Effective control of SARS-CoV-2 transmission between healthcare workers during a period of diminished community prevalence of COVID-19
Abstract
Previously, we showed that 3% (31/1032)of asymptomatic healthcare workers (HCWs) from a large teaching hospital in Cambridge, UK, tested positive for SARS-CoV-2 in April 2020. About 15% (26/169) HCWs with symptoms of coronavirus disease 2019 (COVID-19) also tested positive for SARS-CoV-2 (Rivett et al., 2020). Here, we show that the proportion of both asymptomatic and symptomatic HCWs testing positive for SARS-CoV-2 rapidly declined to near-zero between 25th April and 24th May 2020, corresponding to a decline in patient admissions with COVID-19 during the ongoing UK ‘lockdown’. These data demonstrate how infection prevention and control measures including staff testing may help prevent hospitals from becoming independent ‘hubs’ of SARS-CoV-2 transmission, and illustrate how, with appropriate precautions, organizations in other sectors may be able to resume on-site work safely.
Introduction
The role of nosocomial transmission of SARS-CoV-2 has been highlighted by recent evidence suggesting that 20% of SARS-CoV-2 infections among patients in UK hospitals and up to 89% of infections among HCWs may have originated in hospitals (Evans et al., 2020; Iacobucci, 2020). Since the introduction of ‘lockdown’ in the UK, community transmission rates of SARS-CoV-2 have generally declined (Public Health England (PHE), 2020). Conversely, concerns have been raised that hospitals could become independent ‘hubs’ for ongoing SARS-CoV-2 transmission between patients and HCWs, which would effectively prolong the epidemic (Iacobucci, 2020). In this context, the evolution of the epidemic curves of a hospital’s symptomatic and asymptomatic workforce has not been well described.
We recently initiated a comprehensive HCW screening programme for SARS-CoV-2 in a large teaching hospital in Cambridge, UK. Over a 3-week period from 6th to 24th April 2020, 3% (31/1032) HCWs in the asymptomatic screening arm, 15.4% (26/169) HCWs in the symptomatic screening arm, and 7.7% (4/52) contacts in the symptomatic household contact screening arm tested positive for SARS-CoV-2 (Rivett et al., 2020). Our data from the asymptomatic screening arm were consistent with the results of Shields et al. (Shields et al., 2020). Over the next 4 weeks from 25th April to 24th May 2020, we performed a further 3388 additional tests. Here, we present these longitudinal data, in the context of the hospital patient population and wider local community.
Results
Testing for SARS-CoV-2 RNA was performed with real-time RT-PCR using throat and nose swab samples of HCWs from Cambridge University Hospitals NHS Foundation Trust (CUHNFT) and their symptomatic household contacts. Over the new study period (25th April to 24th May 2020), 2611 additional tests were performed in the HCW asymptomatic screening arm, 555 additional tests in the HCW symptomatic screening arm, and 216 additional tests in the HCW household contact screening arm. A further six tests did not have a clearly recorded arm of origin. Over the entire study period, the median age of HCWs and their household contacts was 36.5 and 35.5 years, respectively. About 68.4% were female and 31.6% were male. Of the individuals testing positive over the entire study period, the median age of HCWs and their household contacts was 32 and 47 years, respectively. About 77.9% of all positive tests were from females and 22.1% from males. Table 1 summarizes the total number of HCWs testing positive through either arm of the screening programme, according to the job role. A comparison of the proportions of hospital employees from each job role that tested positive through the HCW symptomatic screening arm revealed no statistically significant difference (Pearson’s chi-square test p=0.419). Reasonable comparison of the proportions testing positive through the HCW asymptomatic screening arm was not possible due to non-random sampling of different areas of the hospital, meaning some job roles had been more frequently targeted for asymptomatic screening than others.
Combined data for SARS-CoV-2 RNA positive HCWs by role and screening arm, from the present study and our previous study (Rivett et al., 2020).
Difference in proportions of HCWs testing positive through the symptomatic screening arm was analysed using Pearson’s chi-square test.
| Role | HCW asymptomatic screening arm | HCW symptomatic screening arm | Total number of hospital employees |
|---|---|---|---|
| Nurse | 25 | 19 | 3621 |
| Healthcare assistant | 14 | 8 | 1734 |
| Doctor | 8 | 6 | 1871 |
| Cleaners | 2 | 3 | 560 |
| Radiographer | 2 | 1 | 217 |
| Radiology support worker | 0 | 1 | 35 |
| Physiotherapist | 1 | 0 | 116 |
-
Overall, 360 individuals underwent repeat testing, either as part of the asymptomatic screening programme, or for other reasons as previously described (Rivett et al., 2020). The median turnaround time from sample arrival in the laboratory to final verification was 18 hr 45 min. Positive results were called out on the same day, with negative results emailed within 24 hr.
