An effector of the Irish potato famine pathogen antagonizes a host autophagy cargo receptor
Abstract
Plants use autophagy to safeguard against infectious diseases. However, how plant pathogens interfere with autophagy-related processes is unknown. Here, we show that PexRD54, an effector from the Irish potato famine pathogen Phytophthora infestans, binds host autophagy protein ATG8CL to stimulate autophagosome formation. PexRD54 depletes the autophagy cargo receptor Joka2 out of ATG8CL complexes and interferes with Joka2's positive effect on pathogen defense. Thus, a plant pathogen effector has evolved to antagonize a host autophagy cargo receptor to counteract host defenses.
https://doi.org/10.7554/eLife.10856.001eLife digest
Plants and other living organisms can survive stress and starvation by digesting and recycling parts of their own cells. This process is known as autophagy and it involves engulfing cellular material inside spherical structures called autophagosomes, before delivering it to sites in the cell where digestive enzymes can break the material down. A form of autophagy, known as selective autophagy, can specifically degrade toxic substances such as disease-causing microbes. Selective autophagy works through proteins called autophagy cargo receptors that define which molecules are targeted for degradation. However, it was not clear whether autophagy protects plants from infections, or how much disease-causing microbes interfere with this process for their own benefit.
The microbe that causes late blight of potatoes (called Phytophthora infestans) is infamous for triggering widespread famines in Ireland in the 19th century. This disease-causing microbe continues to pose a serious threat to food security today, and parasitizes plant tissues by releasing proteins called effectors that enter the plant’s cells to subvert the plant’s physiology and counteract its defenses.
Dagdas, Belhaj et al. now report that an effector from P. infestans, called PexRD54, can bind to autophagy-related protein from potato, called ATG8CL, and stimulate the formation of autophagosomes. Further experiments revealed that the PexRD54 effector could outcompete a plant autophagy cargo receptor that would otherwise bind to ATG8CL. This plant cargo receptor contributes to the plant’s defences, and by preventing it from interacting with ATG8CL, PexRD54 makes the plant more susceptible to infection by P. infestans.
These findings show that the PexRD54 effector has evolved to interact with an autophagy-related protein to counteract the plant’s defences. Dagdas, Belhaj et al. suggest that PexRD54 might do this by activating autophagy to selectively eliminate some of the molecules that the plant use to defend itself. Furthermore, P. infestans might also benefit from the nutrients that are released when cellular material is broken down via autophagy. Future work could test these two hypotheses and explore whether other effectors from disease-causing microbes work in a similar way.
https://doi.org/10.7554/eLife.10856.002Introduction
Autophagy is conserved catabolic pathway that sequesters unwanted cytosolic components into newly formed double membrane vesicles, autophagosomes, to direct them to the cell’s lytic compartment (He and Klionsky, 2009). The process plays a vital role in survival of the organism by improving cellular adaptation to environmental and stress conditions (Shintani and Klionsky, 2004). Autophagy provides building blocks and energy for elementary cellular processes by degrading dysfunctional or unnecessary cellular components during nutrient deprivation (Shintani and Klionsky, 2004). However, even though autophagy was initially thought to be a bulk degradation process activated during starvation, recent studies showed that it can act selectively, capturing specific substrates through specialized cargo receptors to respond to a variety of environmental and stress conditions (Stolz et al., 2014).
Autophagy is executed through coordinated action of more than 30 core proteins known as the ATG (autophagy-related) proteins (Lamb et al., 2013). Selective autophagy is regulated through specific interactions of autophagy cargo receptors and ATG8 proteins (Stolz et al., 2014). Autophagy cargo receptors carry a short sequence motif called ATG8-interaction motif (AIM) that binds lipidated ATG8 proteins anchored on autophagosomal membranes. Cargo receptors mediate recognition of a diverse set of cargo (Stolz et al., 2014). For instance, mammalian autophagy cargo receptors NDP52 and optineurin can recognize intracellular pathogenic bacteria and mediate their autophagic removal by sorting the captured bacteria inside the ATG8-coated autophagosomes (Boyle and Randow, 2013). Nevertheless, the precise molecular mechanisms of selective autophagy and the components that regulate it remain unknown (Huang and Brumell, 2014; Mostowy, 2013; Randow, 2011).
In plants, autophagy plays important roles in stress tolerance, senescence, development, and defense against invading pathogens (Patel and Dinesh-Kumar, 2008; Lenz et al., 2011; Vanhee and Batoko, 2011; Li and Vierstra, 2012; Lv et al., 2014; Teh and Hofius, 2014). Specifically, autophagy is implicated in the accumulation of defense hormones and the hypersensitive response, a form of plant cell death that prevents spread of microbial infection (Yoshimoto et al., 2009). However, the molecular mechanisms that mediate defense-related autophagy and the selective nature of this process are poorly understood. Furthermore, how adapted plant pathogens manipulate defense-related autophagy and/or subvert autophagy for nutrient uptake is unknown.
In this study, we investigated how a pathogen interferes with and coopts a plant autophagy pathway. The potato blight pathogen, Phytophthora infestans, is a serious threat to food security, causing crop losses that, if alleviated, could feed hundreds of millions of people (Fisher et al., 2012). This pathogen delivers RXLR-type effector proteins inside plant cells to enable parasitism (Morgan and Kamoun, 2007). RXLR effectors form a diverse family of modular proteins that alter a variety of host processes and therefore serve as useful probes to dissect key pathways for pathogen invasion (Morgan and Kamoun, 2007; Bozkurt et al., 2012). Here, we show that the RXLR effector PexRD54 has evolved to bind host autophagy protein ATG8CL to stimulate autophagosome formation. In addition, PexRD54 depletes the autophagy cargo receptor Joka2 out of ATG8CL complexes to counteract host defenses against P. infestans.
Results and discussion
As part of an in plantascreen for host interactors of RXLR effectors, we discovered that the P. infestans effector PexRD54 associates with ATG8CL, a member of the ATG8 family (Materials and methods, Supplementary files 1,2). The association between PexRD54 and ATG8CL was retained under stringent binding conditions in contrast to other candidate interactors (Supplementary file 1). We validated the association with reverse coimmunoprecipitation after co-expressing the potato ATG8CL protein with the C-terminal effector domain of PexRD54 in planta (Figure 1A,B). In addition, PexRD54 expressed and purified from Escherichia coli directly bound ATG8CL in vitro with high affinity and in a one to one ratio (KD = 383 nM based on isothermal titration calorimetry) (Figure 1C). PexRD54 has two predicted ATG8 Interacting Motifs (AIMs) that match the consensus amino acid sequence W/F/Y-x-x-L/I/V (AIM1 and AIM2, Figure 1A). In planta coimmunoprecipitations of single and double AIM mutants of PexRD54 revealed that AIM2, which spans the last four amino acids of the protein (positions 378–381), is required for association with ATG8CL (Figure 1D). PexRD54AIM2 mutant also failed to bind ATG8CL in vitro (Figure 1E). In addition, ATG8CL bound with high affinity to a synthetic peptide (KPLDFDWEIV) that matches the last 10 C-terminal amino acids of PexRD54 (KD = 220 nM) (Figure 1—figure supplement 1). We conclude that the C-terminal AIM of PexRD54 is necessary and sufficient to bind ATG8CL.
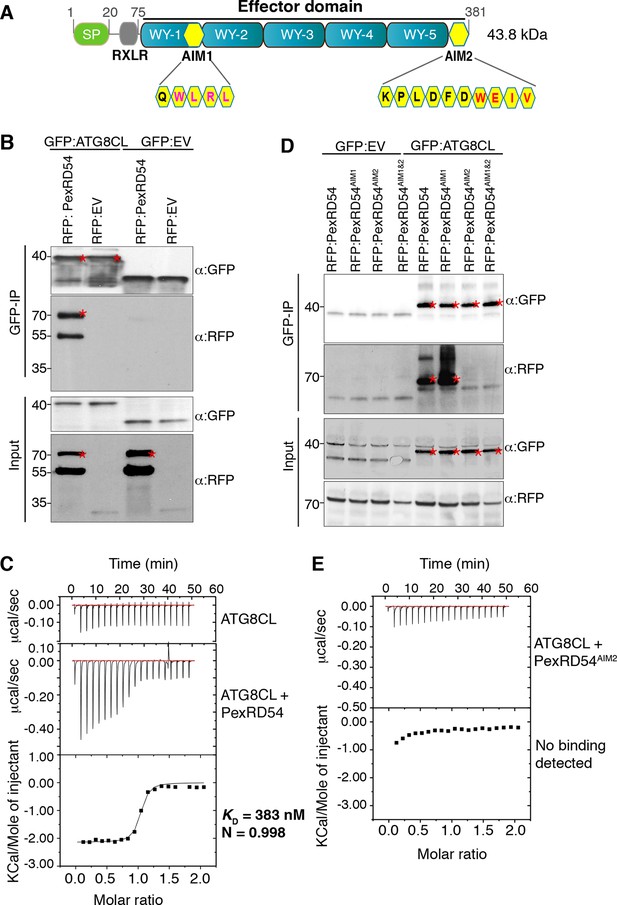
PexRD54 binds to ATG8CL via a C-terminal ATG8 interacting motif (AIM).
