Neuroprotective effects of TRPA1 channels in the cerebral endothelium following ischemic stroke
Figures
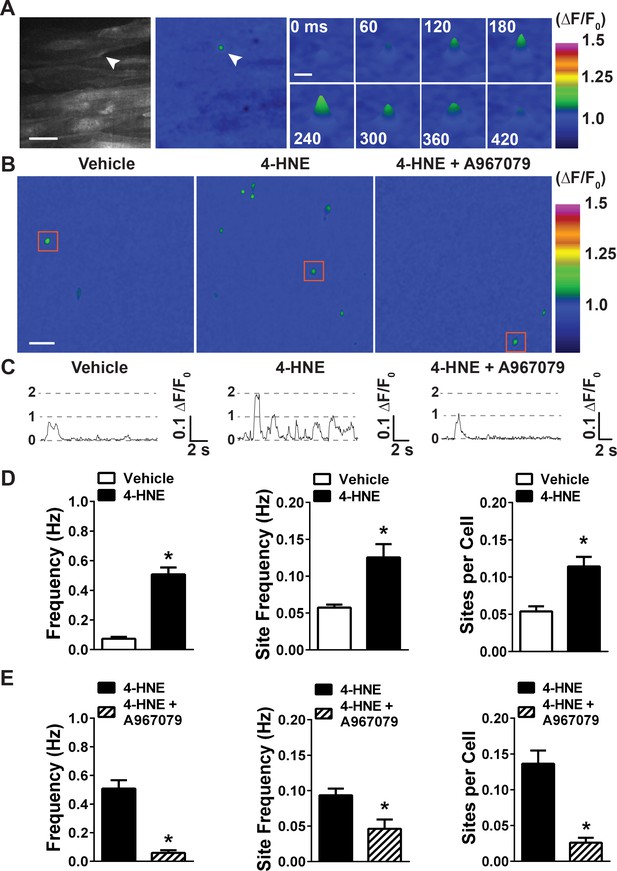
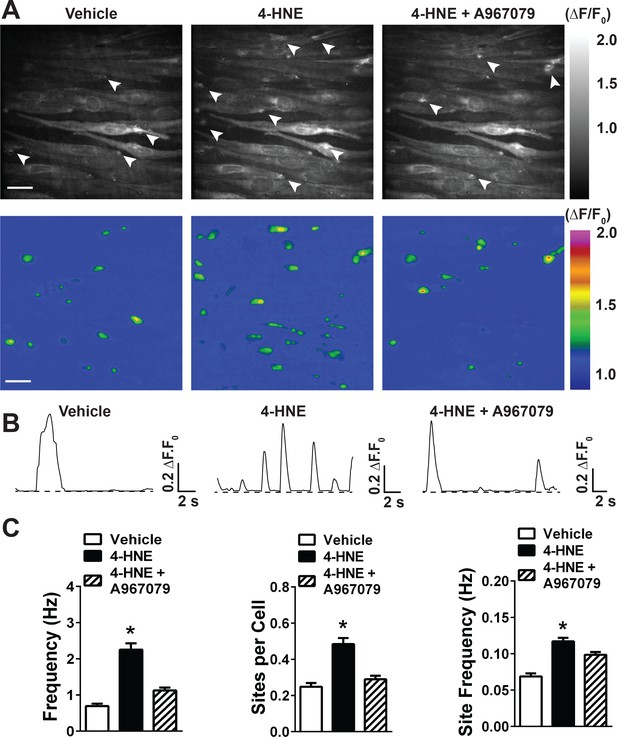
4-HNE stimulates TRPA1 sparklets in the endothelium of intact cerebral arteries.
