone H2B-GFP (D) (red), with S. pombe DNA FISH (green) and DAPI-stained DNA (blue). S. pombe DNA (yellow arrows) and centromere (blue arrows) locations are indicated. Scale bars: 1 μm. (E and F) Average signal intensity profile of the F1.1 insert-bearing chromosome showing FISH and either anti-H4 (E, Figure 1—source data 2) or H2B-GFP (F, Figure 1—source data 3) across several images (n = 28, 27). Error bars represent ± SEM. Average histone levels were compared between the regions of endogenous mouse DNA and S. pombe DNA highlighted by FISH by the KS test (**p<0.001).
-
Figure 1—source data 1
FISH and PI intensity measurements for Figure 1B.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig1-data1-v3.xlsx
-
Figure 1—source data 2
FISH and anti-H4 intensity measurements for Figure 1E.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig1-data2-v3.xlsx
-
Figure 1—source data 3
FISH and H2B-GFP intensity measurements for Figure 1F.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig1-data3-v3.xlsx
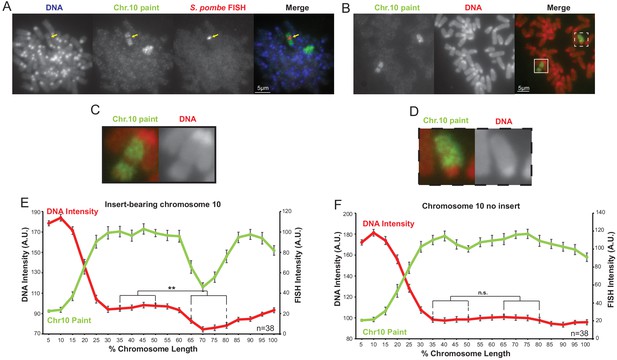
The distinct chromatin structure of the S. pombe DNA insert on chromosome 10 is not an artefact of the FISH procedure.
(A) Two-colour FISH of F1.1 metaphase spreads for S. pombe DNA (red, middle-right, yellow arrows), and mouse chromosome 10 (green, middle-left) with DAPI-stained DNA (blue, far left). (B) Metaphase spread of F1.1 probed with a mouse chromosome 10 paint only (green) with PI-stained DNA (red). The insert-bearing chromosome 10 is recognisable by a distinct region not highlighted by FISH, corresponding to the S. pombe DNA. Scale bars: 5 μm. (C,D) Enlarged images of the regions highlighted in B showing an S. pombe DNA insert-bearing (C) and non-insert bearing (D) copy of chromosome 10. (E,F) Average chromosome profile of the insert-bearing (E, Figure 1—figure supplement 1—source data 1) and homologous non-insert-bearing (F, Figure 1—figure supplement 1—source data 2) copies of chromosome 10 in F1.1 metaphase spreads across several images (n = 38). Error bars represent ± SEM. Average DNA stain intensity was compared between the regions of DNA at 35–50% and 65–80% (where the S. pombe DNA was detected on the insert-bearing chromosome) by the KS test (**p<0.001, n.s. = not significant).
-
Figure 1—figure supplement 1—source data 1
FISH and PI intensity measurements for Figure 1—figure supplement 1E.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig1-figsupp1-data1-v3.xlsx
-
Figure 1—figure supplement 1—source data 2
FISH and PI intensity measurements for Figure 1—figure supplement 1F.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig1-figsupp1-data2-v3.xlsx
