The CD73 immune checkpoint promotes tumor cell metabolic fitness
Abstract
CD73 is an ectonucleotidase overexpressed on tumor cells that suppresses anti-tumor immunity. Accordingly, several CD73 inhibitors are currently being evaluated in the clinic, including in large randomized clinical trials. Yet, the tumor cell-intrinsic impact of CD73 remain largely uncharacterized. Using metabolomics, we discovered that CD73 significantly enhances tumor cell mitochondrial respiration and aspartate biosynthesis. Importantly, rescuing aspartate biosynthesis was sufficient to restore proliferation of CD73-deficient tumors in immune deficient mice. Seahorse analysis of a large panel of mouse and human tumor cells demonstrated that CD73 enhanced oxidative phosphorylation (OXPHOS) and glycolytic reserve. Targeting CD73 decreased tumor cell metabolic fitness, increased genomic instability and suppressed poly ADP ribose polymerase (PARP) activity. Our study thus uncovered an important immune-independent function for CD73 in promoting tumor cell metabolism, and provides the rationale for previously unforeseen combination therapies incorporating CD73 inhibition.
Editor's evaluation
This important study demonstrates a non-canonical, cancer-cell intrinsic role of the ectonucleotidase CD73 in the regulation of cancer cell metabolism. The evidence supporting the claims is convincing and of high quality.
https://doi.org/10.7554/eLife.84508.sa0Introduction
CD73 is an ectonucleotidase that catalyzes the phosphohydrolysis of adenosine monophosphate (AMP), thus contributing to the breakdown of pro-inflammatory adenosine triphosphate (ATP) into immunosuppressive ADO. Accumulation of extracellular ADO in the tumor microenvironment favors cancer progression by activating ADO specific G-coupled protein receptors (GPCRs) expressed on immune cells, endothelial cells, fibroblasts and tumor cells. Of the four ADO receptors, high affinity A2A and low affinity A2B receptors elevate intracellular cyclic AMP (cAMP) levels and contribute to suppress immune cell function. In human cancers, such as triple negative breast cancer (TNBC) and ovarian cancer, CD73 expression is generally associated with poor clinical outcomes, impaired tumor immune surveillance and resistance to chemotherapy (Loi et al., 2013; Bareche et al., 2021). Potent and selective CD73 inhibitors have recently been developed and are now being evaluated in randomized clinical trials with encouraging preliminary results reported in lung cancer and pancreatic cancer patients (Allard et al., 2020; Augustin et al., 2022).
While CD73 inhibitors can stimulate anti-tumor immunity, early studies reported that targeting CD73 can also inhibit human tumor xenografts in severely immunocompromised mice (Zhou et al., 2007; Zhi et al., 2007; Zhi et al., 2010; Rust et al., 2013). Moreover, pharmacological inhibition or gene-deletion of CD73 has also been observed to increase the sensitivity of CD73-expressing tumor cells to cytotoxic drugs in vitro (Loi et al., 2013; Ujházy et al., 1996; Yu et al., 2021). Although activation of pro-survival pathways downstream of A2B receptors has been proposed to explain immune-independent effects of CD73, alternative hypotheses have not been explored (Mittal et al., 2016; Lan et al., 2018).
Rewiring of cancer cells’ metabolism is central to their adaptation to hostile tumor microenvironments (Zhi et al., 2010). While cancer cells can shift their metabolism from highly efficient ATP-producing oxidative phosphorylation (OXPHOS) to low ATP-yielding glycolysis despite presence of oxygen (e.g. Warburg effect), mitochondrial respiration has been shown to be critical for proliferating cells and for maintaining tumor cells invasiveness, metabolic adaptation, chemoresistance and stemness. Accordingly, tumor cells are not only capable of respiration but often require respiration for survival (Ashton et al., 2018).
In proliferating cells, mitochondrial respiration provides electron acceptors to support biosynthesis of aspartate, an indispensable amino acid for nucleotide and protein synthesis. Mitochondrial respiration requires nicotinamide adenine dinucleotide (NAD) in its oxidized and reduced forms to allow movement of electrons through the electron transport chain (ETC). In addition of supporting respiration, NAD is also an important co-substrate for many enzymes, notably poly-ADP-ribose polymerases (PARPs) that play a central role in genomic stability by catalyzing the addition of poly-ADP-ribose to proteins (also known as PARylation; Wei and Yu, 2016; Pommier et al., 2016).
Intracellular NAD levels are maintained either by de novo synthesis from L-tryptophan (Trp), or by more effective salvage pathways that recycle NAD from nicotinamide (NAM), nicotinamide mononucleotide (NMN), or nicotinamide riboside (NR) (Verdin, 2015). Extracellular NMN is dephosphorylated to NR before cellular internalization by equilibrative nucleotide transporters (ENTs). Intracellular NR is then re-phosphorylated by NRK1 into NMN, a substrate for adenylyl transferases (NMNATs) to generate NAD (Ratajczak et al., 2016; Katsyuba et al., 2020). NMN can also be generated from intracellular or extracellular NAM via the rate-limiting enzyme NAMPT (Katsyuba et al., 2020). Interestingly, some studies have suggested a role for CD73 in NAD synthesis from extracellular NR (Garavaglia et al., 2012; Mateuszuk et al., 2020). Yet, the impact of CD73 on tumor cell metabolic pathways remains unknown (Grozio et al., 2013).
We here investigated metabolic consequences of targeting CD73 in a panel of human and murine cancer cells. We observed that CD73 deficiency or pharmacological inhibition induces an important aspartate-dependent metabolic vulnerability in tumor cells characterized by suppressed mitochondrial respiration and increased genomic instability. Our study thus provides a rationale for exploring novel combination strategies with anti-CD73 therapies, such as PARP inhibition and mitochondria targeting agents.
Results
CD73 favors tumor growth independently of its immunosuppressive function
We evaluated the impact of tumor cell-associated CD73 by transiently transfecting a CRISPR-Cas9 construct into human and mouse tumor cell lines followed by flow cytometry-based sorting of polyclonal CD73-negative (CD73neg) and CD73-positive (CD73pos) fractions (Figure 1—figure supplement 1A–F). Consistent with prior work (Rust et al., 2013) and in support of immune-independent effects, CD73 deletion in MDA-MB-231 human triple negative breast cancer (TNBC) cells significantly delayed tumor growth in severely immunodeficient Nod-Rag-gamma (NRG) mice, which lack T cells, B cells, NK cells and functional macrophages (Figure 1A). To further characterize the immune-independent effects of CD73, we next compared the in vitro proliferation rates of CD73pos and CD73neg MDA-MB-231 cells. While NT5E (gene encoding for CD73) gene deletion had no impact in standard cell culture conditions (i.e. 25 mM glucose and 2 mM glutamine), it significantly suppressed cell proliferation and viability in nutrients-limiting conditions (Figure 1B, Figure 1—figure supplement 2), despite no difference in cellular uptake of glucose or glutamine, as shown using a fluorescent glucose analog (i.e. 2-NDBG; Figure 1C) and by measurements of glutamine depletion in media (Figure 1D).
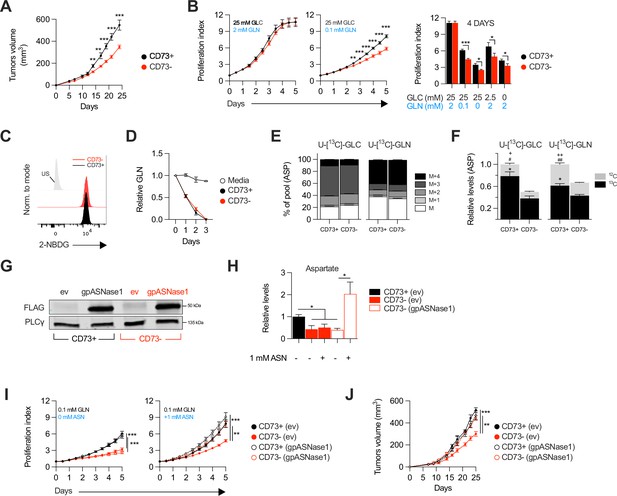
CD73-deficiency delays tumor growth independently from immune suppression through reduced aspartate biosynthesis.