(A) Representative images of endothelial cells from cerebral arteries from Tek:Gcamp6f mice mounted en face, presented in grayscale (left) and pseudocolored (middle and right panels). Scale bar = 15 µm. The images on the right are a timelapse and digital magnification of the TRPA1 sparklet indicated by the arrow in the right and middle panels. Scale bar = 5 µm. (B) Representative pseudocolored images of a 512 × 512 pixels field of view from Tek:Gcamp6f mice showing sparklets (green) after exposure to vehicle, 4-HNE and 4-HNE + A967079. Scale bar = 20 µm. (C) Representative ΔF/F0 vs. time plots for a single sparklet site. 0, 1, two levels indicate hypothesized numbers of TRPA1 channels engaged during each signal. (D) Summary data showing the effects of the TRPA1 channel activator 4-HNE (1 µM) on TRPA1 sparklet frequency, site frequency and number of sites per cell (n = 24 fields of view from four different arteries, N = 4 mice). (E) TRPA1 inhibition with A967079 (1 µM) significantly prevented 4-HNE induction of TRPA1 sparklets in endothelial cells (20 fields of view from five different arteries; N = 3 mice). Data are presented as means ± SEM (*p<0.05, Student’s t-test). TRPA1 sparklets were recorded in the presence of the cell permeable Ca2+ chelator EGTA-AM (10 µM) and the sarcoendoplasmic reticulum Ca2+-ATPase inhibitor cyclopiazonic acid (CPA, 30 µM).
-
Figure 1—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 1.
- https://doi.org/10.7554/eLife.35316.010
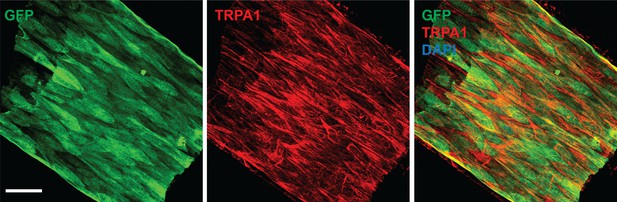
TRPA1 is present in the endothelium of cerebral arteries.
(A) Representative maximum intensity projection laser scanning confocal images showing immunolabeling of TRPA1 channels (red) in EGFP-expressing endothelial cells (green) of a pial artery in the brain of a perfusion-fixed Tekegp mouse. Nuclei of cells were stained by DAPI (blue). Scale bar = 40 µm.
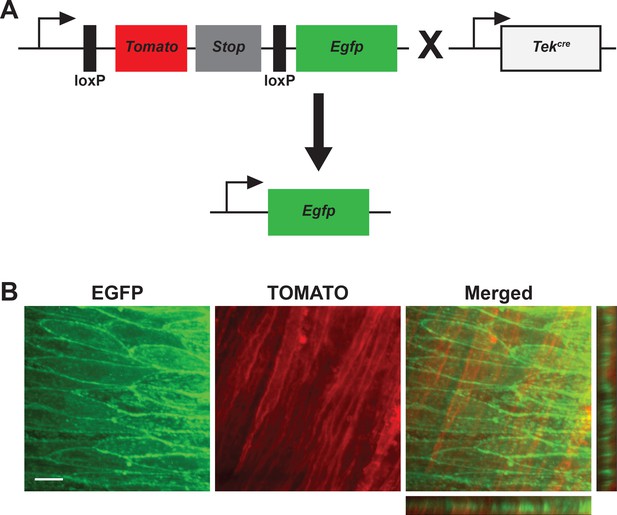
Reporter expression of cre-recombinase using the mT/mG reporter mice.
(A) Diagram of the genetic construct of the mT/mG reporter mice. Upon crossing with a mouse line expressing cre-recombinase, the sequence for Tomato and the Stop codon are excised, and Egfp is expressed. (B) Representative image of an en face cerebral artery from Tek:mT/mG showing EGFP expression (green) in the endothelial cell layer and TOMATO (red) in the underlying smooth muscle layer. The orthogonal sections on the right show that there is no overlay between green and red fluorescence. Scale bar = 20 µm.
