Evolution: Shining a light on the origin of fly species
New species arise when populations of the same species become so different that they no longer or rarely interbreed. Physical barriers, such as an ocean, may facilitate this process, as is the case for the different varieties of Darwin’s finches. But how do new species emerge if they coexist in the same habitat? One explanation could be a process called niche partitioning, whereby competing species use the surrounding environment in different ways, for example by feeding on different resources.
Last year, a study of 62 species of fly belonging to the Drosophila family, led by researchers at the Max Planck Institute of Chemical Ecology, found that the size of a fly’s antenna (the main olfactory organ) is inversely correlated to the size of its eye. i.e. species with larger eyes had smaller antennae and vice versa (Keesey et al., 2019). Both organs develop from the same structure suggesting that this inverse correlation arises through a developmental constraint. Now, in eLife, Ian Keesey, Veit Grabe, Markus Knaden and Bill Hansson – who were involved in the 2019 study – report that light variation within a forest habitat could have contributed to niche partitioning and the speciation of flies belonging to this family (Keesey et al., 2020). The team focused their study on two fly species: Drosophila subobscura and Drosophila pseudoobscura. These species are closely related and known to have large differences in the relative size of their eyes and antennae, but do not usually share the same habitat and are commonly found in Europe and North America respectively (Figure 1).
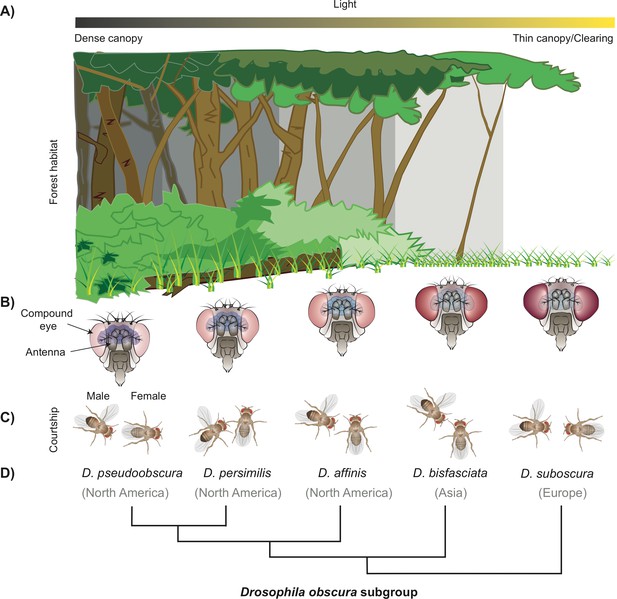
How a subgroup of flies could have become separated by niche partitioning.
(A) The difference in density of the tree canopy covering a forest creates micro-habitats with varying levels of light, which can be a factor for niche partitioning leading to the birth of new fly species from the Drosophila family (B) Diagram showing the five fly species studied belonging to the obscura subgroup, which have an inverse relationship between the size of their eyes and antennae. D. pseudoobscura (left) has the smallest eyes and biggest antennae, and D. subobscura (right) has the biggest eyes and smallest antennae (not drawn to scale). (C) Diagrams illustrating the different mating rituals for each of the five species. D. pseudoobscura flies have the smallest eyes, are the least attracted to light, and have the least vision-dependent courtship (the male courts from the back of the female). D. subobscura, on the other hand, have the biggest eyes, are the most attracted to light, and have the most visually dependent courtship (fully frontal). The other species in the subgroup display a gradient of the morphology, light attraction, and mating behaviour. (D) Phylogenetic tree of these species and the main geographical locations where they can currently be found.
Image credit: Joe Brock.
First, Keesey et al. measured the eye size and other morphological parameters of these two species, including the number of ommatidia – repetitive units that make up the eyes of insects. Ommatidia are a bit like the pixels of a camera, in that the more flies have, the better the spatial resolution of their eyes (Gonzalez-Bellido et al., 2011; Ramaekers et al., 2019). They found that the larger eyes of D. subobscura reflect an increase in the number of ommatidia, rather than an increase in the size of each ommatidium, which suggests this species might have enhanced visual acuity (Figure 1B).