(A) Sub-cutaneous tumor growth of CD73pos (CD73+) and CD73neg (CD73-) MDA-MB-231 tumors in immune-deficient NRG mice. Results shows combined data of two independent experiments cumulating 15 mice per groups. (B) In vitro proliferation of CD73 + and CD73- MDA-MB-231 cells was analyzed by Incucyte live imaging technology in presence of excess or restricted conditions of glucose (GLC) and glutamine (GLN). Data from representative proliferation curves (left panels) and pooled quantification of proliferation after 4 days of culture (right panel) are shown (n=3). (C) FACS analysis of in vitro incorporation of a fluorescent glucose analog (2-NBDG) in CD73 + and CD73- MDA-MB-231 cells (n=3). US = unstained control. (D) Glutamine depletion assay in culture media of MDA-MB-231 CD73 + and CD73- cells measured using a commercial kit (Promega). Cell-free media was used as a control. (E) Mass isotopomer distribution (MID) of U-[13C]-glucose-derived (U-[13C]-GLC; left panel) or U-[13C]-glutamine-derived (U-[13C]-GLN; right panel) aspartate compared between CD73 + and CD73- MDA-MB-231 cells (n=1). M represents the pool of unlabeled aspartate. M+1, M+2, M+3 and M+4 represent the pools of isopotologues labeled with 13C carbon. (F) SITA analysis of glucose (left panel) and glutamine (right panel) contribution to ASP intracellular biosynthesis (n=1). ASP levels were normalized on cell number and are shown relative to CD73 + cells. Stats: *comparison of 13C levels; #Comparison of 12C levels; + Comparison of total 13C+12C levels. (G) Western blot of CD73 + and CD73- MDA-MB-231 cells expressing empty vector (ev) or FLAG-tagged gpASNase1. PLCγ is shown as a loading control. (H) Intracellular ASP levels measured in cells expressing or not (ev) the gpASNase1 upon exposure to exogenous ASN (1 mM) for 24 h. ASP levels were normalized on cell number and are shown relative to CD73+ (ev) cells (n=1). (I) In vitro proliferation of gpASNase1-expressing and non-expressing (ev) MDA-MB-231 cells in presence (right panel) or not (left panel) of exogenous ASN (n=2). (J) In vivo tumor growth of 2×106 gpASNase1-expressing or not (ev) CD73 + and CD73- MDA-MB-231 cells in NRG mice (CD73+ (ev) n=5; CD73- (ev) n=7; CD73+ (gpASNase1) n=8; CD73- (gpASNase1) n=8). Means +/- SEM are shown (*p<0.05; **p<0.01; ***p<0.001 by Student T tests).
-
Figure 1—source data 1
Original raw unedited blots and uncropped blots with the relevant bands labeled that composes Figure 1G.
- https://cdn.elifesciences.org/articles/84508/elife-84508-fig1-data1-v3.zip
-
Figure 1—source data 2
Stable isotope tracing analysis (SITA) raw data for both U-[13C]-glucose and U-[13C]-glutamine tracing in MDA-MB-231 CD73 +and CD73- cells that composes Figure 1E–F and Figure 1—figure supplement 3.
- https://cdn.elifesciences.org/articles/84508/elife-84508-fig1-data2-v3.zip
CD73-deficiency impairs aspartate biosynthesis and tumor growth
We hypothesized that CD73 was involved in promoting tumor cell metabolism. To test this, we performed stable isotope tracer analysis (SITA) of labeled glucose or glutamine in CD73pos and CD73neg MDA-MB-231 cells. Strikingly, while the isopotomers distribution and relative abundance of most intermediate metabolites were unaltered (Figure 1—figure supplement 3), aspartate biosynthesis from glucose or glutamine (Figure 1E) was significantly reduced – by nearly 50% – in CD73neg tumor cells (Figure 1F).
To assess whether this decrease in aspartate biosynthesis was responsible for the suppressed growth of CD73-deficient tumor cells, we tested the ability of aspartate supplementation to restore proliferation of CD73neg MDA-MB-231 cells. We used a strategy developed by Sullivan et al., 2018, whereby an asparaginase enzyme (i.e. gpASNase1) is introduced into cells to restore intracellular aspartate levels upon exposure to exogenous asparagine (since exogenous aspartate cannot be readily incorporated into cells). Stable expression of gpASNase1 (Figure 1G) did not affect CD73 expression or enzymatic activity (Figure 1—figure supplement 4). Asparagine treatment of gpASNase1-expressing cells indeed significantly increased intracellular aspartate levels (Figure 1H) and effectively restored proliferation of CD73neg MDA-MB-231 tumor cells, both in vitro in glutamine-restricted conditions (Figure 1I) and in vivo in immunodeficient NRG mice (Figure 1J). Taken together, our results demonstrated that CD73-deficiency impaired tumor growth via impaired aspartate biosynthesis.
CD73 regulates metabolic fitness of cancer cells
Since aspartate biosynthesis is an essential function of mitochondrial respiration (Birsoy et al., 2015; Garcia-Bermudez et al., 2018; Sullivan et al., 2015), and that aspartate fuels OXPHOS (Cheng et al., 2018), we evaluated the impact of CD73 on mitochondrial respiration. For this, we measured oxygen consumption rate (a surrogate of OXPHOS) and extracellular acidification rate (a surrogate of glycolysis) in response to standard mitochondrial stress tests (Figure 2A). Strikingly, CD73neg MDA-MB-231 cells displayed significantly reduced OXPHOS (Figure 2B) and reduced glycolytic reserve compared to CD73pos cells in both glutamine replete and glutamine limiting conditions (Figure 2C, Figure 2—figure supplement 1). In addition, blocking CD73 with a neutralizing mAb (Häusler et al., 2014) also significantly suppressed OXPHOS and glycolytic reserve (Figure 2B–C). Importantly, similar results were obtained in additional murine cell lines (4T1, SM1 LWT1), human cell lines (SK23MEL, PANC1, SKOV3, and HCC1954) and in primary murine embryonic fibroblasts (MEF) derived from CD73-deficient mice (Figure 2D–H, Figure 2—figure supplement 2A–B).
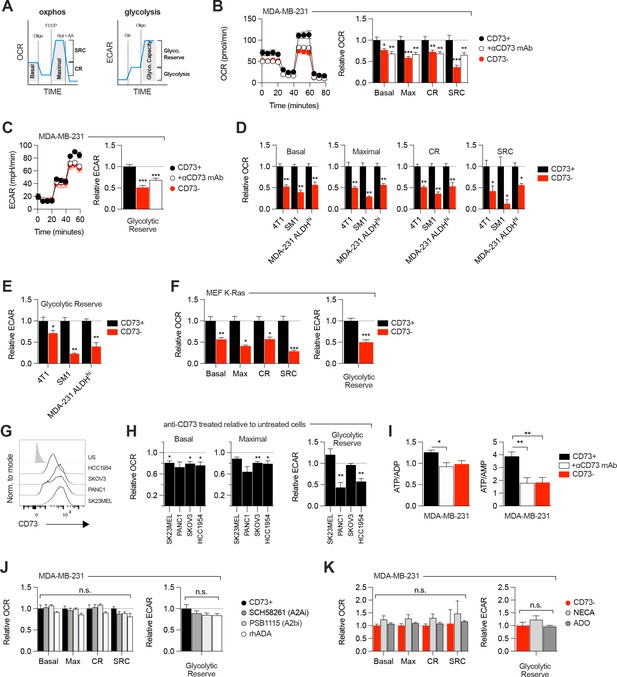
CD73 regulates metabolic fitness of cancer cells independently from adenosine signaling.
(A) Schematized metabolic profiling and parameters generated on a Seahorse analyzer. OXPHOS and glycolytic parameters were calculated using Agilent calculator tool. Rot = rotenone, AA = antimycin A, Oligo = oligomycin, Glc = glucose, OCR = oxygen consumption rate, ECAR = extracellular acidification rate, SCR = spare respiratory capacity which represents the difference between maximal OCR and basal OCR (indicator of cell fitness/flexibility), CR = coupled-respiration which represents the decrease of basal respiration upon inhibition of ATP synthase (indicator of basal respiration used for ATP production to meet energetic needs of the cells). (B) OXPHOS profile (left panel) and parameters (right panel) of CD73pos (CD73+;+/-1 μg/mL anti-CD73 mAb 7G2 for 48 hr) and CD73neg (CD73-) MDA-MB-231 cells. OXPHOS parameters are shown relative to CD73 + cells from pooled experiments (n=3). (C) Glycolytic profile (left panel) and glycolytic reserve (right panel) of CD73+ (+/-1 μg/mL anti-CD73 mAb 7G2 for 48 hr) and CD73- MDA-MB-231 cells. Glycolytic reserve is shown relative to CD73 + cells from pooled experiments (n=3). (D–E) Oxphos parameters (D) and glycolytic reserve (E) compared between various CD73-expressing and CD73 CRISPR-knockout cell lines. ALDHhi cells were sorted from MDA-MB-231 cells using the Aldefluor kit. Data are shown relative to CD73 + cells (MDA-231 ALDHhi n=1, 4T1 and SM1 LWT1 n=2). (F) OXPHOS parameters (left panel) and glycolytic reserve (right panel) compared between primary MEF isolated from WT and CD73-/- C57BL/6 mice and transformed with an oncogenic K-Ras. Data are shown relative to WT cells (n=2). (G) CD73 expression profile on various human cancer cell lines analyzed by FACS. US = unstained control. (H) OXPHOS parameters (left and middle panels) and glycolytic reserve (right panel) of anti-CD73 mAb (7G2)-treated cells compared to untreated cells. Data are shown relative to respective untreated cells (n=1 per cell line). (I) LC-MS analysis of total intracellular levels of ATP/ADP and ATP/AMP ratios in CD73+ (+/-1 μg/mL anti-CD73 mAb 7G2 for 48 h) and CD73- MDA-MB-231 cells (n=2). (J) OXPHOS parameters (left panel) and glycolytic reserve (right panel) of CD73 + MDA MB-231 cells treated with either A2A inhibitor SCH 58261, A2B inhibitor PSB 1115 or human recombinant ADA. Data are shown relative to untreated CD73 + cells (A2ai and A2bi n=2, rhADA n=1). (K) OXPHOS parameters (left panel) and glycolytic reserve (right panel) of NECA- or ADO-treated CD73- MDA-MB-231 cells. Data are shown relative to untreated CD73- cells (NECA n=2, ADO n=1). Means +/- SEM are shown (*p<0.05; **p<0.01; ***p<0.001 by Student T tests).