(A) Domain organization of PexRD54. PexRD54 is a canonical RXLR effector with five WY folds (Win et al., 2012). The amino acid sequences of candidate AIMs are highlighted in yellow color and indicated in brackets. (B) Validation of PexRD54-ATG8CL association in planta. RFP:PexRD54 or RFP:EV (Empty vector) were transiently co-expressed with GFP:ATG8CL or GFP:EV in N. benthamiana leaves. Immunoprecipitates (IPs) obtained with anti-GFP antiserum and total protein extracts were immunoblotted with appropriate antisera. Stars indicate the expected band sizes. (C) PexRD54 binds ATG8CL in vitro. The binding affinity of PexRD54 to ATG8CL was determined using isothermal titration calorimetry (ITC). Upper panel shows heat differences upon injection of ATG8CL into buffer or PexRD54 and the bottom panel show integrated heats of injection (■) and the best fit (solid line) to a single site binding model using MicroCal Origin. KD=383 nM, N=0.998, ΔH= −8.966 kJ.mol-1 and ΔS = 0.092 J.mol-1.K-1. The values of KD, N, ΔH and ΔS are representative of two independent ITC experiments. (D) ATG8 Interacting Motif 2 (AIM2) mediates ATG8CL binding in planta. RFP:PexRD54, RFP:PexRD54AIM1, RFP:PexRD54AIM2 or RFP:PexRD54AIM1&AIM2 were transiently co-expressed with GFP:ATG8CL or GFP:EV in N. benthamiana leaves. IPs obtained with anti-GFP antiserum and total protein extracts were immunoblotted with appropriate antisera. Stars indicate the expected band sizes. (E) AIM2 mediates ATG8CL binding in vitro. The binding affinity of PexRD54AIM2 to ATG8CL was determined using ITC. Upper panel shows heat differences upon injection of PexRD54AIM2 and the bottom panel show integrated heats of injection (■). No binding was detected between PexRD54AIM2 and ATG8CL.
ATG8 occurs as a family of nine proteins in potato (Figure 2—figure supplements 1–2). PexRD54 bound ATG8CL with ~10 times higher affinity than another ATG8 family member, ATG8IL, in both in planta and in vitro assays (Figure 2). These findings prompted us to use ATG8IL as a negative control in the subsequent experiments.
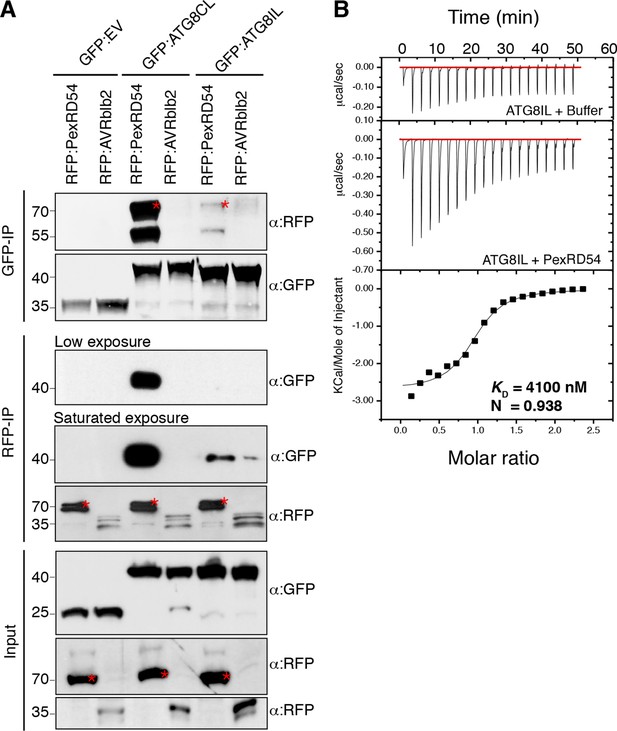
PexRD54 has higher binding affinity to ATG8CL than ATG8IL.
(A) RFP:PexRD54, RFP:AVRblb2 or RFP:EV were transiently co-expressed with GFP:ATG8CL, GFP:ATG8IL or GFP:EV in N. benthamiana leaves and proteins were extracted two days after infiltration and used in immunoprecipitation experiments (IPs). IPs obtained with anti-GFP or anti-RFP antisera and total protein extracts were immunoblotted with appropriate antisera. RFP:AVRblb2 (Bozkurt et al., 2011), an RFP fusion to a different P. infestans RXLR effector, did not associate with ATG8CL or ATG8IL. Both the GFP and RFP IPs indicate higher binding affinity of PexRD54 to ATG8CL than ATG8IL. Stars indicate the expected band sizes. (B) PexRD54 has lower binding affinity to ATG8IL in vitro. The binding affinity of PexRD54 to ATG8IL was determined using isothermal titration calorimetry (ITC). Upper panel shows heat generated upon injection of ATG8IL into buffer or PexRD54 and lower panel shows integrated heats of injection (■) and the best fit (solid line) to a single site binding model using MicroCal Origin. The values of KD = 4100 nM, N = 0.938, ΔH = −11.305 kJ.mol-1 and ΔS= 0.064 J.mol-1K-1 are representative values of two independent ITC experiments. The data show that PexRD54 binds ATG8CL with ~10 times higher affinity than ATGIL.
We then investigated the subcellular localization of PexRD54 within plant cells. N-terminal fusions of PexRD54 to the green fluorescent protein (GFP) or red fluorescent protein (RFP) labelled the nucleo-cytoplasm and mobile punctate structures (Figure 3—figure supplement 1 and Video 1). Immunogold labelling in transmission electron micrographs of cells expressing GFP:PexRD54 revealed a strong signal in electron dense structures that are not peroxisomes (Figure 3—figure supplements 2–3, Dagdas et al., 2016). To determine whether these structures are ATG8CL autophagosomes, we transiently co-expressed RFP:PexRD54 with GFP:ATG8CL in plant cells and observed an overlap between the two fluorescent signals in sharp contrast to RFP:PexRD54AIM2 and RFP:EV negative controls (Figure 3 and Video 2). This indicates that PexRD54 localizes to ATG8CL-marked autophagosomes and its C-terminal AIM is necessary for autophagosome localization. In contrast, RFP:PexRD54 signal overlapped with GFP:ATG8IL-labelled autophagosomes in only 15–20% of observations consistent with its weaker binding affinity to ATG8IL (Figure 3—figure supplement 4).
PexRD54 localizes to mobile endomembrane compartments.
GFP:PexRD54 was transiently expressed in N. benthamiana leaves and examined by confocal laser scanning microscopy, 3 days post infiltration. (Related to Figure 3)
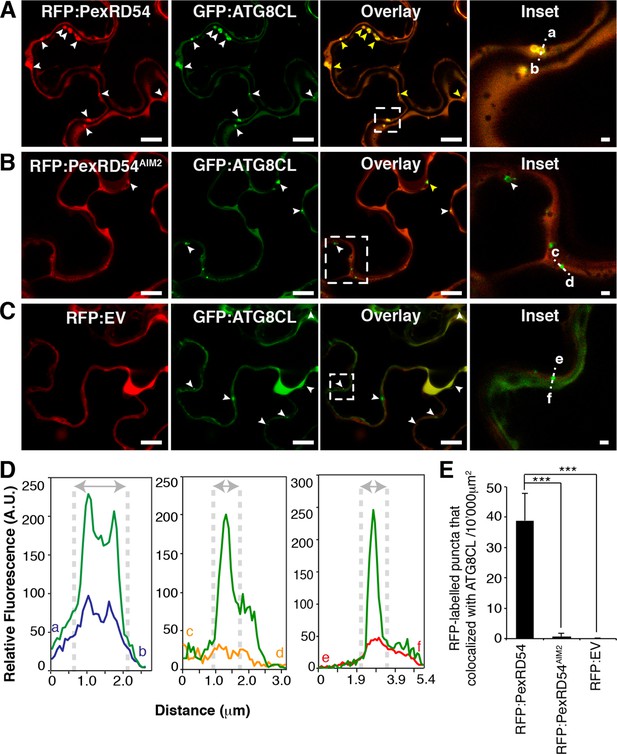
PexRD54 localizes to ATG8CL labelled autophagosomes.