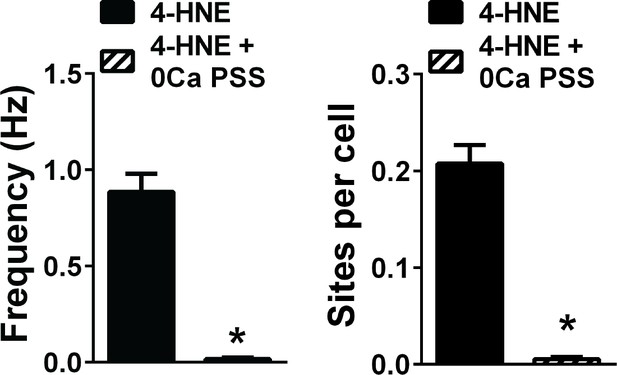
Extracellular Ca2+is required for TRPA1 sparklets.
Removal of extracellular Ca2+ significantly reduces TRPA1 sparklet frequency and number of sparklet sites induced by 4-HNE. *p<0.05, Student’s t-test. n = 36 fields of view from three different Tek:Gcamp6f mice. TRPA1 sparklets were recorded in the presence of the cell permeable Ca2+ chelator EGTA-AM (10 µM) and the sarcoendoplasmic reticulum Ca2+-ATPase inhibitor cyclopiazonic acid (CPA, 30 µM).
-
Figure 1—figure supplement 3—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 1—figure supplement 2.
- https://doi.org/10.7554/eLife.35316.007
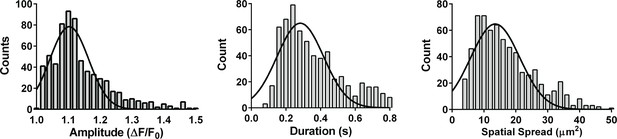
Properties of 4-HNE-induced TRPA1 sparklets.
Frequency distribution plots of 4-hydroxynonenal (4-HNE)-induced TRPA1 sparklets in en face cerebral arteries from Tek:Gcamp6f mice. Recorded events showed a mode amplitude of 1.10 ΔF/F0, a mode duration of 240 ms and a mode spatial spread of 10 µm2. A total of 670 events were plotted to analyze frequency distribution. TRPA1 sparklets were recorded in the presence EGTA-AM (10 µM) and CPA (30 µM).
-
Figure 1—figure supplement 4—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 1—figure supplement 3.
- https://doi.org/10.7554/eLife.35316.009
4-HNE stimulates large Ca2+transients in the endothelium of intact cerebral arteries.
(A) Representative grayscale images of endothelial cells from Tek:Gcamp6f mice mounted en face in the absence of CPA and EGTA-AM. The white arrowheads point to sites of Ca2+ transients. Scale bar = 30 µm. Pseudocolored representations of the images are shown below. Scale bar = 30 µm. (B) Representative ΔF/F0 vs. time plots of Ca2+ transients from a single site of Ca2+ transients. (C) Summary graphs showing that 4-HNE (1 µM) significantly increases frequency, number of active sites, and site frequency of Ca2+ transients in cerebral artery endothelial cells. This response was diminished by the TRPA1 blocker A967079 (1 µM). (25 fields of view from three different arteries; N = 3 mice). Data are presented as means ± SEM (*p<0.05, one-way ANOVA).
-
Figure 2—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 2.