Consistent with the observed defect in mitochondrial respiration, CD73 NT5E gene-targeted tumor cells also displayed reduced ratios of ATP to ADP or AMP (Figure 2I, Figure 2—figure supplement 3A). However, no difference was observed in GOT1 and GOT2 mRNA expression, which encode enzymes that generate aspartate from oxaloacetate (Birsoy et al., 2015; Figure 2—figure supplement 3B), or other tricarboxylic acid (TCA) cycle-related enzymes (Figure 2—figure supplement 3B).
As CD73 is overexpressed in tumor-initiating cells (TIC) to promote stemness (Katsuta et al., 2016; Song et al., 2017), and that TIC rely on OXPHOS to support tumorigenesis (Snyder et al., 2018), we next evaluated the impact of CD73 on the metabolic fitness of putative TIC (ALDHHigh CD44+ CD24-) derived from MDA-MB-231 cell cultures (Figure 2—figure supplement 2C). Similar to the bulk culture, CD73neg TIC also displayed significantly suppressed OXPHOS and reduced glycolytic reserve compared to CD73pos TIC (Figure 2D–E). Taken together, our data demonstrated that CD73 plays a critical role in promoting mitochondrial respiration in proliferating tumor cells.
ADO-independent metabolic functions of CD73
We next evaluated whether the metabolic functions of CD73 were mediated by ADO receptor signaling. MDA-MB-231 cells express A2A and A2B receptors (Figure 1—figure supplement 1F; Fernandez-Gallardo et al., 2016). Nevertheless, treatment with an A2A antagonist (SCH58261) or an A2B antagonist (PSB1115) had no effect on mitochondrial respiration or glycolysis (Figure 2J). To rule-out a potential role for other ADO receptors or ADO intracellular uptake, we treated MDA-MB-231 cells with recombinant adenosine deaminase (rhADA) or the pan-ADO receptor agonist NECA. Depletion of extracellular ADO with rhADA (Figure 2J), or treatment with NECA (Figure 2K), also had no impact on mitochondrial respiration or glycolysis of MDA-MB-231 cells. Our results thus indicated that CD73 promoted metabolic fitness independently of ADO signaling.
CD73 contributes to metabolic fitness of cancer cells through NAD synthesis
NAD + is a coenzyme that plays a central role in mitochondrial respiration (Verdin, 2015). We thus investigated whether CD73 could regulate intracellular NAD levels. Using mass-spectrometry (LC-MS), we observed that NT5E gene-targeting or antibody-mediated CD73 enzymatic inhibition was associated with a significant reduction of intracellular NAD + levels in MDA-MB-231 (Figure 3A) or 4T1.2 cells (Figure 3—figure supplement 1A). Depleting extracellular ADO with exogenous rhADA had no effect on intracellular NAD + levels, further supporting an ADO-independent mechanism (Figure 3A).
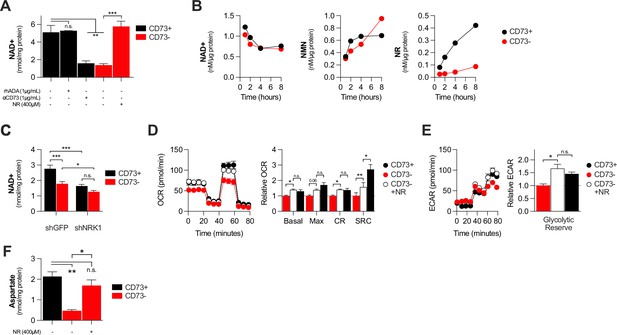
CD73 contributes to metabolic fitness of cancer cells through NAD synthesis.
(A) LC-MS analysis of intracellular nicotinamide adenine levels (NAD+) normalized to protein content of CD73pos (CD73+;+/-1 μg/mL αCD73 mAb or 1 μg/mL rhADA for 48 hr) and CD73neg (CD73-;+/-100 μM NR supplemented 2 x per day for 48 hr) MDA-MB-231 cells (n=2). (B) LC-MS analysis of endogenous extracellular production of NAD+ (left panel), NMN (middle panel) and NR (right panel) in supernatant of CD73 +and CD73- MDA-MB-231 cells. Levels were normalized on protein content of adherent cells (n=1). (C) LC-MS analysis of intracellular nicotinamide adenine levels (NAD+) normalized to protein content of CD73 + and CD73- MDA-MB-231 cells transfected with either shGFP or shNRK1 (n=2). (D) Oxygen consumption rate (OCR) profile (left panel) and OXPHOS parameters (right panel) of CD73- MDA-MB-231 cells cultured with 400 μM NR for 48 hr (n=3). CD73 + cells are shown as a control. (E) Extracellular acidification rate (ECAR) profile (left panel) and glycolytic reserve (right panel) of CD73- MDA-MB-231 cells cultured with 400 μM NR for 48 hr (n=3). CD73 + cells are shown as a control. (F) Intracellular aspartate levels measured in MDA-MB-231 CD73- cells exposed to exogenous NR (400 µM) for 48 hr. ASP levels were normalized on cell number. CD73 + cells are shown as a control. Means +/- SEM are shown (*p<0.05; **p<0.01; ***p<0.001 by one-way ANOVA).
We next tested whether CD73 can serve tumor cells to generate NR, a precursor for NAD synthesis (Ratajczak et al., 2016). Using purified recombinant human CD73 or cell surface CD73, in presence or absence of the CD73 inhibitor APCP, we confirmed hydrolysis of exogenous NMN to NR by mass spectrometry (Figure 2—figure supplement 2B–D). We then tested the impact of CD73 on the extracellular accumulation of NAD+, NMN and NR from MDA-MB-231 cell cultures. Strikingly, we observed that MDA-MB-231 cell cultures accumulated extracellular NMN and rapidly hydrolyzed it to NR in a CD73-dependent manner (Figure 3B). In contrast, CD73 had no impact on extracellular NAD + levels (Figure 3B).
Upon intracellular transport, NR must be phosphorylated by intracellular NRK1 to promote intracellular NAD synthesis (Ratajczak et al., 2016). We thus tested whether NRK1 was involved in CD73-mediated NAD synthesis. In support of a metabolic role for CD73-mediated hydrolysis of extracellular NMN to NR, shRNA-mediated knockdown of NRK1 (Figure 2—figure supplement 2E) prevented CD73 from increasing intracellular NAD + levels in MDA-MB-231 cells (Figure 3C). NRK1-deficiency did reduce NAD + levels in both CD73-proficient and CD73-deficient cells, suggesting that some intracellular NR may come from other sources (Figure 3C).
To further assess the importance of NR production for CD73 function, we tested whether exogenous NR could rescue the metabolic dysfunctions of CD73neg tumor cells. Indeed, treatment of CD73neg MDA-MB-231 tumor cells with exogenous NR for 48 hr significantly increased their intracellular NAD + levels (Figure 3A), their oxygen consumption (Figure 3D) and glycolytic reserves (Figure 3E), to levels similar to CD73-proficient cultures. Finally, further supporting a role for CD73-NAD axis in mitochondrial metabolism we found that NR supplementation of CD73-deficient cells restored aspartate synthesis (Figure 3F). Altogether, our results support a model whereby CD73-dependent extracellular NR promotes intracellular NAD biosynthesis, which in turn enhances mitochondrial respiration and aspartate biosynthesis.
CD73 deficiency decreases PARP activity and sensitizes cancer cells to DNA-damaging agents
In addition of being essential for mitochondrial respiration, NAD is also an important co-factor for PARP enzymes that maintain genomic stability by promoting DNA repair. Interestingly, early studies reported that tumor-derived CD73 can promote chemoresistance (Loi et al., 2013; Ujházy et al., 1996; Yu et al., 2021). To further our understanding of the mechanism of action by which CD73 regulates chemoresistance, we hypothesized that by increasing intracellular NAD levels, CD73 may regulate PARP activity and therefore promote genomic stability. To assess this, we measured PARylation levels by western blotting in H2O2-treated MDA-MB-231 cells. As shown in Figure 4, CD73-deficient tumor cells displayed significantly reduced PARylation compared to CD73-proficient tumor cells (Figure 4A). Moreover, targeting CD73 with a neutralizing mAb also reduced PARylation levels, and NR supplementation rescued PARylation in CD73neg cells (Figure 4B–C). Similar results were obtained in UWB1.289 human ovarian cancer cells (Figure 4—figure supplement 1A–B).
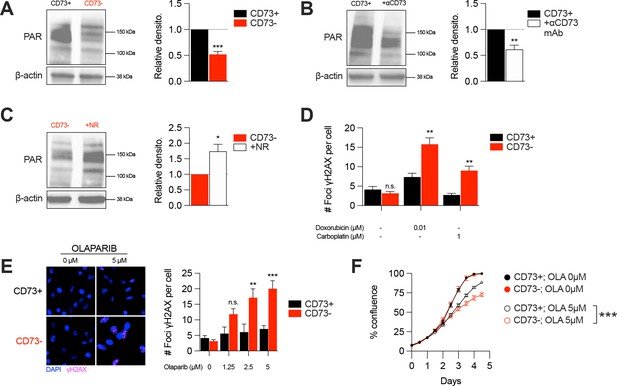
CD73 deficiency decreases PARP activity and sensitizes cancer cells to DNA-damaging agents.