Transient co-expression of GFP:ATG8CL with (A) RFP:PexRD54, (B) RFP:PexRD54AIM2 and (C) RFP control (RFP:EV) in N. benthamiana leaves. Confocal micrographs show single optical sections of RFP:PexRD54, RFP:PexRD54AIM2 and RFP:EV in red, GFP:ATG8CL in green and the overlay indicating colocalization in yellow. White arrowheads point to punctate structures and yellow arrowheads point to puncta where GFP and RFP signals overlap. Far right panels highlight the dotted square region focusing on GFP:ATG8CL labelled puncta in overlaid GFP/RFP channels. Scale bar = 10 μm; scale bar in inset = 1 μm. (D) The intensity plots represent relative GFP and RFP fluorescence signals along the dotted line connecting points a-b, c-d and e-f that span GFP:ATG8CL marked puncta at far right panels. GFP:ATG8CL fluorescence intensity peak overlapped with fluorescence intensity peak of RFP:PexRD54 (left panel) but not with RFP:PexRD54AIM2 (mid panel) or RFP:EV (right panel) validating the localization of RFP:PexRD54 at GFP:ATG8CL labelled autophagosomes. (E) Bar charts showing colocalization of GFP:ATG8CL puncta with RFP:PexRD54, RFP:PexRD54AIM2 or RFP:EV punctate structures. Data are representative of 500 individual images from two biological replicates. Each replicate consists of five independent Z stacks with 50 images each, acquired from five independent leaf areas. (***p<0.001). Error bars represent ± SD.
PexRD54 and ATG8CL colocalize at mobile endomembrane compartments.
RFP:PexRD54 was transiently co-expressed with GFP:ATG8CL in N. benthamiana leaves and examined by confocal laser scanning microscopy, 3 days post infiltration. (Related to Figure 3)
To further confirm that PexRD54-labelled endomembrane compartments are indeed autophagosomes, we investigated the effect of the autophagy inhibitor 3-methyl adenine (3-MA) (Hanamata et al., 2013) on PexRD54 localization. Compared to water, 3-MA treatment reduced the number of PexRD54 and ATG8CL puncta but did not reduce the number of puncta of the trans-Golgi network (TGN) marker VTI12 (Geldner et al., 2009) (Figure 3—figure supplement 5).
Phospholipid modification of a conserved glycine residue at the C-terminus of ATG8 proteins is required for autophagosome formation, and deletion of this terminal glycine yields a dominant negative ATG8 (Hanamata et al., 2013). We deployed a terminal glycine deletion mutant of ATG8CL (ATG8CLΔ) to determine its effect on subcellular distribution of PexRD54 (Figure 3—figure supplement 6). As expected, deletion of the terminal glycine did not affect binding of ATG8CL to PexRD54 (Figure 3—figure supplement 7A). However, GFP:ATG8CLΔ led to the depletion of RFP:PexRD54 labelled puncta presumably because the dominant negative effect of ATG8CLΔ prevented accumulation of RFP:PexRD54 in ATG8CL-labelled autophagosomes (Figure 3—figure supplement 7B–D). In contrast, GFP:ATG8ILΔ, a terminal glycine deletion mutant of ATG8IL, had no effect on the punctate localization of RFP:PexRD54 (Figure 3—figure supplement 7C–D). These experiments independently support the finding that PexRD54 accumulates in ATG8CL autophagosomes.
Increase in ATG8 labelled puncta is widely used as a functional readout of autophagic activity (Hanamata et al., 2013; Bassham, 2015). In samples expressing PexRD54, we noticed a ~fivefold increase in the number of ATG8CL marked autophagosomes compared to control samples expressing PexRD54AIM2 or empty vector control (Figure 4, Video 3). In contrast, PexRD54 did not alter the number of ATG8IL autophagosomes consistent with the weak binding noted between these two proteins (Figure 4A). This indicates that PexRD54 stimulates the formation of ATG8CL autophagosomes.
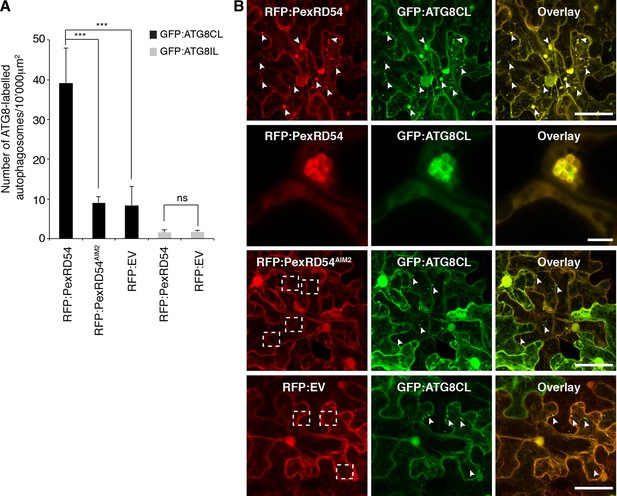
PexRD54 increases the number of ATG8CL autophagosomes.
(A) Co-expression of RFP:PexRD54, but not RFP:PexRD54AIM2 or RFP:EV significantly enhanced the number of GFP:ATG8CL-labelled autophagosomes in N. benthamiana. Bar charts display the number of GFP:ATG8CL or GFP:ATG8IL-labelled autophagosomes in the presence of RFP:PexRD54, RFP:PexRD54AIM2 or RFP:EV. GFP:ATG8CL autophagosomes were significantly enhanced by the expression of RFP:PexRD54 (***p<0.001). GFP:ATG8IL autophagosomes were not significantly enhanced by expression of RFP:PexRD54 (ns=statistically not significant). The data are representative of 500 individual images from two biological replicates. Each replicate consists of five independent Z stacks with 50 images each, acquired from five independent leaf areas. (B) GFP:ATG8CL was transiently co-expressed with RFP:PexRD54, RFP:PexRD54AIM2 or RFP:EV in N. benthamiana leaves and examined by confocal laser scanning microscopy 3 days post infiltration. Maximum projections of images show that RFP:PexRD54 increases the number of GFP:ATG8CL- labelled autophagosomes. RFP:PexRD54AIM2 and RFP:EV were used as controls. Arrowheads point to punctate structures. Regions where GFP or RFP-labelled puncta do not overlap are indicated with dotted squares. Scale bar=50 μm. Zoomed single plane images shown in the second panel indicate that larger puncta co-labelled by RFP:PexRD54 and GFP:ATG8CL are ring-shaped autophagosome clusters. Scale bar=10 μm.
RFP:PexRD54 and GFP:ATG8CL colocalize at mobile ring shaped clusters.
RFP:PexRD54 was transiently co-expressed with GFP:ATG8CL in N. benthamiana leaves and examined by confocal laser scanning microscopy, 3 days post infiltration. (Related to Figure 4)
Next, we set out to determine the effect of PexRD54 on autophagic flux. Treatment of RFP:ATG8CL expressing leaves with the specific vacuolar ATPase inhibitor concanamycin-A (Bassham, 2015) increased the number of ATG8CL-labelled puncta both in the presence of PexRD54 or controls (PexRD54AIM2 or vector control) indicating that PexRD54 does not block autophagic flux (Figure 5A–B). We also confirmed these observations using western blot analyses. PexRD54, but neither PexRD54AIM2 or vector control, increased the levels of GFP:ATG8CL protein 3 days after co-expression in planta (Figure 5C). Treatment of three-day samples with E64d, an inhibitor of vacuolar cysteine proteases (Bassham, 2015), further increased protein levels of GFP:ATG8CL. This further confirms that PexRD54 stimulates autophagy rather than blocking autophagic flux (Figure 5C). PexRD54 did not alter the accumulation of GFP:ATG8IL or control GFP protein, confirming that PexRD54 increases ATG8CL protein accumulation specifically (Figure 5C). Consistent with these observations, we noted an increase in GFP:ATG8CL levels, but not in control GFP, during P. infestans infection relative to the mock infection (Figure 5—figure supplement 1).
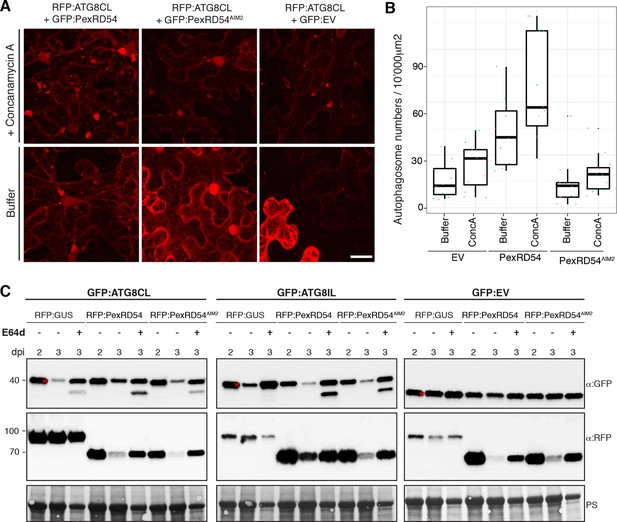
PexRD54 does not block autophagic flux.