- https://doi.org/10.7554/eLife.35316.018
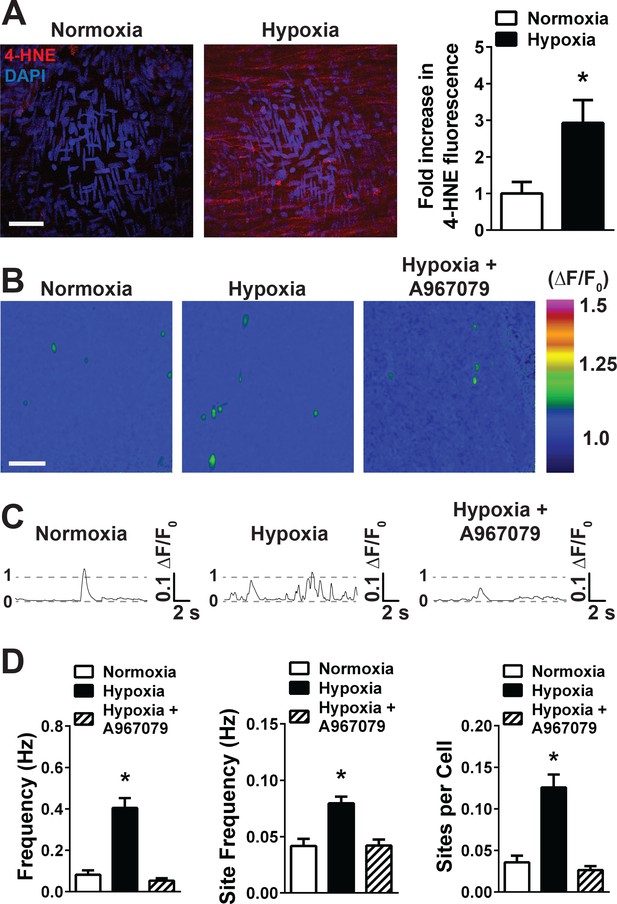
Acute hypoxia increases 4-HNE accumulation and increases TRPA1 sparklet frequency in the cerebral endothelium.
(A) Representative maximum intensity projection of Z-stacks of cerebral arteries mounted en face and exposed to PSS equilibrated with a normoxic (21% O2, 6% CO2, 73% N2, left panel) or hypoxic gas mixture (5% O2, 6% CO2, 89% N2, right panel) and immunolabeled for 4-HNE (red). Scale bar = 40 µm, nuclei of cells are labeled by DAPI (blue). Acute hypoxia significantly increased 4-HNE immunoreactivity in cerebral arteries (*p<0.05 Student’s t-test, N = 10–9 fields of view from three different experiments). (B) Representative pseudocolored images of cerebral arteries from Tek:Gcamp6f mice mounted en face and exposed to normoxic (left panel) or hypoxic (middle and right panels) PSS in the presence or absence of the selective TRPA1 blocker A967079 (1 µM). Green: active TRPA1 sparklet sites. Scale bar = 20 µm. (C) Representative ΔF/F0 vs. time plots for a single sparklet site showing an increase in TRPA1 sparklet frequency during hypoxia which was significantly inhibited by the TRPA1 blocker A967079. (D) Summary data showing the effects of hypoxia on TRPA1 sparklet frequency (left), site frequency (middle) and number of sites per cell (right) in the presence and absence of the selective TRPA1 inhibitor A967079 (1 µM). (*p<0.05, one-way ANOVA; N = 25–28 – 33 fields of view from six different arteries isolated from six mice). TRPA1 sparklets were recorded in the presence EGTA-AM (10 µM) and CPA (30 µM).
-
Figure 3—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 3.
- https://doi.org/10.7554/eLife.35316.027
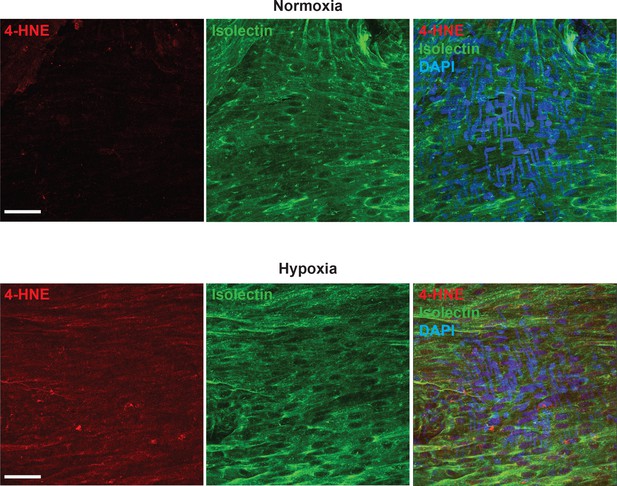
Hypoxia causes 4-HNE accumulation in cerebral arteries.