(A) Western blot (left panel) showing PARylation levels in CD73pos (CD73+) and CD73neg (CD73-) MDA-MB-231 cells. Densitometry (right panel) is shown relative to CD73 + cells (n=5). (B) Western blot (left panel) showing PARylation levels in CD73 + MDA MB-231 cells treated with αCD73 mAb (1 μg/mL; clone 7G2; 48 hr). Densitometry (right panel) is shown relative to untreated CD73 + cells (n=2). (C) Western blot (left panel) showing PARylation levels in CD73- MDA-MB-231 cells treated with NR (400 μM; 48 hr). Densitometry (right panel) is shown relative to untreated CD73- cells (n=2). (D) Number of γH2AX foci per CD73 +and CD73- MDA-MB-231 cells treated with doxorubicin (0.01 μM) or carboplatin (1 μM) for 48 hr (n=2). (E) γH2AX staining of CD73 + and CD73- MDA-MB-231 cells treated with olaparib (0–5 μM) for 48 hr. Left panels show representative images for γH2AX (magenta) and DAPI (blue) staining. Data show number of γH2AX foci per cells (right panel; n=2). (F) In vitro proliferation of CD73 + and CD73- MDA-MB-231 cells treated with (0–5 μM) olaparib for 4 days. Confluence was measured using an Incucyte instrument (n=1). Means +/- SEM are shown (*p<0.05; **p<0.01; ***p<0.001 by Student T tests).
-
Figure 4—source data 1
Original raw unedited blots and uncropped blots with the relevant bands labeled that composes Figure 4A–C.
- https://cdn.elifesciences.org/articles/84508/elife-84508-fig4-data1-v3.zip
Given the impact of CD73 on PARylation, we next compared the levels of DNA damage in CD73pos and CD73neg tumor cells in response to cytotoxic drugs for TNBC or ovarian cancer. Strikingly, treatment of CD73-deficient MDA-MB-231 or UWB1.289 cells with doxorubicin and carboplatin was associated with significantly greater DNA damage compared to CD73-proficient cells (Figure 4D, Figure 4—figure supplement 1C). Since decreasing intracellular NAD levels has also been shown to sensitize tumor cells to PARP inhibitors (Bajrami et al., 2012), we also evaluated the impact of CD73 on olaparib activity. Accordingly, CD73-deficient MDA-MB-231 cells were significantly more sensitive to olaparib activity in vitro (Figure 4E–F). Altogether our results suggest that by regulating intracellular NAD levels, CD73 plays an important role in promoting PARP activity and maintaining genomic stability of tumor cells.
Discussion
In this study, we demonstrated that the ectonucleotidase CD73, often overexpressed on tumor cells and considered as an important cancer immune checkpoint, enhances tumor cell metabolic fitness in a cell-autonomous manner. CD73 promotes mitochondrial respiration, which generates aspartate required for nucleotide and protein synthesis, and maintains genomic stability notably by increasing the activity of NAD+-dependent PARP enzymes. Surprisingly, these metabolic functions of CD73 were independent of ADO signaling, instead relying on the hydrolysis of extracellular NMN to NR and its metabolism by NRK1.
The importance of CD73 for intracellular NAD synthesis has been controversial (Wilk et al., 2020). Using a colorimetric assay, (Sullivan et al., 2018) observed no significant effect of inhibiting or deleting CD73 on intracellular NAD concentrations. These discrepancies from our own data may stem from the different sensitivity of the assays (colorimetric versus LC-MS), the different nature of the inhibitors and the different cell lines studied. Notably, Wilk et al. studied MCF-7 cells that express >10-fold lower levels of CD73 compared to the cell lines we analyzed. Importantly, we validated the impact of CD73 on mitochondrial respiration in a large panel of human and mouse tumor cell lines, as well as in primary fibroblasts derived from CD73 gene-targeted mice. Moreover, the fact that NR supplementation restored intracellular NAD +content in CD73-deficient tumor cells, and that CD73 failed to increase NAD +levels in the absence of NRK1 (since NRK1 is necessary and rate-limiting for the generation of NAD +from exogenous NR Ratajczak et al., 2016), further support the notion that tumor cell-associated CD73 contributes to maintain intracellular NAD pools.
Intriguingly, we observed that NMN accumulates in the extracellular milieu of proliferating MDA-MB-231 tumor cells. The mechanism leading to such extracellular NMN accumulation remains unclear. We ruled out the culture media as a potential source of NMN. Extracellular NMN may come from NAMPT activity (intracellular or extracellular) on nicotinamide (NAM) that reaches the extracellular milieu as a result of cell death or stress (Katsyuba et al., 2020). NAD release may be another source of extracellular NMN via active or passive release mechanisms followed by consumption by glycohydrolases to generate extracellular NAM. Finally, although MDA-MB-231 and 4T1.2 cells do not express CD38, we cannot exclude the involvement of other ecto-NADases.
In the tumor microenvironment, CD38 expression on immune cells may also contribute to consume extracellular NAD+. Production of NAM from NAD-consuming enzymes and NAMPT may thus lead to CD73-mediated NR production. Considering that CD38 can cooperate with CD73 for extracellular ADO production (Morandi et al., 2018), it would be of interest to assess the impact of host CD38 on CD73-mediated tumor cell metabolic fitness. Of note, while some studies suggested that CD73 may hydrolyze extracellular NAD+ (Garavaglia et al., 2012; Jablonska et al., 2021), we and others (Wilk et al., 2020) did not observe this.
Consistent with the observed decrease in intracellular NAD pools, and the importance of NAD for glycolysis (Hopp, 2019; Luengo et al., 2021), we found that CD73-deficient tumor cells also displayed impaired maximal glycolytic capacity following oligomycin treatment, which inhibits ATP synthase and forces cells to redirect pyruvate to lactate conversion via glycolysis (Luengo et al., 2021).
We also demonstrated that CD73-deficient tumor cells had significantly reduced PARP activity, which rely on NAD + as cofactor. Accordingly, targeting CD73 sensitized tumor cells to chemotherapy and PARP inhibition. Other DNA repairing enzymes besides PARPs also require NAD+ (Ruszkiewicz et al., 2022), including the sirtuins SIRT1 and SIRT6 that play critical roles in promoting homologous recombination (HR) and nonhomologous end joining (NHEJ) repairs. It would therefore be of interest to further evaluate the impact of CD73 on sirtuin-mediated HR and NHEJ. A broad impact of CD73 on DNA repair mechanisms might explain the observation that CD73-deficient tumor cells become sensitive to PARP inhibition despite expressing BRCA. Other studies have shown that targeting NAD +synthesis (by blocking NAMPT) enhances the activity of olaparib in BRCA1-competent TNBC cells (Bajrami et al., 2012). Alternatively, targeting NAD + synthesis may decrease BRCA1 expression or activity, as was previously reported (Li et al., 2014).
Interestingly, CD73 deficiency in mice was found associated with reduced serum arginine levels (Mierzejewska et al., 2019). In addition of diet, another source of arginine is through the metabolism of intracellular aspartate via cytosolic argininosuccinate synthase 1 (ASS1) (Garcia-Bermudez et al., 2020). Our study highlighted the importance of CD73 in promoting intracellular aspartate synthesis. It is therefore possible that the systemic reduction in arginine levels observed in CD73-deficient mice may stem from dysfunctional aspartate biosynthesis.
In conclusion, our study sheds new light on the immune-independent functions of CD73 (Figure 5). We demonstrated that CD73 promote tumor growth in a cancer-cell autonomous manner by increasing metabolic fitness. This in turn favors cancer cell adaptation to nutrient-deprived microenvironments and cytotoxic stress by increasing OXPHOS, aspartate biosynthesis and DNA repair. Our study highlights new therapeutic opportunities that may be exploited in novel combination anti-cancer strategies.
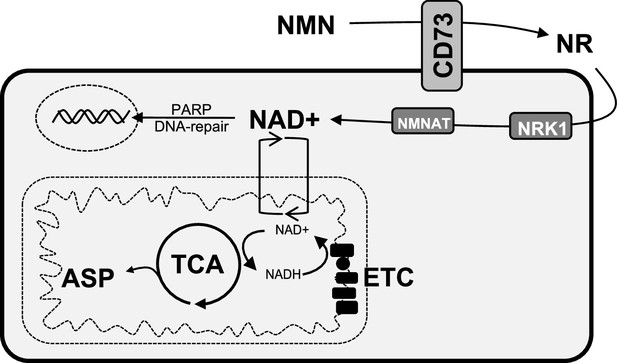
Schematic representation of the role of CD73 in NAD + biosynthesis, metabolic function, and genomic stability in cancer cells.
We propose that CD73 regulates to NAD + biosynthesis intracellularly in a NRK1-dependent manner by hydrolyzing extracellular NMN into NR, which in turn favors DNA-damage response, mitochondrial respiration, aspartate synthesis and tumor growth intrinsically. NMN = nicotinamide mononucleotide, NR = nicotinamide riboside, ASP = aspartate, ETC = electron transport chain.