(A–B) ConcanamycinA treatment increases the number of autophagosomes in PexRD54 expressing samples.RFP:ATG8CL was transienly coexpressed with PexRD54, PexRD54AIM2 and empty vector controls in N. benthamiana. Two days after infiltration, leaves were treated with concanamycinA (conA) or infiltration buffer and number of autophagosomes was counted 24 hr after treatment. ConA treatment significantly increased the number of autophagosomes in PexRD54 expressing cells (p<0.05), confirming PexRD54 does not block autophagic flux. Scale bar=10 μm. (C) E64D treatment increases ATG8CL protein levels in PexRD54 expressing samples. GFP:ATG8CL, GFP:ATG8IL and GFP:EV were transiently coexpressed with RFP:GUS, RFP:PexRD54 or RFP:PexRD54AIM2 in N. benthamiana leaves and protein levels in total extracts were determined two and 3 days post infiltration (dpi). RFP:PexRD54 increased protein levels of GFP:ATG8CL but not GFP:ATG8IL consistent with stronger binding affinity of PexRD54 to ATG8CL. RFP:PexRD54 did not increase protein levels of GFP:EV, suggesting that protein level increase depends on ATG8CL binding and that PexRD54 does not increase protein levels in general. The samples were also treated with E64d to measure autophagic flux. In RFP:PexRD54 coexpressed 3 dpi samples, E64d treatment increased ATG8CL protein levels even more suggesting PexRD54 does not block autophagic flux. Hence, protein level increase is a result of stimulation of autophagy. The blots were stained with Ponceau stain (PS) to show equal loading.
The presence of a functional AIM in PexRD54 prompted us to hypothesize that this effector perturbs the autophagy cargo receptors of its host plants. Recently, Joka2 was reported as a selective autophagy cargo receptor of Solanaceous plants that also binds ATG8 via an AIM (Svenning et al., 2011; Zientara-Rytter et al., 2011) (Figure 6A). Indeed, in planta coimmunoprecipitation assays confirmed that potato Joka2, but not the AIM mutant, Joka2AIM, associated with ATG8CL (Figure 6—figure supplement 1). Joka2 association with ATG8CL was somewhat specific given that this cargo receptor failed to coimmunoprecipitate with ATG8IL (Figure 6—figure supplement 2). Joka2, but not Joka2AIM, also markedly increased the number of GFP:ATG8CL autophagosomes (Figure 6—figure supplement 3, Video 4), and enhanced ATG8CL protein levels (Figure 6—figure supplement 4). This indicates that Joka2 also activates ATG8CL-mediated selective autophagy.
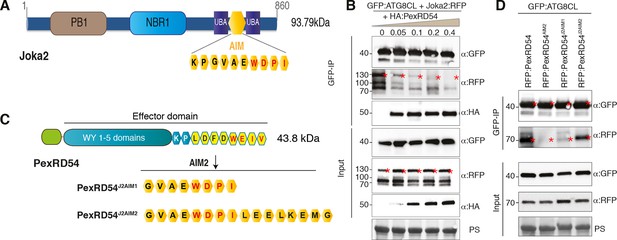
PexRD54 is a competitive antagonist of the plant selective autophagy cargo receptor Joka2.
(A) Domain organization of Joka2. (B) PexRD54 reduces binding of Joka2 to ATG8CL in a dose-dependent manner. GFP:ATG8CL (OD=0.2) and Joka2:RFP (OD=0.2) were transiently co-expressed with varying Agrobacterium concentrations (from OD=0 to OD=0.4) carrying HA:PexRD54 construct in N. benthamiana (OD=optical density of Agrobacterium cells). Joka2:RFP is depleted in GFP:ATG8CL pulldowns as the expression of HA:PexRD54 increased. Immunoprecipitates (IPs) obtained with anti-GFP antiserum and total protein extracts were immunoblotted with appropriate antisera. Stars indicate expected band sizes. (C) Schematic illustrations of PexRD54 variants (PexRD54J2AIM1 and PexRD54J2AIM2) with Joka2 AIM peptides. (D) Replacement of PexRD54 AIM with Joka2 AIM fragments decreases ATG8CL binding affinity. Immunoblots showing binding affinity of PexRD54, PexRD54AIM2, PexRD54J2AIM1 or PexRD54J2AIM2 to GFP:ATG8CL. IPs obtained with anti-GFP antiserum and total protein extracts were immunoblotted with appropriate antisera. Stars indicate expected band sizes.
RFP:ATG8CL and Joka2:GFP colocalize at mobile endomembrane compartments.
RFP:ATG8CL was transiently coexpressed with Joka2:GFP in N. benthamiana leaves and examined by confocal laser scanning microscopy, 3 days post infiltration. (Related to Figure 6)
Given that both PexRD54 and Joka2 bind ATG8CL via their respective AIMs, we hypothesized that PexRD54 interferes with the Joka2-ATG8CL complex. We tested our hypothesis by performing coimmunoprecipitation experiments between Joka2:RFP and GFP:ATG8CL in the presence or absence of PexRD54. Remarkably, ATG8CL complexes were depleted in Joka2 in the presence of PexRD54 relative to the PexRD54AIM2 and vector control (Figure 6—figure supplement 5). Consistently, Joka2 binding to ATG8CL decreased with increasing PexRD54 concentrations (Figure 6B). The distinct AIMs of PexRD54 and Joka2 presumably determine the effect observed in these competition experiments. To further test this, we replaced the functional PexRD54 AIM with two sequences that cover the Joka2 AIM:GVAEWDPI (PexRD54J2AIM1) and GVAEWDPILEELKEMG (PexRD54J2AIM2) (Figure 6C). Both PexRD54J2AIM1 and PexRD54J2AIM2 associated with ATG8CL to a lesser extent than wild-type PexRD54 (Figure 6D), and were less effective than PexRD54 in depleting Joka2 out of ATG8CL complexes (Figure 6—figure supplement 6). These findings reveal that PexRD54 antagonizes Joka2 for ATG8CL binding.
Finally, we investigated the degree to which activation of Joka2-ATG8CL-mediated autophagy contributes to pathogen defense. Overexpression of Joka2, but not Joka2AIM, significantly restricted the size of the disease lesions caused by P. infestans (Figure 7A–B). Conversely, virus-induced gene silencing of Joka2 resulted in increased disease lesions (Figure 7—figure supplement 1). This indicates that Joka2-mediated selective autophagy contributes to defense against this pathogen. Remarkably, PexRD54 counteracted the enhanced resistance conferred by Joka2 whereas PexRD54AIM2 failed to reverse this effect (Figure 7C–D). We conclude that PexRD54 counteracts the positive role of Joka2-mediated selective autophagy in pathogen defense.
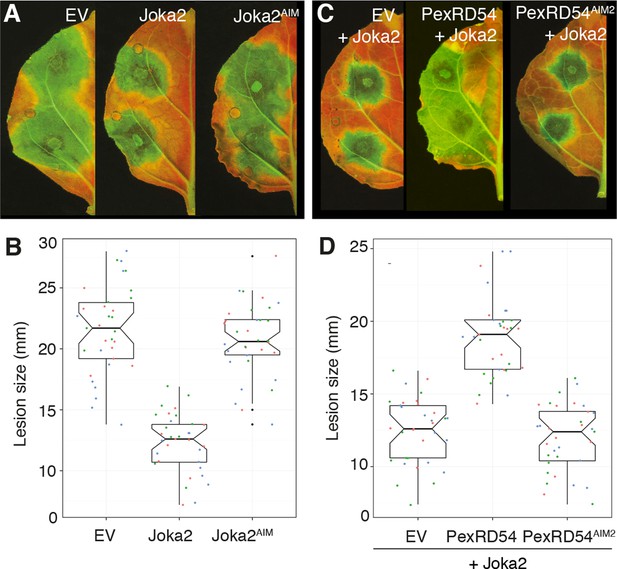
PexRD54 counteracts the enhanced resistance conferred by Joka2.
(A) Overexpression of Joka2:RFP limits P. infestans colonization. Halves of N. benthamiana leaves expressing RFP:EV, Joka2:RFP and Joka2AIM:RFP were infected with P. infestans and pathogen growth was determined by lesion sizes measured 6 days post-inoculation. (B) Categorical scatter plots show lesion diameters of 11 infections sites from three independent biological replicates pointed out by three different colors. Similar p values (p<0.001) were obtained in three independent biological repeats. (C) PexRD54 counteracts the effect of Joka2 on P. infestans colonization. Joka2:RFP was co-expressed with HA:EV, HA:PexRD54 or HA:PexRD54AIM2 in N. benthamiana leaves which are then inoculated with P. infestans. Joka2:RFP failed to limit pathogen growth in the presence of PexRD54, whereas it could still restrict pathogen growth in the presence of PexRD54AIM2 or vector control (EV). (D) Categorical scatter plots show lesion diameters of 11 infections sites from three independent biological replicates pointed out by three different colors. Similar p values (p<0.001) were obtained in three independent biological repeats.