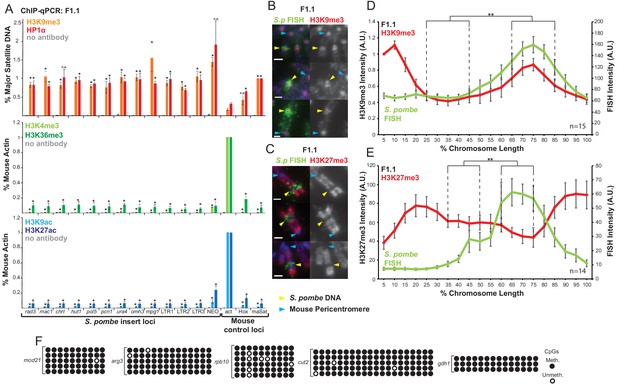
S. pombe DNA in chromosome 10 of F1.1 is coated in H3K9me3-heterochromatin.
(A) ChIP-qPCR of F1.1 interphase cells for the repressive marks H3K9me3 and HP1α (top), activating histone methylation marks H3K4me3 and H3K36me3 (middle) and activating acetylation marks H3K9ac and H3K27ac (bottom) at 13 loci within the S. pombe insert and three mouse control loci. act - highly transcribed control gene Actb; Hox – Hoxc8 region of facultative heterochromatin; maSat - constitutively heterochromatic centromeric major satellite region. Data in Figure 2—source data 1. Error bars represent ± SEM of three independent repeats. Enrichments were normalised to positive control levels and compared to act by the t-test (*p<0.05, n.s. = not significant). (B,C) Immunofluorescence for H3K9me3 (B) or H3K27me3 (C) (red) on F1.1 metaphase spreads showing high H3K9me3 and low H3K27me3 over the S. pombe DNA insert as visualised by FISH (green), with DAPI-stained DNA (blue). S. pombe DNA (yellow arrows) and centromere (blue arrows) locations are indicated. Scale bars: 1 μm. (D,E) Average FISH and H3K9me3 (D Figure 2—source data 2) or H3K27me3 (E Figure 2—source data 3) signal intensity profiles of the insert-bearing chromosome of F1.1 across several images (n = 15, 14). Error bars represent ± SEM. H3K9me3 intensity levels were normalised to the 0–5% region of the chromosome, corresponding to the acrocentric mouse centromere. Average immunofluorescence intensity was compared between the regions of endogenous mouse DNA and S. pombe DNA highlighted by FISH by the KS test (**p<0.001). (F) CpG methylation levels at five S. pombe loci within the F1.1 insert as determined by bisulfite sequencing. Loci were sequenced in 3 to 6 replicates, with each replicate shown. Circles represent methylated (black) and unmethylated (white) CpGs, respectively.
-
Figure 2—source data 1
ChIP results for H3K9me3, HP1α, H3K4me3, H3K36me3, H3K9ac and H3K27ac on F1.1 cells fore Figure 2A.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig2-data1-v3.xlsx
-
Figure 2—source data 2
FISH and anti-H3K9me3 intensity measurements for Figure 2D.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig2-data2-v3.xlsx
-
Figure 2—source data 3
FISH and anti-H3K9me3 intensity measurements for Figure 2E.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig2-data3-v3.xlsx
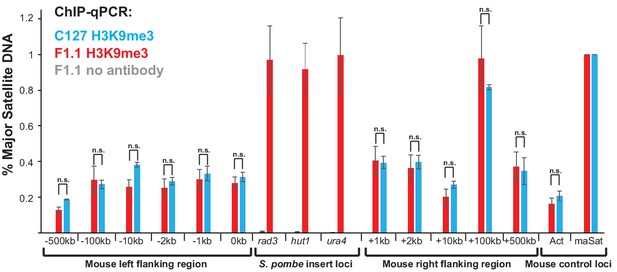
Heterochromatin is confined to the region of foreign S. pombe DNA in F1.1 cells.
ChIP-qPCR for H3K9me3 in F1.1 (red) and C127 (blue) interphase cells at three S. pombe loci within the insert (for F1.1 only), two mouse control loci and eleven mouse loci that flank the S. pombe DNA insertion site at the indicated distance to left or right. act - highly transcribed negative control Actb gene; maSat - centromeric heterochromatin satellite positive control. Enrichments are normalised to maSat positive control levels. Data in Figure 2—figure supplement 1—source data 1. Error bars represent ± SEM of three independent repeats. Enrichment levels between F1.1 and C127 cells were compared by a t-test (*p<0.05, n.s. = not significant).
-
Figure 2—figure supplement 1—source data 1
ChIP results for H3K9me3 on F1.1 and C127 cells for Figure 2—figure supplement 1.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig2-figsupp1-data1-v3.xlsx
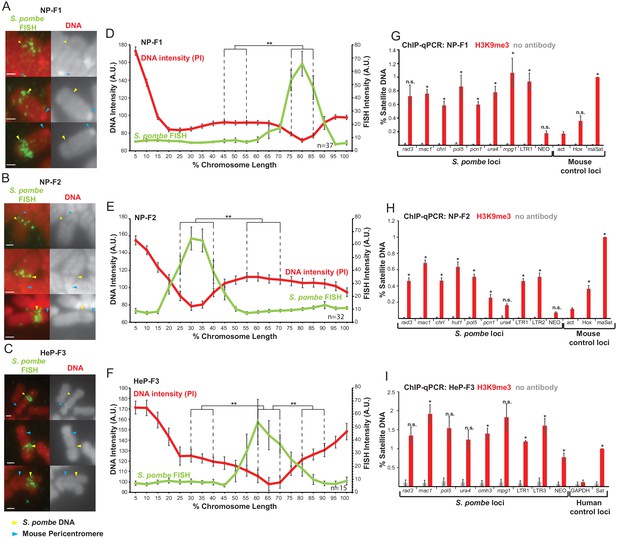
S. pombe DNA assembles heterochromatin and adopts a distinct mitotic chromatin structure when inserted into mammalian chromosomes.