Keesey et al. propose that the ‘flirting’ strategy of males (i.e. their courtship rituals) may have evolved in response to these two species investing differently in the size of their eyes and antennae. D. suboscura males seem to rely on visual displays to attract females, for example by ‘showing-off’ their wings, whereas D. pseudoobscura males only approach females from the back, while singing by vibrating their wings (Figure 1C). This is consistent with previous work which showed that while D. pseudoobscura can mate successfully in the dark, D. suboscura requires light (Wallace and Dobzhansky, 1946). It is possible that the increased visual acuity of D. suboscura facilitated the evolution of visual courtship rituals, causing them to become sexually isolated and diverge from other species. But what other ecological factors could have driven the increased investment in the visual system?
The canopy of trees that covers the natural habitats of these two species varies greatly in density, creating distinct micro-habitats that are either dark and cool, or warm and light (Figure 1A). Further experiments showed that D. suboscura prefer well-lit conditions, while D. pseudoobscura are more likely to prefer darkness. A population of D. suboscura has recently colonised North America, and now share a forest habitat with D. pseudoobscura in some regions (Noor, 1998). It is possible that niche partitioning reduces competition between these two species, if they separate into different canopy regions.
Taken together, these findings show that visual vs. olfactory investment, dependence on vision for mating rituals, and preference for light, all vary in a correlated fashion between these two species. Yet, the order in which these features emerged is difficult to determine. One possibility is that slight differences in light preference would initially segregate flies into two micro-habitats. Flies living in better lit environments would become increasingly more visual, while flies living in the shadows might have evolved a finer sense of smell at the expense of their eyes.
Another possibility is that genetic variation within members of the same species could generate individual flies with larger eyes or antennae: these differences could lead to niche partitioning, as flies with larger eyes would be at an advantage in well-lit forest clearings, and vice versa. This would be followed by the evolution of different light preferences and mating rituals. This hypothesis is partially supported by a previous study showing that small mutations in the regulatory region of a gene called eyeless can change the relative size of these sensory organs within and across species (Ramaekers et al., 2019). Such simple genetic bases potentially makes the size of the eye and the antennae so easily evolvable across species.
To address the evolutionary order of these traits, Keesey et al. expanded their work to include three additional species. The results showed that D. suboobscura and D. pseudoobscura are at the two extremes in a graded variation of these three traits. One of the species examined, called D. persimilis, is the closest relative of D. pseudoobscura and shares the same habitat (Figure 1D). D. persimilis displayed the largest difference to its sibling species in terms of their preference for light, with smaller increases in their eye investment and visual courtship behaviour (Figure 1D). This suggests stronger evolutionary pressures for niche partitioning on light preference behaviour, with visual investment and courtship rituals further increasing this separation.
The idea that the emergence of new fly species might be due to changes in the preference for light is intriguing and inspires many more questions. For example, does this niche partitioning really occur in nature? And if so, what were the initial selection pressures favouring the differential preference for light? Could other factors correlated with canopy thickness – such as reduced risk of desiccation and irradiation – also have contributed towards this variation?
It is also unclear what neurobiological mechanism led to this initial switch in light preference. Although there is no evidence that larger eyes would make animals more attracted to light, these two traits could be linked. For example, changes in the regulatory region of eyeless could simultaneously affect the number of ommatidia and neuronal circuits in the eye. Given these species can be genetically manipulated (Tanaka et al., 2017), future experiments swapping their regulatory region of eyeless could provide some answers.
This study illustrates how studying little known fly species and their ecology can shed light on how brains evolve, and how behavioural changes can shape the evolution of new species.
References
-
Diurnal activity patterns of Drosophila subobscura and D. pseudoobscura in sympatric populationsThe American Midland Naturalist 140:34–41.https://doi.org/10.1674/0003-0031(1998)140[0034:DAPODS]2.0.CO;2
-
Optogenetic activation of the fruitless-labeled circuitry in Drosophila subobscura males induces mating motor actsThe Journal of Neuroscience 37:11662–11674.https://doi.org/10.1523/JNEUROSCI.1943-17.2017
Article and author information
Author details
Publication history
Copyright
© 2020, Gong and Prieto-Godino
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 1,998
- views
-
- 159
- downloads
-
- 0
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.