As demonstrated in mammalian systems, eukaryotic cells employ autophagy to defend against invading pathogens (Boyle and Randow, 2013; Randow and Youle, 2014). In turn, pathogens can deploy effectors to avoid autophagy and enable parasitic infection (Baxt et al., 2013). For instance, to counteract antimicrobial autophagy, intracellular bacterial pathogen Legionella pneumophila secretes a type IV effector protein RavZ that impedes autophagy by uncoupling ATG8-lipid linkage (Choy et al., 2012). In this study, we show that a plant pathogen effector has evolved an ATG8 interacting motif to bind with high affinity to the autophagy protein ATG8CL and stimulate the formation of ATG8CL-marked autophagosomes. Unlike the Legionella effector RavZ, PexRD54 activates selective autophagy possibly to eliminate defense-related compounds or to reattribute cellular resources by promoting nutrient recycling. Our results show that, in addition to disrupting, pathogens can also activate autophagy for their own benefit (Figure 8).
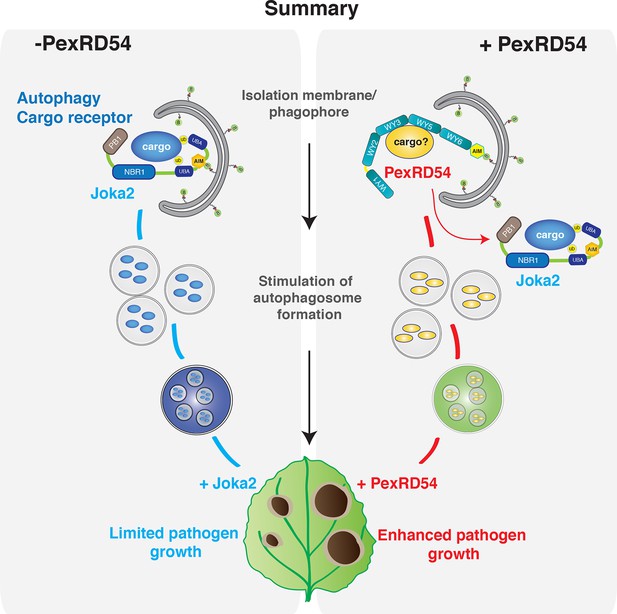
PexRD54 outcompetes the autophagy cargo receptor Joka2 and enhances virulence.
https://doi.org/10.7554/eLife.10856.032Additionally, we show that the effector competes with the host cargo receptor Joka2 and depletes it out of ATG8CL autophagosomes to promote disease susceptibility. Thus P. infestans coopts the host cell’s endomembrane compartment to promote its own growth at the cost of the cell’s physiology (Bozkurt et al., 2015). Joka2 could contribute to immunity by inducing autophagic removal of plant or pathogen molecules that negatively affect host defenses. It will be interesting in the future to determine the identity of potential defense-related cargo carried by Joka2 (Figure 8).
The physiological roles of selective autophagy and the molecular mechanisms involved remain to be determined both in plants and animals. Characterization of additional host cargo receptors and interactome analysis of particular ATG8 proteins should improve our understanding of how selective autophagy operates in response to a variety of stress conditions including immunity. In the potato genome, we identified nine ATG8 genes. Both Joka2 and the effector showed higher affinity to ATG8CL compared to ATG8IL (Figure 2 and Figure 6—figure supplement 2). This indicates that variation between plant ATG8 proteins may contribute to the selective nature of autophagy.
This work further highlights the intricate changes in endomembrane compartment formation that take place during plant-microbe interactions (Bozkurt et al., 2015; Lipka and Panstruga, 2005; Kwon et al., 2008; Ivanov et al., 2010; Wang et al., 2010). Future studies will need to consider pathogen-directed modulation of the spatio-temporal dynamics of autophagy and subcellular trafficking during host infection. It will be interesting to determine whether other plant pathogens also secrete effectors that evolved an ATG8 interacting motif or target autophagy in other ways. These effectors would serve as valuable tools to dissect the mechanisms of defense-related selective autophagy.
Materials and methods
Identification of candidate ATG8 interacting motifs in PexRD54
Request a detailed protocolATG8 interacting motif (AIM), also known as LC3 interacting region (LIR), mediates interaction of cargo receptors or adaptors with ATG8 proteins anchored in autophagosome membranes (Birgisdottir et al., 2013). It follows the W/F/Y-xx-L/I/V amino acid consensus. Initially, we identified two AIM candidates in PexRD54, AIM1 and AIM2 based on the manual search of the consensus sequence mentioned earlier. Then, we confirmed these AIM candidates using the recently published iLIR software (Kalvari et al., 2014). This software assigned AIM1 with an iLIR score of 12 (1.1e-1) and AIM2 with an iLIR score of 23 (3.2e-3).
Sequence analyses and identification of ATG8CL
Request a detailed protocolTo determine the ATG8 variant(s) specifically targeted by PexRD54 among various host ATG8 family members, we performed a BLASTP search (Altschul et al., 1990) against solanaceous plant proteomes including Solanum tuberosum (potato), Solanum lycopersicum (tomato) and Nicotiana benthamiana using Arabidopsis thaliana ATG8C as the query protein sequence. We found nine ATG8 members in S. tuberosum, seven in S. lycopersicum, and eight in N. benthamiana. To verify the gene calls and open reading frame predictions of the putative orthologs in these three species, we performed a sequence alignment of the family members in each species using the Clustal X program (v2) (Larkin et al., 2007) and compared it to the published A. thaliana ATG8 sequences. We found evidence of misannotation for two S. lycopersicum sequences (Solyc10g006270 and Solyc08g078820) and three others in N. benthamiana (NbS00015425g0005, NbS00003316g0005 and NbS00003005g0010). The two S. lycopersium sequences and the NbS00003005g0010 sequence from N. benthamiana carried extra nucleotides before the likely start codon, which were corrected accordingly. A TBLASTN search against the N. benthamiana scaffolds followed by a BLASTP search of the different exons allowed curation of the two other sequences. Clustal X program was used for multiple sequence alignment of ATG8 variants. Boxshade server (http://embnet.vital-it.ch/software/BOX_form.html) was used to visualize the sequence alignment.
Phylogenetic analyses
Request a detailed protocolThe phylogenetic tree of ATG8 homologs in plants was constructed with the neighbor-joining method using ATG8-like curated proteins from S. tuberosum, S. lycopersium, N. benthamina, and A. thaliana. The phylogenetic tree was constructed using MEGA5 (Kumar et al., 2001) with bootstrap values based on 1000 iterations. ATG8CL variants in N. benthamiana and potato have identical amino acid sequences.
Cloning procedures and plasmid constructs
Request a detailed protocolAll primers used in this study are listed in Supplementary file 3. PexRD54 and Joka2 were amplified using polymerase chain reaction (PCR) from genomic DNA of Phytophthora infestans isolate T30-4 and Solanum tuberosum cv. Désirée cDNA, respectively. To amplify both amplicons, we used Phusion proof reading polymerase (New England Biolabs, UK) and primer pairs listed in Supplementary file 3. All amplicons were subsequently cloned into the pENTR/D-Topo Gateway entry vector (Invitrogen, UK). The ATG8CL, ATG8CLΔ, ATG8IL, and ATG8ILΔ entry clones were custom synthesized into pUC57-AmpR using the sequences matching S. tuberosum ATG8CL or ATG8IL genes flanked by attL1 and attL2 gateway sites (Genewiz, UK). The destination constructs GFP:PexRD54, RFP:PexRD54, Joka2:RFP, Joka2:GFP, RFP:ATG8CL, GFP:ATG8CL, GFP:ATG8CLΔ, GFP:ATG8ILΔ, HA:PexRD54 were generated by Gateway LR recombination reaction (Invitrogen) of the corresponding entry clone and Gateway destination vectors pH7WGR2 (N-terminal RFP fusion), pK7WGF2 (N-terminal GFP fusion), pB7FWR2 (C-terminal GFP fusion), pB7RWG2 (C-terminal RFP fusion) and pK7WGF2 (N-terminal HA fusion, generated in house by replacement of GFP with an HA tag), respectively (Karimi et al., 2002). The GFP:PexRD54AIM2, RFP:PexRD54AIM1, RFP:PexRD54AIM2, RFP:PexRD54AIM1+2 RFP:PexRD54KPLDFDWEIV, RFP:PexRD54J2AIM1, RFP:PexRD54J2AIM2, HA:PexRD54AIM2, HA:PexRD54AIM2, HA:PexRD54J2AIM1, HA:PexRD54J2AIM2, Joka2AIM:RFP constructs were generated as follows:PexRD54 and Joka2 mutant constructs (PexRD54AIM1, PexRD54AIM2, PexRD54AIM1+2, PexRD54KPLDFDWEIV, RFP:PexRD54J2AIM1, RFP:PexRD54J2AIM2, and Joka2AIM:RFP) were cloned into the pENTR/D-Topo Gateway entry vector (Invitrogen) by PCR amplification with the primers carrying desired mutations (Supplementary file 3) using PexRD54 and Joka2 entry clones as templates followed by TOPO cloning procedure (Invitrogen). Templates were then eliminated by one-hour Dpn-I (New England Biolabs) restriction digestion at 37°C. Next, the entry clones of PexRD54 and Joka2 mutants were recombined into destination vectors pH7WGR2 or pB7RWG2 by Gateway LR reaction (Invitrogen). pTRBO FLAG:PexRD54 construct used for coimmunoprecipitations was custom synthesized (Genscript, , New Jersey, USA).