Representative maximum intensity projection images of en face cerebral arteries immunolabeled for 4-HNE (red) and stained with isolectin conjugated to AlexaFluor 488 (green) after superfused with normoxic (top images) or hypoxic (bottom panels) physiological saline solution (PSS). The panels on the right are overlay images showing nuclei staining (DAPI, blue). Images are representative of 3 different experiments from 10 basilar arteries. Scale bar = 40 μm.
-
Figure 3—figure supplement 1—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 3—figure supplement 1.
- https://doi.org/10.7554/eLife.35316.024
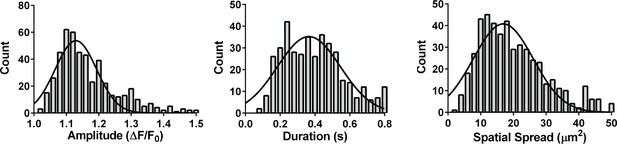
Properties of hypoxia-induced TRPA1 sparklets.
Frequency distribution plots of hypoxia-induced TRPA1 sparklet in en face cerebral arteries from Tek:Gcamp6f mice. Recorded events showed a mode peak amplitude of 1.10 ΔF/F0, a mode duration of 240 ms and a mode spatial spread of 12 µm2. A total of 437 events were plotted to analyze frequency distribution. TRPA1 sparklets were recorded in the presence of EGTA-AM (10 µM) and CPA (30 µM).
-
Figure 3—figure supplement 2—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 3—figure supplement 2.
- https://doi.org/10.7554/eLife.35316.026
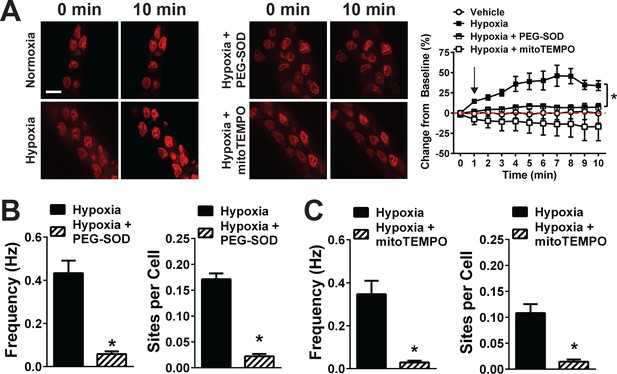
Acute hypoxia induces mitochondrial superoxide generation and increases TRPA1 sparklet frequency in the cerebral endothelium.
(A) Left and middle: Superoxide generation detected with the superoxide-sensitive dye DHE (red) in the endothelium of cerebral arteries mounted en face under normoxia (left), hypoxia (left), hypoxia plus the membrane-permeable PEG-SOD (100 U/ml, middle) and hypoxia plus the mitochondria-targeted SOD mimetic mitoTEMPO (500 nM, middle). Right: Summary data showing changes in DHE fluorescence over time under each condition (*p<0.05, two-way ANOVA; n = 5–7 – 6–7 arteries from five different mice). The arrow in the graph indicates the onset of hypoxia. Scale bar = 10 µm. (B and C) Summary data showing the effects of PEG-SOD (B) and mitoTEMPO (C) on the frequency and number of active TRPA1 sparklet sites in the cerebral artery endothelium of Tek:Gcamp6f mice (*p<0.05, Student’s t-test; n = 25–30 fields of view from six different preparations isolated from six different mice). TRPA1 sparklets were recorded in the presence of EGTA-AM (10 µM) and (CPA, 30 µM).
-
Figure 4—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 4.