(A–C) Metaphase spreads of NP-F1, NP-F2 and HeP-F3 cells showing the distinct structure over the inserted S. pombe DNA. PI-stained DNA (red), S. pombe DNA FISH (green – yellow arrows). Blue arrows indicate centromeres. Scale bar: 1 μm. (D–F) Average FISH and PI signal intensity profiles of the insert-bearing chromosomes in each cell line across several images (n = 37, 32 and 15, Figure 3—source datas 1–3). Error bars represent ± standard error from the mean (SEM). Average DNA stain intensity was compared between the regions of endogenous mammalian DNA and S. pombe DNA highlighted by FISH by the KS test (**p<0.001). (G–I) ChIP-qPCR for H3K9me3 in NP-F1 (G, Figure 3—source data 4), NP-F2 (H, Figure 3—source data 5) and HeP-F3 (I, Figure 3—source data 6) in interphase. qPCR was performed with primers used for analysis of F1.1 (see Figure 2) that were present in the retained S. pombe DNA. Mouse control regions were: negative control Actb gene – act; facultative heterochromatin region – Hox; centromeric heterochromatin major satellite positive control – maSat. Human control regions were: the highly transcribed negative control gene – GAPDH; human alpha satellite DNA positive control – Sat. Error bars represent ± SEM of three independent repeats. Enrichments were normalised to positive control levels and compared to the negative control by the t-test (*p<0.05, n.s. = not significant).
-
Figure 3—source data 1
FISH and PI intensity measurements for Figure 3D.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig3-data1-v3.xlsx
-
Figure 3—source data 2
FISH and PI intensity measurements for Figure 3E.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig3-data2-v3.xlsx
-
Figure 3—source data 3
FISH and PI intensity measurements for Figure 3F.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig3-data3-v3.xlsx
-
Figure 3—source data 4
ChIP results for H3K9me3 on NP-F1 cells for Figure 3G.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig3-data4-v3.xlsx
-
Figure 3—source data 5
ChIP results for H3K9me3 on NP-F2 cells for Figure 3H.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig3-data5-v3.xlsx
-
Figure 3—source data 6
ChIP results for H3K9me3 on HeP3 cells for Figure 3I.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig3-data6-v3.xlsx
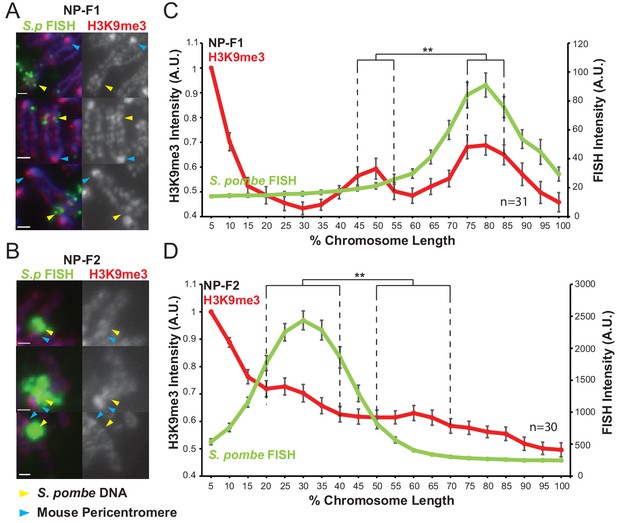
Heterochromatin assembled on S.pombe DNA inserted into NIH3T3 chromosomes is visible by microscopy.
(A,B) Immunofluorescence for H3K9me3 (red) on metaphase spreads of NP-F1 (A) and NP-F2 (B) with DAPI-stained DNA (blue) and S. pombe DNA FISH (green – yellow arrows). Blue arrows indicate centromeres. (C,D) Average FISH and H3K9me3 signal intensity profile of the insert-bearing chromosomes of NP-F1 (C, Figure 3—figure supplement 1—source data 1) and NP-F2 (D, Figure 3—figure supplement 1—source data 2) across several images (n = 31, 30). Error bars represent ± SEM. H3K9me3 intensity levels were normalised to the 0–5% region of the chromosome, corresponding to the acrocentric mouse centromere. Average H3K9me3 intensity was compared between the regions of endogenous mouse DNA and S. pombe DNA highlighted by FISH by the KS test (**p<0.001).
-
Figure 3—figure supplement 1—source data 1
FISH and anti-H3K9me3 intensity measurements for Figure 3—figure supplement 1C.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig3-figsupp1-data1-v3.xlsx
-
Figure 3—figure supplement 1—source data 2
FISH and anti-H3K9me3 intensity measurements for Figure 3—figure supplement 1D.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig3-figsupp1-data2-v3.xlsx
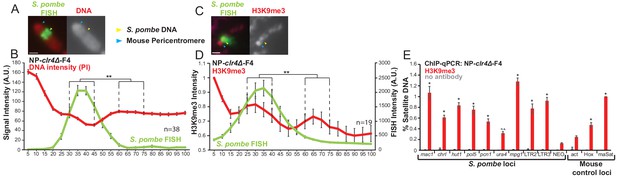
S. pombe DNA from cells lacking H3K9 methylation forms heterochromatin and adopts a distinct structure when incorporated into mammalian chromosomes.