Generation of ATG8 interacting motif (AIM) mutants
Request a detailed protocolTo generate ATG8 interacting motif (AIM) mutants in PexRD54 and Joka2, the conserved tryptophan and leucine residues of the canonical AIMs were both mutated to Alanine as previously done (Zientara-Rytter et al., 2011). In PexRD54AIM1, PexRD54AIM2 and Joka2AIM, the AIM motifs e.g. 'WLRL', 'WEIV' and 'WDPI' were mutated to 'ALRA', 'AEIA' and 'ADPA', respectively.
Biological material
Request a detailed protocolNicotiana benthamiana plants were grown and maintained throughout the experiments in a greenhouse at 22–25°C with high light intensity. Phytophthora infestans cultures were grown in plates with rye sucrose agar (RSA) media for 12–14 days as described elsewhere (Song et al., 2009). Sporangia were harvested from plates using cold water and zoospores were collected 1–3 hr after incubation at 4°C. Infection assays were performed by droplet inoculations of zoospore solutions of P. infestans on 3–4-week-old detached N. benthamiana leaves as described previously (Song et al., 2009; Saunders et al., 2012). For all infection assays, P. infestans isolate 88,069 was used.
Transient gene-expression assays in N. benthamiana
Request a detailed protocolTransient gene-expression in planta was performed by delivering T-DNA constructs with Agrobacterium tumefaciens GV3101 strain into 3–4-week-old N. benthamiana plants as described previously (Bozkurt et al., 2011). For transient co-expression assays, A. tumefaciens strains carrying the plant expression constructs were mixed in a 1:1 ratio in agroinfiltration medium [10 mM MgCl2, 5 mM 2-(N-morpholine)-ethanesulfonic acid (MES), pH 5.6] to a final OD600 of 0.2, unless otherwise stated.
Coimmunoprecipitation (Co-IP) experiments and liquid chromatography tandem mass spectrometry (LC-MS/MS) analysis
Request a detailed protocolProteins were transiently expressed by A. tumefaciens-mediated transient expression (agroinfiltration) in N. benthamiana leaves and harvested 2 or 3 days post infiltration. Co-IP experiments and preparation of peptides for liquid chromatography–tandem mass spectrometry (LC-MS/MS) was performed as described previously (Bozkurt et al., 2011). Except the stringent PexRD54 immunoprecipitation assay (Stringent IP), all IPs were done using 150 mM NaCl buffer and 0.15% detergent concentration. For the stringent IP, the concentrations of salt and the detergent were increased to 250 mM and to 0.5%, respectively. LC-MS/MS analysis was performed with a LTQ Orbitrap mass spectrometer (Thermo Fisher Scientific, UK) and a nanoflow-HPLC system (nanoACQUITY; Waters Corp., UK) as described previously (Petre et al., 2015). LC-MS/MS data processing and protein identification were done as described previously (Petre et al., 2015).
In planta association of PexRD54 and Joka2 with either ATG8CL, ATG8CLΔ or ATG8IL constructs was tested by co-IP assays as follows: constructs were transiently co-expressed in N. benthamiana leaves by agroinfiltration followed by protein extraction 2–3 days post infiltrations. Protein extraction, purification and western blot analysis steps were performed as described previously (Saunders et al., 2012; Bozkurt et al., 2011; Oh et al., 2009). Monoclonal FLAG M2 antibody (Sigma-Aldrich, UK), polyclonal GFP/RFP antibodies (Invitrogen, UK), and polyclonal HA (Sigma-Aldrich, UK) antibody were used as primary antibodies, and anti-mouse antibody (Sigma-Aldrich, UK) and anti-rat (Sigma-Aldrich, UK) antibodies were used as secondary antibodies.
Determination of ATG8CL protein levels
Request a detailed protocolTo test if PexRD54 increased protein levels of ATG8CL, GFP:ATG8CL was co-expressed with RFP:GUS, RFP:PexRD54 and RFP:PexRD54AIM2 in N. benthamiana leaves. Leaf samples were collected 2 and 3 days after infiltration. Total proteins were extracted as described previously (Saunders et al., 2012; Oh et al., 2009) and immunoblots were performed using the appropriate antisera. The same experimental setup was also used to test whether Joka2 increases protein levels of ATG8CL. Joka2:RFP, Joka2AIM:RFP and RFP:GUS constructs were co-expressed with GFP:ATG8CL construct in N. benthamina leaves and total proteins were extracted 2 and 3 days after infiltration. Immunoblots were developed using appropriate antisera. To assay ATG8CL protein levels during infection with P. infestans, N. benthamiana leaves were first infiltrated with GFP:ATG8CL construct and infected one day later with droplets from a zoospore solution of P. infestans as described earlier (Song et al., 2009). Water droplets were used as mock treatment. Protein extracts were prepared 2 and 3 days after infection and protein levels of GFP:ATG8CL were detected by immunoblots using a polyclonal GFP-HRP antibody (Santa Cruz, Texas, USA).
PexRD54/Joka2 in planta competition assays for ATG8CL binding
Request a detailed protocolJoka2:RFP was transiently co-expressed with GFP:ATG8CL in the presence of HA:PexRD54, HA:PexRD54AIM, or HA:EV constructs in N. benthamiana leaves. Total proteins extracts prepared 2 days after infiltration were then used in anti-GFP Co-IPs. Purified protein complexes were separated by SDS/PAGE and immunoblotted to detect RFP, GFP and HA signals. Similar competition experiment was conveyed with increased HA:PexRD54 A. tumefaciens concentrations (OD600=0, 0.05, 0.1, 0.2 or 0.4) while Joka2:RFP and GFP:ATG8CL A. tumefaciens concentrations were kept fixed at OD600= 0.2. Protein extracts prepared two days after infiltration were used in GFP-IPs as described above. Joka2-ATG8CL binding assays were carried out by co-expressing Joka2:RFP and GFP:ATG8CL in the presence of HA:PexRD54, HA:PexRD54AIM2, HA:PexRD54J2AIM1 (PexRD54 AIM (LDFDWEIV) replaced by Joka2 AIM (GVAEWDPI)), HA:PexRD54J2AIM2 (PexRD54 AIM replaced by Joka2 AIM plus 8 additional amino acids at the C-terminus (GVAEWDPILEELKEMG)) or HA:EV. GFP-IPs were performed as described above.
Infection assays
Request a detailed protocolInfection assays assessing the effect of Joka2 overexpression on P. infestans colonization were performed as follow: Joka2:RFP, Joka2AIM:RFP or RFP EV were transiently overexpressed side by side on either halves of independent N. benthamiana leaves. Twenty-four hours after expression, the infiltrated leaves were detached and inoculated with P. infestans 88,069 on two or three spots on each half leaf. P. infestans growth was monitored by UV photography 6 days after infection. Colonization was quantified by measuring the diameter of the lesion on each inoculated spot and values from three independent biological replicates were used to generate the scatter plots.
To demonstrate whether PexRD54 could alleviate the effect of Joka2 on pathogen growth, Joka2:RFP was co-expressed with HA:PexRD54, HA:PexRD54AIM2 or HA:EV in N. benthamiana leaves, which were infected and monitored as described above. For all infection assays, experiments were repeated three times with minimum 10 infection spots. Lesion diameters were measured 6 days after infection.
Joka2 silencing assays
Request a detailed protocolVirus induced gene silencing of Joka2 was performed in N. benthamiana as described previously (Liu et al., 2002). Two different TRV2/pYL279 constructs were designed to target both 5’-and 3’ ends of Joka2. TRV:Joka2-1 and TRV2:Joka2-2 targeted the region between 340 and 639 and between 1942 and 2241, respectively. The primers were used for generating TRV2:Joka2-1 and TRV2:Joka2-2 are listed in Supplementary file 3. TRV2:GFP was used as a negative control as described previously (Chaparro-Garcia et al., 2015).