- https://doi.org/10.7554/eLife.35316.033
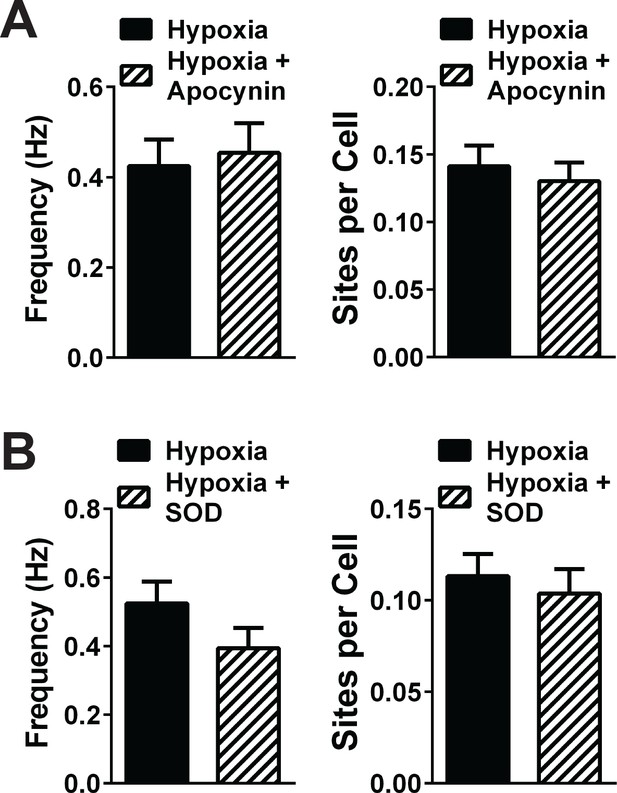
NOX2 inhibition and extracellular SOD did not significantly inhibit hypoxia-induced TRPA1 sparklets.
(A) Summary graph showing that sparklet frequency and number of TRPA1 sparklet sites induced by hypoxia are not significantly changed by apocynin. N = 25–30 fields of view from three different Tek:Gcamp6f mice. (B) Summary graph showing that TRPA1 sparklet frequency and number of sparklet sites induced by hypoxia are not significantly altered by incubation with extracellular SOD. N = 25–30 fields of view from three different Tek:Gcamp6f mice. TRPA1 sparklets were recorded in the presence of EGTA-AM (10 µM) and CPA (30 µM).
-
Figure 4—figure supplement 1—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 4—figure supplement 1.
- https://doi.org/10.7554/eLife.35316.032
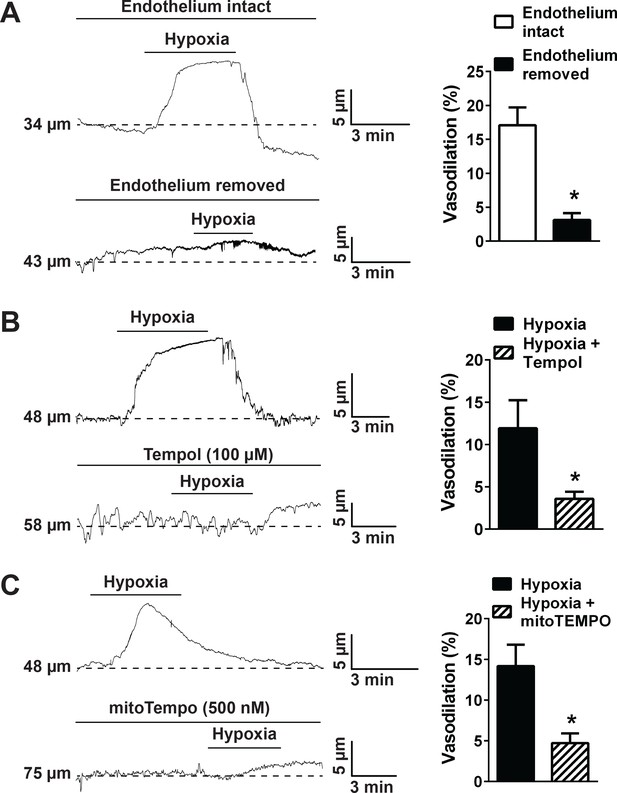
Hypoxia acts via mitochondrial superoxide to induce endothelium-dependent dilation of pressurized cerebral arteries.