(A) Metaphase spread of NP-clr4Δ-F4 showing the distinct structure over the S. pombe DNA insert, with PI-stained DNA (red) and S. pombe DNA FISH (green – yellow arrows). Blue arrows indicate the centromere. Scale bar: 1 μm. (B) Average FISH and DNA stain (PI) signal intensity profile of the insert-bearing chromosomes of NP-clr4Δ-F4 (n = 38, Figure 4—source data 1). Error bars represent ± SEM. Average DNA stain intensity was compared between a region of endogenous mouse DNA and S. pombe DNA highlighted by FISH by the KS test (**p<0.001). (C) Immunofluorescence for H3K9me3 (red) on a NP-clr4Δ-F4 metaphase spread with DAPI-stained DNA (blue) and S. pombe DNA FISH (green - yellow arrow). Blue arrows indicate the centromere. Scale bars: 1 μm. (D) Average FISH and H3K9me3 signal intensity profile along the NP-clr4Δ-F4 insert-bearing chromosome from several images (n = 19, Figure 4—source data 2). Error bars represent ± SEM. H3K9me3 intensity levels were normalised to the 0–5% region of the chromosome, corresponding to the acrocentric mouse centromere. Average H3K9me3 intensity was compared between the regions of endogenous mouse DNA and S. pombe DNA highlighted by FISH by the KS test (*p<0.01). (E) ChIP-qPCR on NP-clr4Δ-F4 interphase cells for H3K9me3 at several loci within the S. pombe insert and three mouse control loci. Negative control gene Actb - act, facultative heterochromatin - Hox, centromeric heterochromatn major satellite positive control – maSat. Data in Figure 4—source data 3. Error bars represent ± SEM of three independent repeats. Enrichments were normalised to maSat levels and compared to act by the t-test (*p<0.05, n.s. = not significant).
-
Figure 4—source data 1
FISH and PI intensity measurements for Figure 4B.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig4-data1-v3.xlsx
-
Figure 4—source data 2
FISH and anti-H3K9me3 intensity measurements for Figure 4D.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig4-data2-v3.xlsx
-
Figure 4—source data 3
ChIP results for H3K9me3 on NP-clr4Δ-F4 cells for Figure 4E.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig4-data3-v3.xlsx
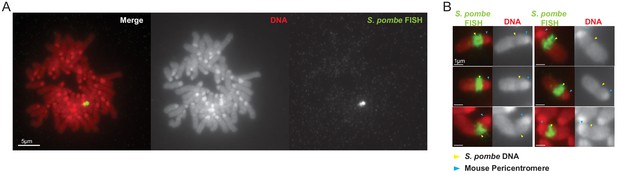
Distinct structure formed by DNA from S. pombe clr4Δ cells when inserted into a mouse NIH3T3 chromosome.
(A and B) FISH for S. pombe DNA on a NP-clr4Δ-F4 metaphase spread showing the single large insertion of S. pombe DNA (A) and close up images showing the distinct structure adopted over the inserted S. pombe DNA (B). PI-stained DNA (red), S. pombe DNA FISH (green - yellow arrows). Blue arrows in B indicate bright PI- stained centromeres. Scale bars: 5 μm (A) and 1 μm (B).
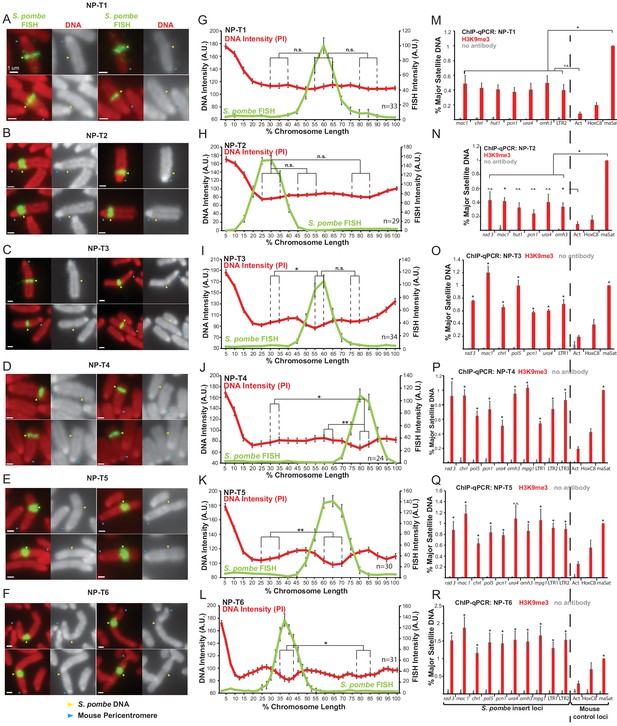
Distinct metaphase structure correlates with H3K9me3 levels over regions of S. pombe DNA inserted in mammalian chromosomes.