Methods
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Antibody | Anti-human CD73 (7G2, mouse monoclonal) | Invitrogen | Cat#410200, RRID: AB_2533492 | Inhibition (1 µg/mL) |
| Antibody | Anti-human CD73 BV421 (mouse monoclonal) | BD Bioscience | Cat#562430, RRID: AB_11153119 | FACS (1:200) |
| Antibody | Anti-mouse CD73 PE-Cy7 (Rat monoclonal) | Thermo Fisher Scientific | Cat#25-0731-82, RRID: AB_10853348 | FACS (1:200) |
| Antibody | Anti-β-actin (mouse monoclonal) | Abcam | Cat#ab8226 | WB (1:5000) |
| Antibody | Anti-PLCγ (mouse polyclonal) | Millipore | Cat#05–163 | WB (1:2000) |
| Antibody | Anti-human CD73 (mouse monoclonal) | Abcam | Cat#Ab91086 | WB (1:1000) |
| Antibody | Anti-FLAG (rabbit polyclonal) | Millipore | Cat#F7425 | WB (1:1000) |
| Antibody | Anti-Cas9 S. pyogenes (rabbit monoclonal) | Cell Signaling Technology | Cat#19526 S | WB (1:1000) |
| Antibody | Anti-PAR (mouse monoclonal) | Trevigen | Cat#4335-MC-100 | WB (1:1000) |
| Antibody | HRP-conjugated anti-mouse IgG secondary (goat polyclonal) | Millipore | Cat#AP124P | WB (2.5:10,000) |
| Antibody | IRDye 800CW anti-Mouse IgG secondary (donkey polyclonal) | LI-COR | Cat#926–32212, RRID: AB_621847 | WB (1:10,000) |
| Antibody | IRDye 680RD anti-rabbit IgG secondary (donkey polyclonal) | LI-COR | Cat#926–68073, RRID: AB_10954442 | WB (1:10,000) |
| Antibody | Anti-phospho-Histone H2A.X Ser139 (mouse monoclonal) | Millipore | Cat#05–636 | IF (1:2000) |
| Antibody | Anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Alexa Fluor 488 (donkey polyclonal) | Thermo Fisher Scientific | Cat#A-21202, RRID: AB_141607 | IF (1:800) |
| Chemical compound, drug | Adenosine 5'-(α,β-methylene) diphosphate (APCP) | Sigma-Aldrich | Cat#M3763, CAS: 3768-14-7 | |
| Chemical compound, drug | Nicotinamide riboside (NR) | Cayman Chemical | Cat#23132, CAS: 1341-23-7 | |
| Chemical compound, drug | Nicotinamide mononucleotide (NMN) | Sigma-Aldrich | Cat#N3501, CAS: 1094-61-7 | |
| Chemical compound, drug | 5'-N-Ethylcarboxamidoadenosine (NECA) | Tocris | Cat#1691, CAS: 35920-39-9 | |
| Chemical compound, drug | SCH58261 | Tocris | Cat#2270, CAS: 160098-96-4 | |
| Chemical compound, drug | PSB1115 | Tocris | Cat#2009, CAS: 152529-79-8 | |
| Chemical compound, drug | Olaparib (AZD2281) | Selleckchem | Cat#S1060, CAS: 763113-22-0 | |
| Chemical compound, drug | Doxorubicin | CRCHUM pharmacy | CAS: 23214-92-8 | |
| Chemical compound, drug | Carboplatin | CRCHUM pharmacy | CAS: 41575-94-4 | |
| Chemical compound, drug | 2-deoxy-2-[(7-nitro-2,1,3- benzoxadiazol-4-yl)amino]- D-glucose (2-NBDG) | Cayman Chemical | Cat#11046, CAS: 186689-07-6 | |
| Chemical compound, drug | U-[13C]-glucose | Cambridge Isotope Laboratories | Cat#CLM-1396, CAS: 110187-42-3 | |
| Chemical compound, drug | U-[13C]-glutamine | Cambridge Isotope Laboratories | Cat#CLM-1822, CAS: 184161-19-1 | |
| Chemical compound, drug | Oligomycin | Sigma-Aldrich | Cat#O4876, CAS: 1404-19-9 | |
| Chemical compound, drug | Carbonyl cyanide p- trifluoromethoxyphenylhydrazone (FCCP) | Sigma-Aldrich | Cat#C2920, CAS: 370-86-5 | |
| Chemical compound, drug | Rotenone | Sigma-Aldrich | Cat#R8875, CAS: 83-79-4 | |
| Chemical compound, drug | Antimycin A from Streptomyces sp. | Sigma-Aldrich | Cat#A8674, CAS: 1397-94-0 | |
| Chemical compound, drug | CelLytic M buffer | Sigma-Aldrich | Cat#C2978 | |
| Chemical compound, drug | Halt protease and phosphatase cocktail inhibitors | Sigma-Aldrich | Cat#78440 | |
| Chemical compound, drug | Bradford protein assay dye reagent | Bio-Rad | Cat#500–0006 | |
| Chemical compound, drug | Protein Block DAKO solution | Agilent | Cat#X0909 | |
| Chemical compound, drug | Fluoromount Aqueous Mounting Medium | Sigma-Aldrich | Cat#F4680 | |
| Chemical compound, drug | Formalin solution, neutral buffered, 10% | Sigma-Aldrich | Cat#HT5011 | |
| Chemical compound, drug | Triton X-100 reduced | Sigma-Aldrich | Cat#X100RS; CAS: 92046-34-9 | |
| Chemical compound, drug | 7-amino-actinomycin D (7-AAD) | eBioscience | Cat#00-6993-50 | |
| Peptide, recombinant protein | Recombinant human adenosine deaminase (rhADA) | R&D systems | Cat#7048-AD, Acc#P00813 | |
| Peptide, recombinant protein | Recombinant human CD73 (rhCD73) | R&D systems | Cat#5795-EN, Acc#AAH659937 | |
| Commercial assay, kit | Malachite green phosphate detection kit | R&D systems | Cat#DY996 | |
| Commercial assay, kit | SuperScript VILO cDNA Synthesis Kit | Thermo Fisher Scientific | Cat#11754050 | |
| Commercial assay, kit | RNeasy mini kit | QIAGEN | Cat#74104 | |
| Commercial assay, kit | ALDEFLUOR kit | Stemcell | Cat#01700 | |
| Commercial assay, kit | Amersham ECL prime detection reagent | GE healthcare lifescience | Cat#RPN2232 | |
| Commercial assay, kit | Glutamine/Glutamate-Glo Assay kit | Promega | Cat#J8021 | |
| Cell line (homo-sapiens) | MDA-MB-231 (TNBC) | ATCC | HTB-26 | |
| Cell line (homo-sapiens) | PANC-1 (pancreatic cancer) | ATCC | CRL-1469 | |
| Cell line (homo-sapiens) | SK23MEL (metastatic melanoma) | MSK Cancer Center | RRID: CVCL_6027 | |
| Cell line (homo-sapiens) | HCC1954 (breast cancer) | ATCC | CRL-2338 | |
| Cell line (homo-sapiens) | OV1369 (ovarian cancer) | Dr. Anne-Marie Mes-Masson | Fleury et al., 2019 | |
| Cell line (homo-sapiens) | SKOV3 (ovarian cancer) | ATCC | HTB-77 | |
| Cell line (homo-sapiens) | HEK293FT (human embryonic kidney) | Dr. Francis Rodier | RRID: CVCL_6911 | |
| Cell line (homo-sapiens) | UWB1.289+BRCA1 | Dr. Madhuri Koti | RRID: CVCL_B078 | |
| Cell line (Mus musculus) | 4T1.2 (mammary cancer) | ATCC | CRL-2539 | |
| Cell line (Mus musculus) | SM1 LWT1 (metastatic melanoma) | Dr. Mark Smyth | ||
| Cell line (Mus musculus) | MEF from C57BL/6 J | This paper | Generated in Dr. Stagg’s laboratory | |
| Cell line (Mus musculus) | MEF from Nt5e-/- C57BL/6 J | This paper | Generated in Dr. Stagg’s laboratory | |
| Strain, strain background (mouse) | NOD.Cg-Rag1tm1Mom Il2rgtm1Wjl/SzJ (NRG) | Jackson Laboratory | JAX: 007799 | |
| Strain, strain background (mouse) | C57BL/6 J | Jackson Laboratory | JAX: 000664 | |
| Strain, strain background (mouse) | B6.129S1-Nt5etm1Lft/J (CD73-/- C57BL/6 J) | Dr. Linda Thompson | JAX: 018986 | |
| Sequence-based reagent | Nt5e | Thermo Fisher Scientific | Cat#4331182, Mm00501910_m1 | |
| Sequence-based reagent | Actb | Thermo Fisher Scientific | Cat#4331182, Mm00607939_s1 | |
| Sequence-based reagent | NMRK1 | Thermo Fisher Scientific | Cat#4331182, Hs00944470_m1 | |
| Sequence-based reagent | ACTB | Thermo Fisher Scientific | Cat#4331182, Hs99999903_m1 | |
| Sequence-based reagent | ADORA2A | Thermo Fisher Scientific | Cat#4331182, Hs00169123_m1 | |
| Sequence-based reagent | ADORA2B | Thermo Fisher Scientific | Cat#4331182, Hs00386497_m1 | |
| Recombinant DNA reagent | pLenti-PGK-ER- KRAS G12V (plasmid) | Dr. Francis Rodier | Rodier et al., 2009; Addgene#35635 | |
| Recombinant DNA reagent | pLHCX (plasmid) | Dr. Lucas Sullivan | Sullivan et al., 2018 | |
| Recombinant DNA reagent | pLHCX-gpASNase1 | Dr. Lucas Sullivan | Sullivan et al., 2018; Addgene#121526 | |
| Recombinant DNA reagent | shGFP | Sigma-Aldrich | TRCN0000075219 | |
| Recombinant DNA reagent | shNRK1 | Sigma-Aldrich | TRCN0000160469 | |
| Other | DMEM | Wisent | Cell culture reagent | |
| Other | Glucose/glutamine/sodium pyruvate-free DMEM | Wisent | Cell culture reagent | |
| Other | RPMI 1640 | Wisent | Cell culture reagent | |
| Other | OSE | Wisent | Cell culture reagent | |
| Other | Dialyzed FBS | Wisent | Cell culture reagent | |