Between 25th April and 24th May 2020, a total of 34 new positive tests were reported. In the HCW symptomatic and HCW symptomatic household contact screening arms combined (reflecting all individuals with self-reported symptoms at the time of testing), 13/771 (1.7%) tests were positive, which was significantly lower than 30/221 (13%) in the original study period (Fisher’s exact test p<0.0001). In the HCW asymptomatic screening arm, 21/2611 (0.8%) tests were positive, which again was significantly lower than 31/1032 (3%) in the original study period (Fisher’s exact test p<0.0001). As we previously observed (Rivett et al., 2020), individuals captured in the HCW asymptomatic screening arm were generally asymptomatic at the time of screening; however, these individuals could be divided into subgroups. In the first subgroup, 8/21 (38%) HCWs had no symptoms at all. Of these, 5/8 (63%) remained entirely asymptomatic 5–7 weeks after their positive test, whereas 2/8 (25%) developed symptoms 24–48 hr after testing. One HCW could not be contacted to obtain further history. In the second subgroup, 6/21 (29%) had retrospectively experienced some symptoms prior to screening. Of these, 5/6 (83%) had symptoms with a high pre-test probability of COVID-19 (Rivett et al., 2020) commencing >7 days prior to screening, of whom 3/5 had appropriately self-isolated then returned to work, and 1/5 was tested shortly after developing symptoms. 1/6 (17%) had symptoms with a low pre-test probability of COVID-19 (Rivett et al., 2020) commencing <7 days prior to screening and had not self-isolated. In the third subgroup, 7/21 (33%) were detected through repeat sampling of HCW who previously tested positive. Of these, 4/7 (57%) were tested to determine their suitability to return to work with severely immunocompromised/immunosuppressed patients, as dictated by UK national guidance (National Institute for Health and Care Excellence (NICE), 2020). The remaining 3/7 (43%) were from HCWs tested incidentally for the second time in the asymptomatic HCW screening programme. The median interval between serial positive tests was 16.5 days (IQR 9.5–19.5). All cases were attributable to prolonged SARS-CoV-2 RNA detection from a single infection, rather than re-infection. Our approach to patients with repeatedly positive SARS-CoV-2 PCR tests is described in the Methods.
The fraction of positive tests among the HCW asymptomatic, and HCW symptomatic and household contact screening groups combined varied over time (Figure 1A, Table 2). In particular, during the last 2 weeks of the study period (11th to 24th May 2020), we identified only four positive SARS-CoV-2 samples from 2016 tests performed, two from the HCW asymptomatic and two from the HCW symptomatic/symptomatic household contact arms. This fall in positive HCW tests mirrored the decline in both patients testing positive at CUHNFT and those tested throughout the wider region (Figure 1B). Similar trends were observed in a smaller cohort study of HCWs in London (Treibel et al., 2020).
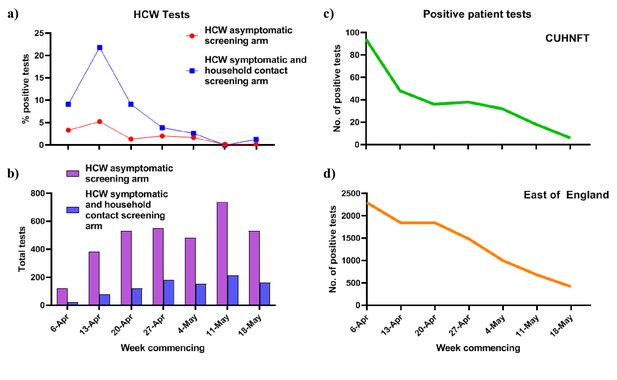
Trends in positive SARS-CoV-2 PCR tests among HCWs, hospital patients and the wider community over time.
(a) Positive SARS-CoV-2 tests for asymptomatic and symptomatic screening arms by week. (b) Total HCW SARS-CoV-2 tests in CUHNFT performed by week. (c) Total positive SARS-CoV-2 patient tests in Cambridge University Hospital NHS Foundation Trust (CUHNFT) by week. (d) Total positive SARS-CoV-2 tests in the East of England (EOE) by week.