(A–F) Metaphase spreads of S. pombe DNA insert-bearing chromosomes from S. pombe DNA transfected cell lines NP-T1, T2, T3, T4, T5 and T6. PI-stained DNA (red), S. pombe DNA FISH (green - yellow arrows), centromeres (blue arrows). Scale bars: 1 μm. (G–L) Average FISH and DNA stain (PI) signal intensity profiles along S. pombe DNA insert-bearing chromosomes of NP-T1 to NP-T6 (n = 33, 29, 34, 24, 30, 31, Figure 5—source datas 1–6). Error bars represent ± SEM. Average DNA stain intensity was compared between regions of endogenous mouse DNA and S. pombe DNA highlighted by FISH by the KS test (*p<0.01, **p<0.001, n.s. = not significant). (M–R) ChIP-qPCR on NP-T1 to NP-T6 interphase cells for H3K9me3 at several loci within the S. pombe insert and three mouse control loci. Negative control gene Actb - act, facultative heterochromatin - Hox, centromeric heterochromatin major satellite positive control – maSat. Data in Figure 5—source datas 7–12. Error bars represent ± SEM of three independent repeats. Enrichments were normalised to maSat levels and compared to Act (and maSat for NP-T1 and NP-T2) by the t-test (*p<0.05, n.s. = not significant).
-
Figure 5—source data 1
FISH and PI intensity measurements for Figure 5G.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-data1-v3.xlsx
-
Figure 5—source data 2
FISH and PI intensity measurements for Figure 5H.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-data2-v3.xlsx
-
Figure 5—source data 3
FISH and PI intensity measurements for Figure 5I.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-data3-v3.xlsx
-
Figure 5—source data 4
FISH and PI intensity measurements for Figure 5J.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-data4-v3.xlsx
-
Figure 5—source data 5
FISH and PI intensity measurements for Figure 5K.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-data5-v3.xlsx
-
Figure 5—source data 6
FISH and PI intensity measurements for Figure 5L.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-data6-v3.xlsx
-
Figure 5—source data 7
ChIP results for H3K9me3 on NP-T1 cells for Figure 5M.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-data7-v3.xlsx
-
Figure 5—source data 8
ChIP results for H3K9me3 on NP-T2 cells for Figure 5N.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-data8-v3.xlsx
-
Figure 5—source data 9
ChIP results for H3K9me3 on NP-T3 cells for Figure 5O.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-data9-v3.xlsx
-
Figure 5—source data 10
ChIP results for H3K9me3 on NP-T4 cells for Figure 5P.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-data10-v3.xlsx
-
Figure 5—source data 11
ChIP results for H3K9me3 on NP-T5 cells for Figure 5Q.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-data11-v3.xlsx
-
Figure 5—source data 12
ChIP results for H3K9me3 on NP-T6 cells for Figure 5R.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-data12-v3.xlsx
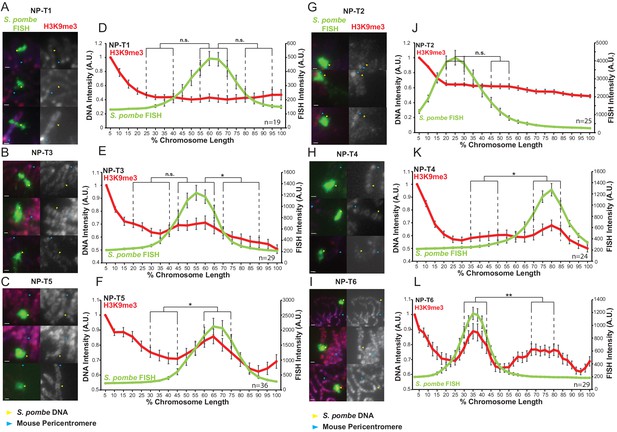
Variable levels of H3K9me3 accumulate on S. pombe DNA transgenomes in mammalian chromosomes.