| Other | Seahorse base media | Agilent | Cell culture reagent | |
| Other | Glutamax | Gibco | Cat#35050–061 | Cell culture reagent |
| Other | D-(+)-glucose (cell culture grade) | Sigma-Aldrich | Cat#G7021, CAS: 50-99-7 | Cell culture reagent |
| Other | L-glutamine (cell culture grade) | Sigma-Aldrich | Cat#G8540, CAS: 56-85-9 | Cell culture reagent |
| Software, algorithm | FlowJo (10.8.0) | FlowJo | RRID:SCR_008520 | |
| Software, algorithm | GraphPad Prism 9 (9.2.0) | GraphPad | RRID:SCR_002798 | |
| Software, algorithm | StepOne (2.3) | Thermo Fisher Scientific | RRID:SCR_014281 | |
| Software, algorithm | Incucyte base analysis software | Sartorius | RRID:SCR_019874 | |
| Software, algorithm | Seahorse Wave software | Agilent | RRID:SCR_014526 | |
| Software, algorithm | Image Lab Software (6.0.1) | Bio-Rad | RRID:SCR_014210 | |
| Software, algorithm | Visiomorph software | B&B microscopes | http://www.bbmicro.com/products/product.php?pid=204 |
Cell lines generation and culture
NT5E gene-editing was generated by electroporation of all-in-one CRISPR/Cas9 vector (px330, Addgene) expressing the 20mer target sequence GCAGCACGTTGGGTTCGGCG (exon1), provided by Michael Hoelzel (University of Bonn, Germany). Cells were sorted based on CD73 expression (CD73 +and CD73-) within 1 week after transfection. Enzymatic functionality was verified using the commercially available malachite green kit assay (R&D system).
Empty vector (pLHCX-ev) and plasmid coding for a FLAG-tagged guinea pig asparaginase (pLHCX-gpASNase1) enzyme were kindly provided by Dr. Lucas B. Sullivan (Fred Hutchinson Cancer Research Center, Seattle, WA, USA; Sullivan et al., 2018). CD73 +and CD73- MDA-MB-231 cells were infected with a retrovirus containing either pLHCX-ev or pLHCX-gpASNase1 and selected with 750 µg/mL of Hygromycin B-containing media until all uninfected control cells died. For NRK1 knockdown in MDA-231 cells, CD73 +and CD73- cells were infected with a retrovirus containing either a control shRNA (shGFP) or shNRK1 (Sigma) and selected with 1 µg/mL of puromycin-containing media until all uninfected control cells died. Plasmids were purchased from Sigma.
MEF were isolated from embryos at 14.5 days of gestation from WT and Nt5e-/- C57BL/6 mice. Briefly, pregnant females were euthanized, uterus were dissected out and individual embryos separated from their yolk sacs into PBS-containing petri dishes. Limbs, tail, head and liver were cut off embryos before being digested and broken up by up-and-down pipetting in freshly thawed 0.25% trypsin solution and incubated at 37 °C in water bath for 5 min cycles until only insoluble cartilage would remain and not settle down. Pooled dissociated cells were filtered through a 40 µM mesh, centrifuged 10 min at 1200 rpm, resuspended in DMEM with 1% pen/strep antibiotics, seeded in 100 mm x 15 mm petri dishes (2 plates per embryo) and incubated in a 37 °C/5% CO2 incubator. Growth media was replaced the next day and rapidly transformed within 7 days upon isolation with a lentiviral vector expressing an oncogenic K-RasG12V (pLenti PGK RasV12), kindly provided by Dr. Francis Rodier (Université de Montréal, Montréal, QC, CA; Rodier et al., 2009). Lentivirus was generated in HEK293FT cells by transfection using standard techniques and cells selected with 75 µg/mL of hygromycin B until all uninfected cells were dead. Immortalized MEF clones upon K-Ras transformation were pooled and used for experimentation.
Cells were purchased from ATCC and frozen aliquots were thawed and used for study within 20 in vitro passages without re-authentication. All cells were grown at 37 °C in a 5% CO2 humid atmosphere. Cells were sub-cultured or media was changed every 3 days. Cell lines were routinely tested and confirmed negative for mycoplasma. MDA-MB-231, MDA-MB-231 ALDHhi, 4T1.2, SM1 LWT1, PANC-1 cell lines were cultured in antibiotics-free DMEM (Wisent) supplemented with 10% FBS (referred to as growth media). SK23MEL and HCC1954 cell lines were cultured in antibiotics-free RPMI 1640 (Wisent) supplemented with 10% FBS. SKOV3 cells were cultured in OSE (Wisent) supplemented with 10% FBS and 1% pen/strep antibiotics. Mouse embryonic fibroblasts (MEF; generated and immortalized as described in method details) were cultured in DMEM (Wisent) supplemented with 10% FBS and 1% pen/strep antibiotics. UWB1.289+BRCA1 cells were culture in 50/50 RPMI (Wisent) and MEGM (Lonza) media supplemented with 3% FBS and 200 µg/mL G-418. All cell lines were negative for mycoplasma (Lonza, MycoAlert). Pyruvate-, glucose- and glutamine-free DMEM media (Wisent) is defined as assay media and supplementations of the media are described in method details depending on the experiment.
Animal experimentation
NOD-Rag1null IL2rgnull (NRG; Jackson Laboratory) and Nt5e-/- C57BL/6 J mice (obtained from Dr. Linda Thompson; OMRF, Oklahoma City, OK, USA) were bred and housed at the CRCHUM which holds a certificate of Good Animal Practice from the Canadian Council on Animal Care (CCAC). C57BL/6 J mice were purchased from Jackson Laboratory and housed at the CRCHUM. All the experimental procedures were authorized, and all animals were handled according to an approved Institutional Animal Care and Use Committee (IACUC) protocol (#C20010JSs) of the Centre de Recherche du Centre Hospitalier de l'Université de Montréal. For in vivo tumor growth, 8–12 weeks-old healthy female NRG mice were injected with 2x106 MDA-MB-231 cells sub-cutaneously and tumor growth was monitored thrice weekly. Tumor volume was measured by caliper in two dimensions, and volumes were estimated using the equation V = (large diameter ×small diameter2)×0.5. The total number of mice analyzed for each experiment is detailed in figure legends. C57BL/6 J and Nt5e-/- C57BL/6 J mice were used for mouse embryoblastic fibroblast (MEF) derivation (see cell lines generation and culture section).
Flow cytometry
Single cell suspensions were strained (40 µM nylon mesh) and incubated with fluorescence-conjugated antibodies (1:200 PE-Cy7 rat anti-mouse CD73 or 1:200 BV421 mouse anti-human CD73) for 30 min at 4 °C in ice-cold PBS + 2% FBS+2 mM EDTA (FACS buffer). Fluorescence was acquired with a LSRFortessa flow cytometer (BD) or sorted with a FACSAria III cell sorting system (BD) equipped with FACSDiva software (BD). Dead cells were excluded using a fixable viability dye eF506 (Thermo Fisher Scientific; cat.: 65-0866-14). FACS data were further analyzed with FlowJo software (version 10.8.0). For viability assay, cells were stained with 7-AAD (1:20) for 15 min at room temperature, washed twice with FACS buffer and immediately acquired on a FACS machine.
Glucose incorporation assay
Cells were cultured in assay media supplemented with 10% FBS and 2 mM glutamine (Sigma) for 30 min before being harvested, washed with PBS 1 X twice and incubated with 100 µM of 2-NBDG (Cayman Chemical) for 30 min at 37 °C in a 5% CO2 humid atmosphere. Cells were washed twice with PBS 1 X and resuspended in FACS buffer (see flow cytometry section). Fluorescence was analyzed in the FITC channel on a LSRFortessa flow cytometer (BD).
Glutamine depletion assay
10 000 MDA-MB-231 CD73 +and CD73- cells were seeded per wells of a 96-well plate and cultured in assay media complemented with 2 mM glutamine (Sigma), 25 mM glucose (Sigma) and 10% FBS for 3 days. Supernatant was collected daily and stored at –20 °C until analysis. Glutamine levels in media was measured using a commercially available kit (Promega) according to manufacturer’s protocol.