-
Figure 1—source data 1
Source data for trends in SARS-CoV-2 PCR positive HCWs, hospital patients and individuals in the wider community.
- https://cdn.elifesciences.org/articles/59391/elife-59391-fig1-data1-v3.xlsx
Positive tests and total number of SARS-CoV-2 tests performed in each screening arm categorised according to week since starting the healthcare worker testing programme (6th April–24th May 2020).
| Week commencing | ||||||||
|---|---|---|---|---|---|---|---|---|
| 6th April | 13th April | 20th April | 27th April | 4th May | 11th May | 18th May | Total | |
| HCW asymptomatic screening arm | 4/121 | 20/383 | 7/529 | 11/550 | 8/483 | 1/738 | 1/840 | 52/3644 (1.4%) |
| HCW symptomatic screening arm | 1/15 | 14/60 | 11/95 | 7/119 | 3/104 | 0/164 | 2/168 | 38/725 (5.2%) |
| HCW symptomatic household contacts | 1/7 | 3/18 | 0/26 | 0/62 | 1/50 | 0/51 | 0/53 | 5/267 (1.8%) |
| Unknown | 0/0 | 0/2 | 0/13 | 0/0 | 0/4 | 0/1 | 0/1 | 0/21 |
| All | 6/143 (4.1%) | 37/463 (7.9%) | 18/663 (2.7%) | 18/731 (2.4%) | 12/641 (1.8%) | 1/954 (0.1%) | 3/1062 (0.2%) | 95/4657 (2%) |
In our original study between 6th and 24th April 2020, we described in detail two clusters of HCW infections (Rivett et al., 2020). From 25th April to 24th May 2020, we detected one additional cluster on a general medical ward with a separate area for patients with proven COVID-19 and another area for those without. This was identified through targeted screening of the ward over a 24 hr period from 4th to 5th May 2020, in response to four staff testing positive through the HCW symptomatic arm of the screening programme from 27th to 30th April 2020. Reactive screening of a further 40 staff from the same ward identified a further three positive asymptomatic HCWs. In addition, a further two HCWs tested positive in an asymptomatic screen of 30 individuals from a closely related clinical area (designated for non-COVID patients) on 6th May 2020.
Discussion
Our data demonstrate a dramatic fall in the prevalence of symptomatic and asymptomatic SARS-CoV-2 infection among HCWs in our hospital during the study period. On average, the number of secondary infections among HCWs arising from each infected HCW (effectively, the reproduction number (R) for SARS-CoV-2 transmission between HCWs) must therefore be <1.
As well as an acquisition from other HCWs, infections among HCWs may also be acquired from patients, as well as other individuals outside the hospital. Our study period coincided with a decline in the rate of infection across our local community, and our data are consistent with a reduction in transmission within the hospital, a reduction in community-based acquisition of infection by HCWs, or (most likely) a combination of both. In the absence of detailed epidemiological data, it is not possible to formally differentiate between these possibilities or determine their relative effect sizes. Nonetheless, our identification of HCW infection clusters in specific areas of the hospital highlighted the potential for workplace acquisition of SARS-CoV-2, which may lead to self-sustaining outbreaks if left uninterrupted (Rivett et al., 2020; Meredeth et al., 2020). For each of these clusters, timely identification of HCW infection proved effective in terminating chains of hospital transmission between staff, preventing ongoing nosocomial infection.
With the incidence of infection having fallen significantly in hospitalised patients, HCWs and the wider community, many hospitals across the UK and further afield have been afforded precious time to build the infrastructure necessary to establish comprehensive screening programmes in anticipation of a possible second epidemic peak. For hospitals already operating newly established screening programmes, the challenge now is to up-scale to the point that screening can occur at a frequency that permits pre-symptomatic capture of as close to 100% of all new infections as possible. This approach will enable staff to be removed from the workplace at the time of peak infectivity (He et al., 2020). The minimum screening frequency required needs to be carefully modelled, with recent estimates suggesting the need for weekly testing to prevent 16–33% of onward transmission from HCWs, depending on the time taken for results to be reported, and another study estimating the need for daily screening to prevent 65% of HCW-to-HCW transmission events (Evans et al., 2020; Grassly et al., 2020). In practice, we have observed good results in our hospital with a current frequency of asymptomatic screening every 2–4 weeks. Those being screened are prioritised by anticipated ward-based exposure to COVID-19, with additional targeted screens triggered by excess staff sickness or the identification of symptomatic cases on specific wards (Rivett et al., 2020). In addition to asymptomatic screening, testing of symptomatic HCWs is essential for preventing excessive erosion of the hospital workforce by self-isolation on the basis of symptoms alone, and testing of symptomatic HCW household contacts negates the need for unnecessary self-quarantine periods for co-habiting HCWs. We found uptake to the HCW symptomatic household contact screening arm of our programme to be notably lower than the HCW symptomatic arm despite regular communications to advertise the service within CUHNFT. This lack of uptake may reflect a lack of awareness that symptomatic non-HCWs were eligible for testing, provided they shared a household with a hospital employee. Many non-hospital employees may also have been more inclined to attend national testing centres or be less aware of the spectrum of COVID-19 symptoms.