(A–C, G–I) Immunofluorescence for H3K9me3 (red) on NP-T1-to-T6 metaphase spreads, S. pombe DNA FISH (green - yellow arrows), DAPI-stained DNA (blue), centromeres (blue arrows). Scale bars: 1 μm. (D–F, J–L) Average FISH and H3K9me3 signal intensity profiles of the insert-bearing chromosomes of NP-T1-to-T6 across several images (n = 19, 29, 36, 25, 24, 29, Figure 5—figure supplement 1—source datas 1–6). Error bars represent ± SEM. H3K9me3 intensity levels were normalised to the 0–5% region of the chromosome, corresponding to the acrocentric mouse centromere. Average H3K9me3 intensity was compared between the regions of endogenous mouse DNA and S. pombe DNA highlighted by FISH by the KS test (*p<0.01, **<0.001, n.s. = not significant).
-
Figure 5—figure supplement 1—source data 1
FISH and anti-H3K9me3 intensity measurements for Figure 5—figure supplement 1D.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-figsupp1-data1-v3.xlsx
-
Figure 5—figure supplement 1—source data 2
FISH and anti-H3K9me3 intensity measurements for Figure 5—figure supplement 1E.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-figsupp1-data2-v3.xlsx
-
Figure 5—figure supplement 1—source data 3
FISH and anti-H3K9me3 intensity measurements for Figure 5—figure supplement 1F.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-figsupp1-data3-v3.xlsx
-
Figure 5—figure supplement 1—source data 4
FISH and anti-H3K9me3 intensity measurements for Figure 5—figure supplement 1J.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-figsupp1-data4-v3.xlsx
-
Figure 5—figure supplement 1—source data 5
FISH and anti-H3K9me3 intensity measurements for Figure 5—figure supplement 1K.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-figsupp1-data5-v3.xlsx
-
Figure 5—figure supplement 1—source data 6
FISH and anti-H3K9me3 intensity measurements for Figure 5—figure supplement 1L.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig5-figsupp1-data6-v3.xlsx
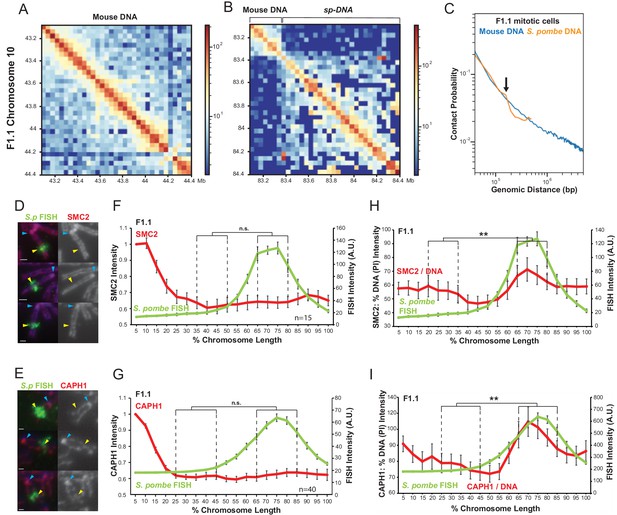
Chromatin assembled on S. pombe DNA in F1.1 cells is organised into smaller loops than flanking mouse chromatin in mitosis.
(A,B) Hi-C contact heatmaps for the longest contig containing S. pombe DNA and flanking mouse DNA from the F1.1 de novo genome assembly (B) and a contiguous region of mouse chromosome 10 of the same size (A). (C) Chromatin interactions as a function of genomic distance in F1.1 mitotic cells. Contact probability plotted for mouse sequences and S. pombe sequences mapped to the F1.1 de novo assembly. Arrow indicates drop in S. pombe interactions. (D,E) Immunofluorescence for SMC2 (D) or CAPH1 (E) on F1.1 metaphase spreads (red); S. pombe DNA FISH (green - yellow arrows), DAPI-stained DNA (blue), centromeres (blue arrows). Scale bars: 1 μm. (F,G) Average FISH and SMC2 (F, Figure 6—source data 1) or CAPH1 (G, Figure 6—source data 2) signal intensity profiles along the insert-bearing chromosome of F1.1 from several images (n = 15, 40). SMC2 and CAPH1 intensity levels were normalised to the 0–5% region of the chromosome, corresponding to the mouse centromere. (H,I) Normalisation of the SMC2 (H) and CAPH1 (I) profiles to DNA levels as measured by PI intensity. FISH intensities remain unaltered. Error bars represent ± SEM. Values were compared between a region of endogenous mouse DNA and the region of S. pombe DNA highlighted by FISH by the KS test (**p<0.001, n.s. = not significant).
-
Figure 6—source data 1
FISH and anti-SMC2 intensity measurements for Figure 6F.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig6-data1-v3.xlsx
-
Figure 6—source data 2
FISH and anti-SMC2 intensity measurements for Figure 6G.