ALDH assay
Tumor-initiating cells were sorted based on ALDH activity and tested on seahorse analyzer as previously described (Lee et al., 2017) using the ALDEFLUOR kit (Stemcell). Briefly, cells were harvested and resuspended at 2.5x106 cells/mL of ALDEFLUOR assay buffer. Five µL/mL of the ADLEFLUOR reagent was added to each cell line test tubes and 0.5 mL per test tubes were immediately aliquoted in control tubes in which 10 µL of ALDEFLUOR DEAB reagent were added. Cells were incubated for 30 min at 37 °C washed and resuspended in ALDEFLUOR assay buffer and rapidly sorted on a FACSAria III cell sorting system (BD). 5x104 freshly sorted ALDHhi cells were seeded in seahorse XF24 plate in growth media and incubated overnight before seahorse analysis as described in analysis of extracellular flux section.
Proliferation assays
A total of 2.5x103 cells were seeded in a 48-well plate and cultured in growth media for 8 hr before being washed and switched to assay media supplemented with 10% FBS and 0.1–2 mM glutamine (Glutamax; Gibco) and/or 2.5–25 mM glucose (Sigma). The moment at which the media was changed is defined as baseline (day 0). Percentage of well confluency was measured by applying a phase mask over 4 X photos (2 photos per well) taken every 2 hr for 5 days using the Incucyte technology. For each independent experiments, experimental conditions were tested in triplicate. Proliferation was calculated by reporting the confluence level every 0.5 days relative to confluence level at baseline for each well.
Stable isotope tracing analysis (SITA)
For SITA, 3x106 cells/well were seeded in a six-well plate and cultured in 2 mL assay media supplemented with 10% dialyzed FBS (Wisent) and either 25 mM U-[13C]-glucose (Cambridge Isotope Laboratories) and 4 mM glutamine (Sigma) or 4 mM U-[13C]-glutamine (Cambridge Isotope Laboratories) and 25 mM glucose (Sigma) for 3 hr at 37 °C. Metabolites were extracted from cells using dry ice-cold 80% methanol, followed by sonication and removal of cellular debris by centrifugation at 4 °C. Metabolite extracts were dried, derivatized as tert-butyldimethylsilyl esters, and analyzed via GC-MS by the metabolomic core from the Goodman cancer research centre (McGill University). Labeled metabolite abundance was expressed relative to the internal standard D27 (D-myristic acid) and normalized to protein content. Mass isotopomer distribution was determined using a custom algorithm developed at McGill University (Ma et al., 2017).
LC-MS analyses
For intracellular measurements of AMP, ADP, ATP, NAD + and aspartate, 3x106 cells were seeded in 100 mm petri dishes. Cells were incubated with drugs (7G2 mAb, rhADA or NR) for 48 hr in assay media supplemented with 25 mM glucose, 2 mM Glutamax and 10% dialyzed FBS. Media was washed once with PBS 1 X, remove completely and cell culture dishes were quickly snap frozen on liquid nitrogen. Cells were scraped on ice and collected in 675 µL ice-cold extraction buffer [80% (vol/vol) methanol, 2 mM ammonium acetate, pH 9.0, with 10 µM AMP-13C10,15N5 (IS-AMP) as internal standard], transferred into polypropylene (PP) tubes, and sonicated in a cup-horn sonicator at 150 W for 2 min (cycles of 10 s on, 10 s off) in an ethanol/ice bath. Cell extracts were centrifuged at 4 °C for 10 min at 25,830×g, and supernatants were collected in ice-cold 2 mL PP tubes, to which 250 µL water was added. Polar metabolites were extracted with 1080 µL of chloroform:heptane (3:1 vol/vol) by 2×10 s vortex followed by 10 min incubation on ice and 15 min centrifugation at 4 °C, 12,500×g. From the upper phase, 600 µL was collected without carrying out any interface material and transferred into new cold 2 mL PP tubes. These tubes were centrifuged again, and 400 µL supernatant were collected into cold 1.5 mL PP tubes. Samples were frozen in liquid nitrogen and dried in two steps – first, in a SpeedVac Concentrator for ∼2 hr (maximal vacuum, no heat; Savant) at 4 °C to remove methanol; and second, by lyophilization for 90 min (Labconco FreeZone) – and then stored at –80 °C until used. Samples were reconstituted in 14 µL of Milli-Q water, and injections of 3 µL were performed in duplicate on an electrospray ionization LC-MS/MS system composed of an Agilent 1200 SL device (for LC) and a triple-quadrupole mass spectrometer (4000Q TRAP MS/MS; Sciex). Samples were separated by gradient elution of 12 min on a Poroshell 120 EC-C18, 2.1×75 mm, 2.7 µm column (Agilent Technologies) using mobile phase consisting of an aqueous solvent A (10 mM tributylamine, 15 mM acetic acid, pH 5.2) and an organic solvent B (95% (vol/vol) acetonitrile in water, 0.1% formic acid), at a flow rate of 0.75 mL/min and column oven temperature of 40 °C. Quantification was performed as described in Guay et al., 2013.
For extracellular measurements of NAD+, NMN and NR, 0.2 µg/mL of human recombinant CD73 or 5x104 cells seeded in a 96-wells plate were cultured in assay buffer (2 mM MgCl2, 125 mM NaCl, 1 mM KCl, 10 mM Glucose, 10 mM HEPES pH 7.2, ddH2O) with or without exogenous NMN (0.2 mM) for 1–8 hr. Supernatant was collected in 40% methanol +40% acetonitrile dry ice-cold extraction solution (2:2:1 methanol:acetonitrile:aqueous ratio). Samples were flash frozen on dry ice and stored at –80 °C. For LC-MS/MS analysis, samples (500 µL) were thawed and AMP-13C10,15N5 (IS-AMP) was added as internal standard. Samples were incubated 1 hr at 4 °C with vigorous agitation and centrifuged 10 min, 20K×g, 4 °C. Supernatants were transferred to new polypropylene tubes, dried down at 10 °C by centrifugal vacuum evaporation, then reconstituted in 50 µL 80% acetonitrile in water before LC-MS/MS analysis. Samples (5 µL injections) were separated by HILIC (Nexera X2, Shimadzu) at 0.25 mL/min using a gradient elution (A=MilliQ water, B=90% acetonitrile in water, both A and B containing 10 mM ammonium acetate pH 9.0 and 5 µM Agilent’s deactivator additive; gradient: 0 min 90% B, 2 min 90% B, 12 min 60% B, 15 min 60% B, 16 min 90% B (re-equilibration), 24 min 90% B) on a Poroshell 120 HILIC-Z 2.1×100 mm, 2.7 μm HPLC column (Agilent) following a guard column of similar material, both kept at 30 °C. Metabolites were detected after ESI on a triple quadrupole mass spectrometer (QTRAP 6500, SCIEX) with polarity switching looking for the following transitions: negative ions IS-AMP 360.8/79.0, NAD 661.8/540.0; positive ions NR 255.0/123.1, NMN 334.9/123.2. Depending on the experiment, results normalized on protein content are shown as area of peak obtain by mass spectrometry analysis or absolute concentrations. For quantification of absolute concentration, a mix of pure standards prepared in 80% acetonitrile in water was used to build a calibration curve. No traces of NAD, NMN or NR were found in the assay buffer.
Extracellular flux analysis
Oxygen consumption rate (OCR) and extracellular acidification rate (ECAR) were measured in real-time using Seahorse (XF24 and XFe96) extracellular flux analyzers (Agilent). Cells were seeded in XF24 (5x104) or XFe96 (1x104) and incubated between 12- and 48 hr depending on the experiment. For assays with 7G2 mAb, SCH58261, PSB1115, rhADA, NECA or ADO, drugs were added to the media after seeding and changed after 24 hr with fresh media. The day of the assay, cells were washed and incubated for 60 min in a CO2-free incubator in seahorse assay base media (Agilent) containing 25 mM glucose (Sigma) and 2 mM glutamine (Sigma). For mitochondrial stress test, ATP synthase was inhibited by injection of 1 µM oligomycin (Sigma), followed by 1 µM FCCP (Sigma) to induce mitochondrial uncoupling to determine the spare/maximal respiratory capacity. Non-mitochondrial respiration was determined after rotenone (Sigma; 1 µM)/antimycin A (Sigma; 0.1 µM) injection. For glycolysis measurements, cells were glucose-starved for 60 min in seahorse assay base media containing 2 mM glutamine (Sigma). Glycolysis was measured upon glucose injection (Sigma; 25 mM) and glycolytic reserve upon oligomycin (Sigma; 1 µM) injection. Upon completion of the assay, data were normalized by cell counts (XF24) or crystal violet (XFe96) to correct for seeding density and analysis was performed using Seahorse Wave software (2 min mix, 2 min wait, 2 min measurements).