Importantly, our data demonstrate that CUHNFT was not acting as an independent ‘hub’ for ongoing COVID-19 transmission among HCWs. The absence of nosocomial transmission likely reflects the combined efficacy of HCW testing, stringent prospective, and reactive infection prevention and control measures, and appropriate social distancing among the workforce. These findings should give reassurance to both hospital staff and patients that healthcare facilities remain safe places to give and receive care. Furthermore, since CUHNFT, with approximately 11,000 staff members (many of whom are based in the hospital) is a major regional employer, we predict that comparable organisations in other sectors may also be able to resume on-site work safely by instigating similar precautions.
Materials and methods
Staff screening protocols
View detailed protocolWe previously described protocols for staff screening, sample collection, laboratory processing, and results reporting in detail (Rivett et al., 2020). These methods remained unchanged throughout this study period. Two parallel streams of entry into the testing programme included (i) HCW symptomatic, and HCW symptomatic household contact screening arms and (ii) an HCW asymptomatic screening arm. In the former, any patient-facing or non-patient-facing HCW could voluntarily refer themselves or a household contact, should they develop symptoms suggestive of COVID-19. In the latter, HCWs could volunteer to take part in a rolling programme of testing for all patient-facing and non-patient-facing staff working in defined clinical areas thought to be at risk of SARS-CoV-2 transmission. Testing was performed (i) at temporary on-site ‘Pods’; (ii) via self-swabbing kits delivered to HCWs in their area of work. All individuals in each arm of the programme performed a self-swab at the back of the throat then the nasal cavity, followed by RNA extraction and amplification using real-time RT-PCR (Sridhar et al., 2020). Cluster investigation was initiated when three or more HCWs working in the same clinical area tested positive for SARS-CoV-2 in 1 week .
Management of HCW with repeat positive tests
Request a detailed protocolCurrent National Institute for Health and Care Excellence (NICE) guidelines require a negative test before returning to work with immunocompromised patients (National Institute for Health and Care Excellence (NICE), 2020). In accordance with the UK national guidance, individuals with repeat positive screens following a minimum period of 7 days self-isolation were advised to continue working if they were not scheduled to come into close contact with heavily immunocompromised patients, provided they remained asymptomatic (UK Government, 2020). This approach to managing repeat positive screens is further supported by recent data from the Korea Centers for Disease Control and Prevention, which showed no clear evidence of onward transmission to the contacts of 285 repeat-positive individuals, 108 of whom had samples taken for attempted viral culture, which was universally unsuccessful (Korea Centers for Disease Control & Prevention (KCDC), 2020). Additional small studies have also demonstrated an inability to culture virus from clinical samples obtained later than 8 days after symptom onset, suggesting prolonged detection of viral RNA is unlikely to indicate an ongoing risk of transmission (Wölfel et al., 2020; Bullard et al., 2020).
Data extraction and analysis
Request a detailed protocolSwab result data for HCWs and patients were extracted directly from the hospital-laboratory interface software, Epic (Verona, WI) and from SARS-CoV-2 point of care testing. Data for SARS-CoV-2 infections from the local community were extracted from Public Health England’s Data Dashboard (Public Health England (PHE), 2020). Data were collated using Microsoft Excel, and figures produced with GraphPad Prism (GraphPad Software, La Jolla, CA). Fisher’s exact test was used to compare the proportion of HCWs testing positive in this study period to that of our previous study period (Rivett et al., 2020). Pearson’s chi-square test was used for comparison of the proportions of HCWs testing positive in each job role.
Data availability
All data generated or analysed during this study are included in the manuscript and supporting files.