- https://cdn.elifesciences.org/articles/57212/elife-57212-fig6-data2-v3.xlsx
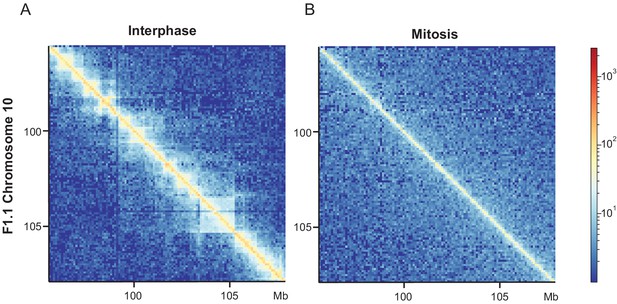
Loss of long-range interphase chromatin contacts in F1.1 mitotic cells.
Relative Hi-C contact probability heatmaps for the same region on mouse chromosome 10 in F1.1 cells in interphase (A) and mitosis (B).
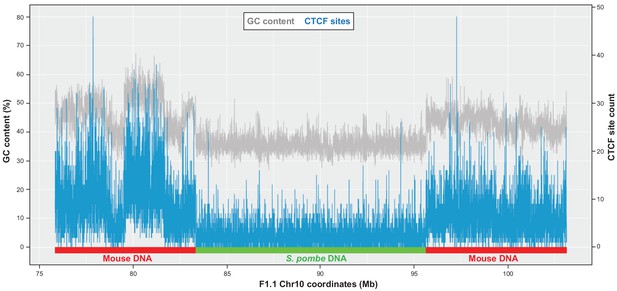
CTCF sites are not enriched over the Sp-DNA insert.
Predicted number of CTCF consensus motifs and GC content plotted across the S. pombe DNA present in the F1.1 genome and flanking mouse regions on chromosome 10. Window size is 3 kb.
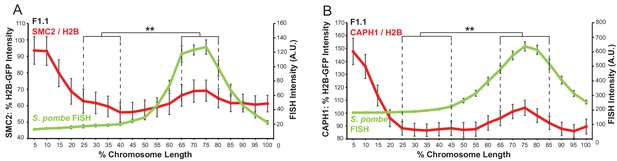
Condensin levels increase relative to chromatin over the region of S. pombe DNA insertion in a mammalian chromosome.
(A,B) Normalisation of the overall condensin (SMC2, A) and condensin I (CAPH1, B) profiles to chromatin levels as measured by H2B-GFP fluorescence intensity across the F1.1 metaphase chromosome 10 bearing S. pombe DNA. FISH intensities remain unaltered. Error bars represent ± SEM. Intensity ratios were compared between a region of endogenous mouse DNA and the region of S. pombe DNA highlighted by FISH by the KS test (**p<0.001).
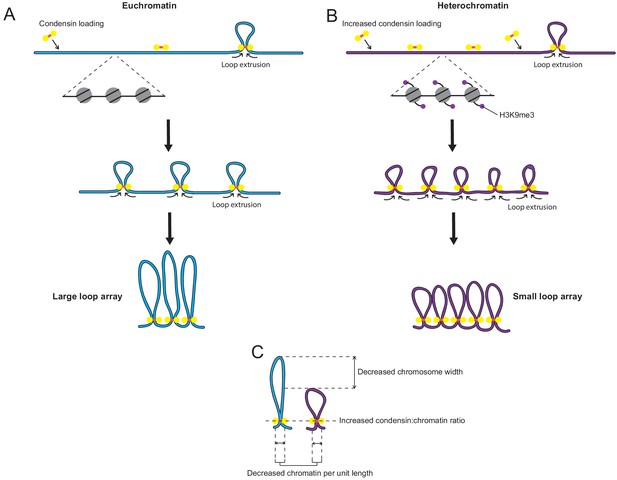
Model: Increased loading of condensin on heterochromatin leads to smaller chromatin loops on the mitotic chromosome.