Suspensions of Agrobacterium tumefaciens strain GV3101 harboring TRV1/pYL155 and TRV2:Joka2-1 or TRV2:Joka2-2 were mixed in a 2:1 ratio in infiltration buffer (10 mM MES (2-[N-morpholino]ethanesulfonic acid), 10 mM magnesium chloride (MgCl2), pH 5.6) to a final OD600 of 0.3. As a control, we used TRV2:GFP. Two-week-old N. benthamiana plants were infiltrated with A. tumefaciens for VIGS assays and upper leaves were used 2–3 weeks later for P. infestans infections. UV photographs were taken 5 days post infection. Each experiment was repeated at least three times with minimum 10 infection spots and disease lesion areas were measured using ImageJ. Each lesion size value was normalized following the formula 'Nr = Or * Ar /A', where N is the normalized lesion size value, O is the original lesion size value, r is the biological repeat, Xr is the average of all O values of the biological repeat r, and A is the average of all O values. Silencing levels were confirmed using RT-PCR. Total RNA was extracted using RNAeasy Plant Mini Kit (Qiagen, UK) and treated with Ambion TURBO DNA-free according to manufacturer’s protocol. 1.0 μg of DNase treated RNA was used for cDNA synthesis using SuperScript III Reverse Transcriptase (Invitrogen). RT-PCR was performed with the following program: 1 cycle with 3 min at 95°C, followed by 35 cycles with 95°C at 30 s, 56°C at 30 s and 72°C at 30 s. Primers pairs used for cDNA amplification were Joka2-TRV1-F and Joka2-TRV1-R, and Joka2-TRV2-R and Joka2-TRV2-F (described in Supplementary file 3). NbEF1α was used to normalize transcript abundance (Segonzac et al., 2011).
Live-cell imaging by confocal laser scanning microscopy
Request a detailed protocolAll microscopy analyses were performed on live leaf tissue 3 days post agroinfiltration. N. benthamiana leaf patches were cut and mounted in water and analyzed on a Leica TCS SP5 confocal microscope (Leica Microsystems, Germany) using 63x water immersion objective. The GFP and RFP probes were excited using 488 and 561 nm laser diodes and their fluorescent emissions were collected at 495–550 nm and 570–620 nm, respectively. To avoid bleed-through from different fluorophores, co-localization images were taken using sequential scanning between lines and acquired using multi-channels.
Image processing and data analysis
Request a detailed protocolConfocal microscopy images were processed with the Leica LAS AF software, ImageJ (2.0) and Adobe PHOTOSHOP CS5 (12.0) programs. Images for quantification of autophagosome numbers were obtained from 50 Z stacks consisting of 1 μm depth field multi-layered images with similar settings for all samples. To detect and quantify punctate structures in one channel (green channel or red channel or overlay channel (green channel images superimposed with red channel ones)), the Z stacks were separated into individual images with the ImageJ (2.0) program and analyzed. The counting procedure was based on a naked-eye detection of punctate structures to avoid cytoplasm noise and dual counting of autophagosomes within the same stack. Histograms were generated with mean of punctate numbers generated from stacks obtained in two independent biological experiments. Statistical differences were assessed by means of a two-tailed t-test assuming unequal variance as implemented in StatPlus LE package (AnalystSoft, Washington, USA). Measurements were significant when p<0.05 and highly significant when p<0.001.
Electron microscopy and immunogold labelling
Request a detailed protocolLeaf samples were embedded in LR White as described (Liu et al., 2002) except sections were picked up on gold grids before immunogold labeling. All labeling procedures were carried out at room temperature. Grids were floated, section-side down, on drops of 50 mM glycine/PBS (150 mM NaCl, 10 mM phosphate, pH 7.4) for 15 min then on Aurion blocking buffer (5% BSA/0.1% cold water fish gelatin/5–10% normal goat serum/15 mM NaN3/PBS, pH7.4) (Aurion, the Netherlands) for 30 min then briefly equilibrated in incubation buffer (0.1% (v/v) BSA-C (actetylated BSA; Aurion) /PBS, pH 7.3) before a 90 min’ incubation with the primary antibody. Serial sections on separate grids were labelled with either a rabbit polyclonal anti-GFP Abcam ab6556 (Abcam, UK) at 1/250, or a rabbit polyclonal anti-catalase AS09 501 (Newmarket Scientific, UK) at 1/1000. Grids were washed 6x5 min in drops of incubation buffer then placed on the drops of goat anti-rabbit secondary antibody conjugated to 10 nm gold (BioCell, Agar Scientific Ltd., Essex, UK), diluted to 1/50 in incubation buffer, for 90 min. After 6x5 min washes in incubation buffer and 2x5 min washes in PBS, the grids were briefly washed in water and contrast stained with 2% (w/v) uranyl acetate. Grids were viewed in a FEI Tecnai 20 transmission electron microscope (FEI, the Netherlands) at 200 kV and digital TIFF images were taken using an AMT XR60B digital camera (Deben, UK) to record TIFF files. Bar charts were generated by counting the clustered (>4 gold particles close to each other) gold particles on each image.
Chemical treatments
Request a detailed protocol3-Methyl adenine (3-MA), a phosphatidylinositol 3-kinase (PI3K) inhibitor is widely used to inhibit autophagosome formation (Hanamata et al., 2013). N. benthamiana leaves transiently expressing GFP:ATG8CL-RFP:PexRD54, GFP:PexRD54-RFP:ATG8CL or YFP:VTI12 were infiltrated with 5 mM 3-MA. Both GFP:PexRD54 and RFP:ATG8CL constructs were expressed side by side to monitor the effect of 3-MA treatment on stimulation of autophagosome formation by PexRD54. Punctate structures were visualized using confocal microscopy 6–10 hr after 3-MA treatment. Bar charts were generated with the number of punctate structures obtained from maximum projections of Z-stack images of two independent biological experiments.
The cysteine protease inhibitor E64d is widely used for measuring autophagic flux (Bassham, 2015). To determine whether PexRD54 blocked autophagic flux, at 2 dpi we infiltrated leaves with 100 μM E64D and kept them overnight in the dark. At 3 dpi, we collected E64d treated and untreated samples and analyzed total protein levels using appropriate antisera. Concanamycin A (2 μM in agro infiltration medium) was infiltrated into leaves of N. benthamiana transiently expressing ATG8CL and PexRD54, PexRD54AIM2 or empty vector control constructs. The leaves were than incubated in dark at 20°C for 24 hr. ATG8CL-labelled puncta were visualized using confocal microscopy 24 hr after concanamycin A treatment.
Cloning, expression and purification of PexRD54 and PexRD54AIM2 variants for in vitro studies
Request a detailed protocolDNA encoding PexRD54 residues Val92 to Val381 (lacking secretion and translocation signals) was amplified from RFP:PexRD54 (using primers shown in Supplementary file 3) and cloned into the vector pOPINS3C, resulting in an N-terminal 6xHis-SUMO tag with PexRD54, linked by a 3C cleavage site (Berrow et al., 2007). Recombinant protein was produced using E. coli BL21-Arabinose Inducible (AI) cells. Cell cultures were grown in Power Broth at 37°C to an A650 0.4–0.6 followed by induction with 0.2% (w/v) L-arabinose and overnight incubation at 18°C. Pelleted cells were resuspended in buffer A (50 mM Tris-HCl pH 8, 500 mM NaCl, 50 mM glycine, 5% (v/v) glycerol and 20 mM imidazole supplemented with EDTA free protease inhibitor tablets (one tablet per 40 ml buffer)) and lysed by sonication. The clarified cell lysate was applied to a Ni2+-NTA column connected to an AKTA Xpress system. 6xHis-SUMO-PexRD54 was step-eluted with elution buffer (buffer A containing 500 mM imidazole) and directly injected onto a Superdex 75 26/600 gel filtration column pre-equilibrated in buffer C (20 mM HEPES pH 7.5, 150 mM NaCl). The fractions containing 6xHis-SUMO-PexRD54 were pooled and concentrated to 2–3 mg/mL. The 6xHis-SUMO tag was cleaved by addition of 3C protease (10 µg/mg fusion protein) and incubated at 4°C overnight. Cleaved PexRD54 was further purified using Ni2+-NTA column (collecting eluate) followed by gel filtration. The purified protein was concentrated as appropriate, and the final concentration was judged by absorbance at 280 nm (using a calculated molar extinction coefficient of PexRD54, 57,040 M-1cm-1).
The PexRD54AIM2 variant was amplified from RFP:RD54AIM2 (using primers shown in Supplementary file 3) and cloned into the vector pOPINS3C as above. Recombinant protein was expressed and purified as for wild-type PexRD54; with protein concentration measured using a calculated molar extinction coefficient of 51,350 M-1cm-1.
Cloning, expression and purification of ATG8CL and ATG8IL
Request a detailed protocolDNA encoding Met1 to Ser119 of ATG8CL and Gly2 to Ser119 of ATG8IL were amplified from GFP:ATG8CL and GFP:ATG8IL (using primers shown in Supplementary file 3) and cloned into the vector pOPINF, generating a cleavable N-terminal 6xHis-tag with ATG8CL and ATG8IL. Recombinant proteins were produced using E. coli strain BL21 (DE3) grown in lysogeny broth at 37°C to an A600 of 0.4–0.6 followed by induction with 1 mM IPTG and overnight incubation at 18°C. Pelleted cells were resuspended in buffer A and pure, concentrated protein prepared as described for PexRD54 above (concentration determined using a calculated molar extinction coefficient of 7680 M-1cm-1 and 9080 M-1cm-1 for ATG8CL and ATG8IL, respectively).