References
-
Predicting infectious SARS-CoV-2 from diagnostic samplesClinical Infectious Diseases 22:ciaa638.https://doi.org/10.1093/cid/ciaa638
Article and author information
Author details
Funding
Wellcome (108070/Z/15/Z)
- Michael P Weekes
Wellcome (215515/Z/19/Z)
- Stephen Baker
Wellcome (207498?Z/17/Z)
- Ian G Goodfellow
Wellcome (206298/B/17/Z)
- Ian G Goodfellow
Wellcome (210688/Z/18/Z)
- Paul J Lehner
Wellcome (200871/Z/16/Z)
- Kenneth GC Smith
Medical Research Council (MR/P008801/1)
- Nicholas J Matheson
Addenbrooke's Charitable Trust, Cambridge University Hospitals
- Ian G Goodfellow
- Paul J Lehner
- Stephen Baker
- Michael P Weekes
NHS Blood and Transplant (WPA15-02)
- Nicholas J Matheson
National Institute for Health Research
- Afzal Chaudhry
- John R Bradley
- Gordon Dougan
Cancer Research UK (C38317/A24043)
- Jamie Young
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Acknowledgements
This work was supported by the Wellcome Trust Senior Research Fellowships 108070/Z/15/Z to MPW, 215515/Z/19/Z to SGB and 207498/Z/17/Z to IGG; Collaborative award 206298/B/17/Z to IGG; Principal Research Fellowship 210688/Z/18/Z to PJL; Investigator Award 200871/Z/16/Z to KGCS; Addenbrooke’s Charitable Trust (to MPW, SGB, and PJL); the Medical Research Council (CSF MR/P008801/1 to NJM); NHS Blood and Transfusion (WPA15-02 to NJM); National Institute for Health Research (Cambridge Biomedical Research Centre at CUHNFT), to JRB, AC and GD, Cancer Research UK (PRECISION Grand Challenge C38317/A24043 award to JY).
The CITIID-NIHR COVID-19 BioResource Collaboration
Principal Investigators: Stephen Baker, John Bradley, Gordon Dougan, Ian Goodfellow, Ravi Gupta, Paul J Lehner, Paul A Lyons, Nicholas J Matheson, Kenneth GC Smith, M Estee Torok, Mark Toshner, Michael P Weekes
Infectious Diseases Department: Nicholas K Jones, Lucy Rivett, Matthew Routledge, Dominic Sparkes, Ben Warne
SARS-CoV-2 testing team: Claire Cormie, Sally Forrest, Harmeet Gill, Iain Kean, Joana Pereira-Dias, Nicola Reynolds, Sushmita Sridhar, Michelle Wantoch, Jamie Young
COG-UK Cambridge Sequencing Team: Sarah Caddy, Laura Caller, Theresa Feltwell, Grant Hall, William Hamilton, Myra Hosmillo, Charlotte Houldcroft, Aminu Jahun, Fahad Khokhar, Luke Meredith, Anna Yakovleva
NIHR BioResource: Helen Butcher, Daniela Caputo, Debra Clapham-Riley, Helen Dolling, Anita Furlong, Barbara Graves, Emma Le Gresley, Nathalie Kingston, Sofia Papadia, Hannah Stark, Kathleen E Stirrups, Jennifer Webster
Research nurses: Joanna Calder, Julie Harris, Sarah Hewitt, Jane Kennet, Anne Meadows, Rebecca Rastall, Criona O,Brien, Jo Price, Cherry Publico, Jane Rowlands, Valentina Ruffolo, Hugo Tordesillas
CRUK: Michael Gill, Jane Gray, Greg Hannon
NIHR Cambridge Clinical Research Facility: Karen Brookes, Laura Canna, Isabel Cruz, Katie Dempsey, Anne Elmer, Naidine Escoffery, Stewart Fuller, Heather Jones, Carla Ribeiro, Caroline Saunders, Angela Wright
Cambridge Cancer Trial Centre: Rutendo Nyagumbo, Anne Roberts
Clinical Research Network Eastern: Ashlea Bucke, Simone Hargreaves, Danielle Johnson, Aileen Narcorda, Debbie Read, Christian Sparke, Lucy Worboys
Administrative staff, CUHNFT: Kirsty Lagadu, Lenette Mactavous
CUHNFT NHS Foundation Trust: Tim Gould, Tim Raine, Ashley Shaw
Cambridge Cancer Trials Centre: Claire Mather, Nicola Ramenatte, Anne-Laure Vallier
Legal/Ethics: Mary Kasanicki
CUHNFT Improvement and Transformation Team: Penelope-Jane Eames, Chris McNicholas, Lisa Thake
Clinical Microbiology & Public Health Laboratory (PHE): Neil Bartholomew, Nick Brown, Martin Curran, Surendra Parmar, Hongyi Zhang
Occupational Health: Ailsa Bowring, Mark Ferris, Geraldine Martell, Natalie Quinnell, Giles Wright, Jo Wright
Health and Safety: Helen Murphy
Department of Medicine Sample Logistics: Benjamin J Dunmore, Ekaterina Legchenko, Stefan Gräf, Christopher Huang, Josh Hodgson, Kelvin Hunter, Jennifer Martin, Federica Mescia, Ciara O’Donnell, Linda Pointon, Joy Shih, Rachel Sutcliffe, Tobias Tilly, Zhen Tong, Carmen Treacy, Jennifer Wood