Genome-wide transcriptional analysis
Total RNA was isolated from MDA-MB-231 cells using the RNeasy Plus Mini Kit (Qiagen) according to the manufacturer’s instructions. RNA was quantified using a DS-11 spectrophotometer (DeNovix). Samples were sent to Génome Québec (Montréal, QC, CA) for gene expression analysis using an Affymetrix microarray chip. Expression values were computed using the robust multi-array analysis (RMA) normalization method (‘affy’ package in Bioconductor; Irizarry et al., 2003; Gautier et al., 2004). When multiple probe sets mapped to the same official gene symbol, we computed their average value. Differential expression (DE) analysis was performed using ‘limma’ (Ritchie et al., 2015) standard analysis pipeline. Significant DE gene were defined as genes with absolute fold-change ≥1.5 and adjusted p-value ≤0.05. Statistical analyses were performed between NT5E mRNA expression and genes expression using Spearman correlation.
Quantitative real-time PCR analysis
Total RNA was isolated and quantified as described in genome-wide transcriptional analysis section. Reverse transcription of 1 µg RNA was performed using the SuperScript VILO cDNA Synthesis Kit (Thermo Fisher Scientific) and ADORA2A, ADORA2B, NMRK1 and Nt5e gene expression encoding for A2A, A2B, NRK1 and CD73 respectively was analyzed using Taqman probes for mouse Nt5e (Thermo Fisher Scientific; Mm00501910_m1), or human NMRK1 (Thermo Fisher Scientific; Hs00944470_m1), human ADORA2A (Thermo Fisher Scientific; Hs00169123_m1) or human ADORA2B (Thermo Fisher Scientific; Hs00386497_m1) relative to mouse Actb (Thermo Fisher Scientific; Mm00607939_s1) or human ACTB (Thermo Fisher Scientific; Hs99999903_m1) using the StepOne PCR machine (Thermo Fisher Scientific) and software (version 2.3).
Western blot
Adherent cells were washed with ice-cold PBS and lysed and scrapped in CelLytic M buffer (Sigma) with 1 X Halt protease and phosphatase cocktail inhibitors (Thermo Fisher Scientific) before being centrifuged for 15 min at 20,000 g at 4 °C. Proteins were harvested from supernatant and quantified by using Bradford protein assay dye reagent (Bio-Rad). Twenty-five µg of protein from whole cell extract were loaded in 4–10% acrylamide gels and transferred on nitrocellulose membranes. Membranes were stained overnight in 5% BSA-containing PBS Tween 0.1% with following antibodies: mouse anti-β-actin (1:5000), mouse anti-PLCγ (1:2000), mouse anti-CD73 (human; 1:1000), mouse anti-FLAG (1:1000) and rabbit anti-Cas9 (S. pyogenes; 1:1000). For PARylation assay, 1x106 cells were seeded in six-well plates and drugs was added in media for 48 hr. Before proceeding to protein extraction, cells were treated with 2 mM H2O2 for 20 min at room temperature. A total of 50 µg proteins were load in a 4–10% acrylamide gels, transferred on nitrocellulose membrane and stained overnight in 5% milk-containing PBS Tween 0.1% with mouse anti-PAR (1:1000). Proteins were revealed with fluorescent secondary anti-rabbit or anti-mouse antibodies (1:10 000; LI-COR) using the LI-COR fluorescent scanner or by chemiluminescence (Bio-Rad) using HRP-conjugated secondary anti-mouse antibody (2.5:10,000) and Amersham ECL prime detection reagent (GE healthcare lifescience).
Immunostaining
For γH2AX immunofluorescence staining, 5x104 cells were seeded on glass slides (Thermo Fisher Scientific) and incubated overnight before adding treatments (Doxorubicin, carboplatin, olaparib). Forty-eight hr later, cells were fixed with formalin 10% (Sigma) for 10 min at room temperature, washed with PBS 1 X and then permeabilized with 0.25% Triton-X100 (Sigma) in PBS 1 X for 15 min. Slides were incubated 1 hr with DAKO blocking solution (Agilent) and incubated overnight at 4 C with anti-phospo-H2AX (1:2000). Cells were then washed three times with PBS 1X-Tween 0.05% followed by incubation with the secondary antibody (1:800; donkey anti-mouse IgG Alexa Fluor 488; Thermo Fisher Scientific). The glass slides were stained with DAPI, washed, and mounted with Fluoromount aqueous mounting medium (Sigma). Stained sections were scanned (Leica) by the molecular pathology core of the CRCHUM and photographs were analyzed using Visomorph viewer software for automatic counting of γH2AX foci.
Data representation and statistical analysis
Mean +/- SEM are shown. Each experimental conditions for all independent experiments were tested in duplicates or triplicates excepted for XFe96 extracellular flux analysis (n=4–6 per condition tested). The numbers of repeated independent experiments are indicated in figure legends. Student T-test were performed when comparing 2 conditions. 1-way ANOVA was used when comparing 3 or more conditions. Statistical significance is shown in comparison to the control condition (untreated or CD73+), unless indicated otherwise by a bracket. Results are considered significant when p<0.05. All statistical analyses were performed using graph pad prism software (version 9.0.2).
Data availability
All data generated or analyzed during this study are included in the manuscript and supporting file.
References
-
The adenosine pathway in Immuno-oncologyNature Reviews. Clinical Oncology 17:611–629.https://doi.org/10.1038/s41571-020-0382-2
-
Oxidative Phosphorylation as an emerging target in cancer therapyClinical Cancer Research 24:2482–2490.https://doi.org/10.1158/1078-0432.CCR-17-3070
-
Next steps for clinical translation of adenosine pathway inhibition in cancer ImmunotherapyJournal for Immunotherapy of Cancer 10:e004089.https://doi.org/10.1136/jitc-2021-004089
-
Synthetic lethality of PARP and NAMPT inhibition in triple-negative breast cancer cellsEMBO Molecular Medicine 4:1087–1096.https://doi.org/10.1002/emmm.201201250
-
High-dimensional analysis of the adenosine pathway in high-grade Serous ovarian cancerJournal for Immunotherapy of Cancer 9:e001965.https://doi.org/10.1136/jitc-2020-001965
-
Targeting extracellular nutrient Dependencies of cancer cellsMolecular Metabolism 33:67–82.https://doi.org/10.1016/j.molmet.2019.11.011
-
Cd73 protein as a source of extracellular precursors for sustained NAD+ biosynthesis in Fk866-treated tumor cellsThe Journal of Biological Chemistry 288:25938–25949.https://doi.org/10.1074/jbc.M113.470435
-
Anti-Cd39 and anti-Cd73 antibodies A1 and 7G2 improve targeted therapy in ovarian cancer by blocking adenosine-dependent immune evasionAmerican Journal of Translational Research 6:129–139.
-
The new insight into extracellular NAD(+) degradation-the contribution of Cd38 and Cd73 in Calcific aortic valve diseaseJournal of Cellular and Molecular Medicine 25:5884–5898.https://doi.org/10.1111/jcmm.15912
-
Cd73 as a therapeutic target for Pancreatic Neuroendocrine tumor stem cellsInternational Journal of Oncology 48:657–669.https://doi.org/10.3892/ijo.2015.3299
-
NAD(+) homeostasis in health and diseaseNature Metabolism 2:9–31.https://doi.org/10.1038/s42255-019-0161-5
-
Impaired L-arginine metabolism marks endothelial dysfunction in Cd73-deficient miceMolecular and Cellular Biochemistry 458:133–142.https://doi.org/10.1007/s11010-019-03537-4
-
Laying a trap to kill cancer cells: PARP inhibitors and their mechanisms of actionScience Translational Medicine 8:362.https://doi.org/10.1126/scitranslmed.aaf9246
-
Fueling genome maintenance: on the versatile roles of NAD(+) in preserving DNA integrityThe Journal of Biological Chemistry 298:102037.https://doi.org/10.1016/j.jbc.2022.102037
-
Cancer stem cell metabolism and potential therapeutic targetsFrontiers in Oncology 8:203.https://doi.org/10.3389/fonc.2018.00203
-
Aspartate is an endogenous metabolic limitation for tumour growthNature Cell Biology 20:782–788.https://doi.org/10.1038/s41556-018-0125-0
-
Evidence for the involvement of Ecto-5'-Nucleotidase (Cd73) in drug resistanceInternational Journal of Cancer 68:493–500.https://doi.org/10.1002/(SICI)1097-0215(19961115)68:4<493::AID-IJC15>3.0.CO;2-6
-
Functions of Parylation in DNA damage repair pathwaysGenomics, Proteomics & Bioinformatics 14:131–139.https://doi.org/10.1016/j.gpb.2016.05.001
-
RNA interference of Ecto-5'-Nucleotidase (Cd73) inhibits human breast cancer cell growth and invasionClinical & Experimental Metastasis 24:439–448.https://doi.org/10.1007/s10585-007-9081-y
Article and author information
Author details
Funding
Canadian Institutes of Health Research
- John Stagg
Fonds de recherche du Québec
- John Stagg
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Acknowledgements
The authors thank Julien Lamontagne, Alexia Grangeon, Maxime Cahuzac, Dominique Gauchat, Daina Avizonis and Bozena Samborska for technical assistance.
Ethics
All the experimental procedures were authorized, and all animals were handled according to an approved Institutional Animal Care and Use Committee (IACUC) protocol (#C20010JSs) of the Centre de Recherche du Centre Hospitalier de l'Université de Montréal.
Copyright
© 2023, Allard et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 2,258
- views
-
- 314
- downloads
-
- 18
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Citations by DOI
-
- 18
- citations for umbrella DOI https://doi.org/10.7554/eLife.84508