(A) Diagram of regular chromatin organisation by loop extrusion in mitosis. Condensin binds to the chromatin fibre and extrudes loops until blocked by other condensin complexes on either side. This results in an array of large side-by-side loops organised around a condensin scaffold. (Goloborodko et al., 2016; Gibcus et al., 2018). (B) When condensin loading is increased over heterochromatin loop extrusion is halted earlier by the greater concentration of condensin complexes present on the chromatin fibre. This results in an array of smaller loops. (C) This small-loop array results in a decrease in chromatin per unit length, an increase in the ratio of condensin to chromatin and a narrowing of the chromosome width.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Cell line (M. musculus) | C127 (female, mammary tumour) | ATCC | CRL-1804; | |
| Cell line (M. musculus) | F1.1 | Allshire et al., 1987 | Kept as cryopreserved stocks in the Allshire lab | |
| Cell line (M. musculus) | NIH3T3 | ATCC | CRL-1858 | |
| Cell line (H. sapiens) | HeLa | ATCC | CCL-2 | |
| Cell line (M. musculus) | NP-F1 | This paper | NIH3T3-S. pombe fusion cell line. Aka NPA4. Available from Allshire lab. | |
| Cell line (M. musculus) | NP-F2 | This paper | NIH3T3-S. pombe fusion cell line. Aka NPF3-19. Available from Allshire lab. | |
| Cell line (M. musculus) | NP-T1 | This paper | NIH3T3-S. pombe DNA transfection cell line. Aka NPD3. Available from Allshire lab. | |
| Cell line (M. musculus) | NP-T2 | This paper | NIH3T3-S. pombe DNA transfection cell line. Aka NPD5. Available from Allshire lab. | |
| Cell line (M. musculus) | NP-T3 | This paper | NIH3T3-S. pombe DNA transfection cell line. Aka NPT-C606. Available from Allshire lab. | |
| Cell line (M. musculus) | NP-T4 | This paper | NIH3T3-S. pombe DNA transfection cell line. Aka CPT-C500. Available from Allshire lab. | |
| Cell line (M. musculus) | NP-T5 | This paper | NIH3T3-S. pombe DNA transfection cell line. Aka NPT-C480. Available from Allshire lab. | |
| Cell line (M. musculus) | NP-T6 | This paper | NIH3T3-S. pombe DNA transfection cell line. Aka NPT-C482. Available from Allshire lab. | |
| Cell line (M. musculus) | NP-clr4Δ-F4 | This paper | NIH3T3-S. pombe fusion cell line. Aka NPF-Clr-I. Available from Allshire lab. | |
| Cell line (H. sapiens) | HeP-F3 | This paper | HeLa-S. pombe fusion cell line. Aka HeP3. Available from Allshire lab. | |
| Strain, strain background (S. pombe) | FY43 | Allshire et al., 1987 | h-Int5(pUraSV2Neo) ade6-210 leu1-32 ura4-D18 | |
| Antibody | Anti-H3K9me3 (rabbit polyclonal) | Active Motif | 39161; RRID:AB_2532132 | ChIP(4 µg/mL) IF(1:1000) |
| Antibody | Anti-H3K4me3 (rabbit polyclonal) | Active Motif | 39159; RRID:AB_2615077 | ChIP(4 µg/mL) |
| Antibody | Anti-H3K36me3 (rabbit polyclonal) | Active Motif | 61101; RRID:AB_2615073 | ChIP(4 µg/mL) |
| Antibody | Anti-H3K9ac (rabbit polyclonal) | Active Motif | 39137; RRID:AB_2561017 | ChIP(4 µg/mL) |
| Antibody | Anti-H3K27me3 (rabbit polyclonal) | Abcam | Ab4729; RRID:AB_2118291 | ChIP(4 µg/mL) |
| Antibody | Anti-HP1alpha (goat polyclonal) | Abcam | Ab77256; RRID:AB_1523784 | ChIP(4 µg/mL) IF(1:500) |
| Antibody | Anti-SMC2 (rabbit polyclonal) | Losada Lab | IF(1:1000); Obtained from Ana Losada | |
| Antibody | Anti-SMC3 (rabbit polyclonal) | Losada Lab | IF(1:1000); Obtained from Ana Losada | |
| Antibody | Anti-H4 (rabbit polyclonal) | Sigma-Aldrich | SAB4500312; RRID:AB_10743081 | IF(1:1000) |
| Antibody | Anti-CAPH1 (rabbit polyclonal) | Abcam | Ab154105 | IF(1:1000) |
| Antibody | Anti-rabbit-A594 (donkey polyclonal) | Invitrogen | A21207; RRID:AB_141637 | IF(1:10000) |
| Antibody | Avidin-FITC | Vector Labs | A-2001–5; RRID:AB_2336455 | FISH(1:500) |
| Antibody | Biotinylated anti-Avidin (goat polyclonal) | Vector Labs | BA-0300-.5; RRID:AB_2336108 | FISH(1:100) |
| Antibody | Texas Red anti-sheep (rabbit polyclonal) | Vector Labs | TI-6000; RRID:AB_2336219 | FISH(1:100) |
| Antibody | Rhodamin anti-dig (sheep polyclonal) | Roche | 11207750910; RRID:AB_514501 | FISH(1:20) |
| Recombinant DNA reagent | H2B-GFP | Addgene | 11680 | |
| Commercial assay or kit | EpiTect Bisuflite kit | Qiagen | 59104 | |
| Chemical compound, drug | LightCycler 480 SYBR Green Master Mix | Roche | 04707516001 | |
| Software, algorithm | FIMO | Grant et al., 2011 | ||
| Software, algorithm | HiCExplorer | Ramírez et al., 2018 |