Department of Medicine Sample Processing and Acquisition: Laura Bergamaschi, Ariana Betancourt, Georgie Bowyer, Aloka De Sa, Maddie Epping, Andrew Hinch, Oisin Huhn, Isobel Jarvis, Daniel Lewis, Joe Marsden, Simon McCallum, Francescsa Nice, Ommar Omarjee, Marianne Perera, Nika Romashova, Mateusz Strezlecki, Natalia Savoinykh Yarkoni, Lori Turner
Epic team/other computing support: Barrie Bailey, Afzal Chaudhry, Rachel Doughton, Chris Workman
Statistics/modelling: Caroline Trotter
Department of Engineering: William David Cordova Jiménez, Jack Levy, Fatima NB Samad
Ethics
Human subjects: As a study of healthcare-associated infections, this investigation is exempt from requiring ethical approval under Section 251 of the NHS Act 2006 (see also the NHS Health Research Authority algorithm, available at http://www.hra-decisiontools.org.uk/research/, which concludes that no formal ethical approval is required). Our study was performed as a service evaluation of the Cambridge Universith Hospitals NHS Foundation Trust screening programme. The service provided was not changed in any way in order to undertake this evaluation.
Copyright
© 2020, Jones et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 3,824
- views
-
- 524
- downloads
-
- 37
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Citations by DOI
-
- 37
- citations for umbrella DOI https://doi.org/10.7554/eLife.59391
Download links
Downloads (link to download the article as PDF)
Open citations (links to open the citations from this article in various online reference manager services)
Cite this article (links to download the citations from this article in formats compatible with various reference manager tools)
Further reading
-
- Epidemiology and Global Health
- Microbiology and Infectious Disease
The BNT162b2 mRNA COVID-19 vaccine (Pfizer-BioNTech) is being utilised internationally for mass COVID-19 vaccination. Evidence of single-dose protection against symptomatic disease has encouraged some countries to opt for delayed booster doses of BNT162b2, but the effect of this strategy on rates of asymptomatic SARS-CoV-2 infection remains unknown. We previously demonstrated frequent pauci- and asymptomatic SARS-CoV-2 infection amongst healthcare workers (HCWs) during the UK’s first wave of the COVID-19 pandemic, using a comprehensive PCR-based HCW screening programme (Rivett et al., 2020; Jones et al., 2020). Here, we evaluate the effect of first-dose BNT162b2 vaccination on test positivity rates and find a fourfold reduction in asymptomatic infection amongst HCWs ≥12 days post-vaccination. These data provide real-world evidence of short-term protection against asymptomatic SARS-CoV-2 infection following a single dose of BNT162b2 vaccine, suggesting that mass first-dose vaccination will reduce SARS-CoV-2 transmission, as well as the burden of COVID-19 disease.
-
- Epidemiology and Global Health
- Medicine
Significant differences exist in the availability of healthcare worker (HCW) SARS-CoV-2 testing between countries, and existing programmes focus on screening symptomatic rather than asymptomatic staff. Over a 3 week period (April 2020), 1032 asymptomatic HCWs were screened for SARS-CoV-2 in a large UK teaching hospital. Symptomatic staff and symptomatic household contacts were additionally tested. Real-time RT-PCR was used to detect viral RNA from a throat+nose self-swab. 3% of HCWs in the asymptomatic screening group tested positive for SARS-CoV-2. 17/30 (57%) were truly asymptomatic/pauci-symptomatic. 12/30 (40%) had experienced symptoms compatible with coronavirus disease 2019 (COVID-19)>7 days prior to testing, most self-isolating, returning well. Clusters of HCW infection were discovered on two independent wards. Viral genome sequencing showed that the majority of HCWs had the dominant lineage B∙1. Our data demonstrates the utility of comprehensive screening of HCWs with minimal or no symptoms. This approach will be critical for protecting patients and hospital staff.