Isothermal titration calorimetry (ITC)
Request a detailed protocolCalorimetry experiments were carried out at 15°C in 20 mM HEPES pH 7.5, 500 mM NaCl, using an iTC200 instrument (MicroCal Inc.). For protein:protein interactions, the calorimetric cell was filled with 100 μM PexRD54 and titrated with 1.1 mM ATG8CL or ATG8IL from the syringe. A single injection of 0.5 μl of ATG8CL or ATG8IL was followed by 19 injections of 2 μl each. Injections were made at 150 s intervals with a stirring speed of 750 rpm. For the heats of dilution control experiments, equivalent volumes of ATG8CL or ATG8IL were injected into buffer using the parameters above. For protein:peptide interactions, the calorimetric cell was filled with 90 μM ATG8CL or ATG8IL and titrated with 1 mM peptide from the syringe. The titrations were performed at 25°C, but otherwise as above. The raw titration data were integrated and fitted to a one-site binding model using the MicroCal Origin software.
Accession numbers
Request a detailed protocolPexRD54 (PITG_09316); StATG8CL (PGSC0003DMP400038670), StATG8IL (PGSC0003DMP400009229), SlATG8CL (Solyc10g006270 and Solyc07g064680), SlATG8IL (Solyc01g068060), NbATG8CL (Nb_S00003316g0005, KR021366), NbATG8IL (Nb_S00005942g0011, KR021365), and Joka2 (XM_006344410).
Data availability
-
Phytophthora infestans T30-4 secreted RxLR effector peptide protein, putative (PITG_09316) mRNA, complete cdsPublicly available at NCBI Nucleotide (Accession no: XM_002903553.1).
-
Solanum tuberosum polypeptide PGSC0003DMP400038670Publicly available at SolGenomics Database (Accession no: PGSC0003 DMP400038670).
-
Solanum tuberosum polypeptide PGSC0003DMP400009229Publicly available at SolGenomics Database (Accession no: PGSC0003 DMP400009229).
-
Tomato locus Solyc10g006270Publicly available at SolGenomics Database (Accession no: Solyc10g006270).
-
Tomato locus Solyc07g064680Publicly available at SolGenomics Database (Accession no: Solyc07g064680).
-
Tomato locus Solyc01g068060Publicly available at SolGenomics Database (Accession no: Solyc01g068060).
-
An effector of the Irish potato famine pathogen antagonizes a host autophagy cargo receptorPublicly available at NCBI (Accession no: KR021366).
-
An effector of the Irish potato famine pathogen antagonizes a host autophagy cargo receptorPublicly available at NCBI (Accession no: KR021365).
-
PREDICTED: Solanum tuberosum protein NBR1 homolog (LOC102582603), transcript variant X2, mRNAPublicly available at NCBI nucleotide (Accession no: XM_006344410.2).
-
Data from: An effector of the Irish potato famine pathogen antagonizes a host autophagy cargo receptorAvailable at Dryad Digital Repository under a CC0 Public Domain Dedication.
References
-
Basic local alignment search toolJournal of Molecular Biology 215:403–410.https://doi.org/10.1016/S0022-2836(05)80360-2
-
The LIR motif - crucial for selective autophagyJournal of Cell Science 126:3237–3247.https://doi.org/10.1242/jcs.126128
-
The role of ‘eat-me’ signals and autophagy cargo receptors in innate immunityCurrent Opinion in Microbiology 16:339–348.https://doi.org/10.1016/j.mib.2013.03.010
-
Oomycetes, effectors, and all that jazzCurrent Opinion in Plant Biology 15:483–492.https://doi.org/10.1016/j.pbi.2012.03.008
-
Phytophthora infestans effector AVRblb2 prevents secretion of a plant immune protease at the haustorial interfaceProceedings of the National Academy of Sciences of the United States of America 108:20832–20837.https://doi.org/10.1073/pnas.1112708109
-
In vivo imaging and quantitative monitoring of autophagic flux in tobacco by-2 cellsPlant Signaling & Behavior 8:e22510.https://doi.org/10.4161/psb.22510
-
Regulation mechanisms and signaling pathways of autophagyAnnual Review of Genetics 43:67–93.https://doi.org/10.1146/annurev-genet-102808-114910
-
Bacteria–autophagy interplay: a battle for survivalNature Reviews Microbiology 12:101–114.https://doi.org/10.1038/nrmicro3160
-
Intracellular plant microbe associations: secretory pathways and the formation of perimicrobial compartmentsCurrent Opinion in Plant Biology 13:372–377.https://doi.org/10.1016/j.pbi.2010.04.005
-
GATEWAY vectors for agrobacterium-mediated plant transformationTrends in Plant Science 7:193–195.https://doi.org/10.1016/S1360-1385(02)02251-3
-
MEGA2: molecular evolutionary genetics analysis softwareBioinformatics 17:1244–1245.https://doi.org/10.1093/bioinformatics/17.12.1244
-
The autophagosome: origins unknown, biogenesis complexNature Reviews Molecular Cell Biology 14:759–774.https://doi.org/10.1038/nrm3696
-
Clustal w and clustal x version 2.0Bioinformatics 23:2947–2948.https://doi.org/10.1093/bioinformatics/btm404
-
Autophagy: a multifaceted intracellular system for bulk and selective recyclingTrends in Plant Science 17:526–537.https://doi.org/10.1016/j.tplants.2012.05.006
-
Dynamic cellular responses in plant–microbe interactionsCurrent Opinion in Plant Biology 8:625–631.https://doi.org/10.1016/j.pbi.2005.09.006
-
Virus-induced gene silencing in tomatoThe Plant Journal 31:777–786.https://doi.org/10.1046/j.1365-313X.2002.01394.x
-
RXLR effectors of plant pathogenic oomycetesCurrent Opinion in Microbiology 10:332–338.https://doi.org/10.1016/j.mib.2007.04.005
-
Autophagy and bacterial clearance: a not so clear pictureCellular Microbiology 15:395–402.https://doi.org/10.1111/cmi.12063
-
Candidate effector proteins of the rust pathogen melampsora larici-populina target diverse plant cell compartmentsMolecular Plant-Microbe Interactions 28:689–700.https://doi.org/10.1094/MPMI-01-15-0003-R
-
Self and nonself: how autophagy targets mitochondria and bacteriaCell Host & Microbe 15:403–411.https://doi.org/10.1016/j.chom.2014.03.012
-
Apoplastic effectors secreted by two unrelated eukaryotic plant pathogens target the tomato defense protease Rcr3Proceedings of the National Academy of Sciences of the United States of America 106:1654–1659.https://doi.org/10.1073/pnas.0809201106
-
Cargo recognition and trafficking in selective autophagyNature Cell Biology 16:495–501.https://doi.org/10.1038/ncb2979
-
Membrane trafficking and autophagy in pathogen-triggered cell death and immunityJournal of Experimental Botany 65:1297–1312.https://doi.org/10.1093/jxb/ert441
Article and author information
Author details
Funding
European Research Council
- Abbas Maqbool
- Benjamin Petre
- Joe Win
- Sophien Kamoun
Gatsby Charitable Foundation
- Yasin F Dagdas
- Khaoula Belhaj
- Neftaly Cruz-Mireles
- Jan Sklenar
- Joe Win
- Frank Menke
- Sophien Kamoun
Biotechnology and Biological Sciences Research Council
- Pooja Pandey
- Nadra Tabassum
- Tolga O Bozkurt
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Acknowledgements
We thank Elaine Barclay, Liliana M. Cano, Frédéric Fragnière, Marina Franceschetti, Artemis Giannakopoulou, Ricardo Oliva, Egem Ozbudak, Stephen Whisson for technical support and/or providing materials. We are grateful to Zlay Taftacs and all members of the Kamoun Lab for helpful suggestions. This project was funded by the Gatsby Charitable Foundation, European Research Council (ERC), Biotechnology Biological Sciences Research Council (BBSRC) and the John Innes Foundation.
Copyright
© 2016, Dagdas et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 11,306
- views
-
- 2,369
- downloads
-
- 201
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Citations by DOI
-
- 201
- citations for umbrella DOI https://doi.org/10.7554/eLife.10856
Download links
Downloads (link to download the article as PDF)
Open citations (links to open the citations from this article in various online reference manager services)
Cite this article (links to download the citations from this article in formats compatible with various reference manager tools)
Further reading
-
- Microbiology and Infectious Disease
- Plant Biology
Pathogens target proteins involved in autophagy to inhibit immune responses in plants.