(A) Representative traces (left) and summary data (right) showing hypoxia-induced dilation in intact and endothelium-denuded cerebral pial arteries (*p<0.05, Student’s t-test; n = 5 arteries from three different mice). (B) Representative traces of lumen diameter of a pressurized cerebral pial artery (left) and summary data (right) showing hypoxia-induced dilation in the presence of the cell-permeant SOD mimetic Tempol (100 µM). (*p<0.05, Student’s t-test; n = 5 arteries from three different mice.) (C) Representative traces of the lumen diameter of a pressurized cerebral pial artery (left) and summary data (right) showing hypoxia-induced dilation in the presence of the cell-permeant mitochondrial membrane-targeted SOD mimetic mitoTEMPO (500 nM). (*p<0.05, Student’s t-test; n = 6 arteries from three different mice). The arteries used for pressure myography experiments were not treated with EGTA-AM or CPA.
-
Figure 5—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 5.
- https://doi.org/10.7554/eLife.35316.039
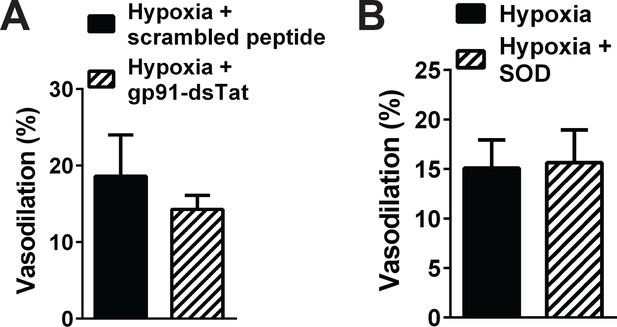
NOX2 inhibition and quenching of extracellular O2-and H2O2 did not significantly alter cerebral artery dilation induced by hypoxia.
Summary data showing that hypoxia-induced dilation of cerebral pial arteries was not significantly altered by NOX2 inhibition with gp91-dsTat (1 μM, (A), and by removal of extracellular O2 with SOD (200 U/mL, (B). n = 3 arteries from three different mice for the gp91-dsTat experiments, and n = 5 arteries from three different mice for the extracellular SOD experiments. Arteries used for pressure myography experiments were not incubated with EGTA-AM or CPA.
-
Figure 5—figure supplement 1—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 5—figure supplement 1.
- https://doi.org/10.7554/eLife.35316.036
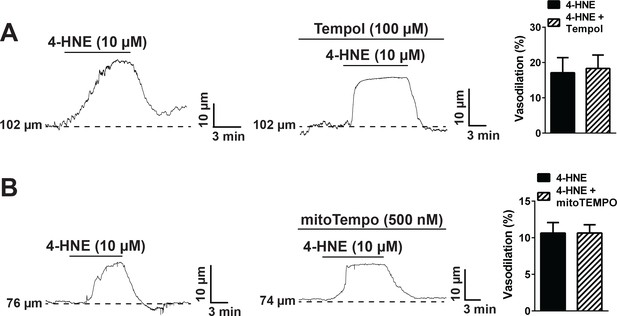
Superoxide dismutase mimetics do not directly inhibit TRPA1 channels.
(A) The cell permeable superoxide dismutase (SOD) mimetic Tempol (100 µM) did not alter dilation of pressurized cerebral arteries to the TRPA1 agonist 4-hydroxynonenal (4-HNE), as observed by the representative traces on the left and middle, and summary graph on the right. N = 3 arteries from three different mice. (B) Similarly, the mitochondria-targeted SOD mimetic mitoTEMPO (500 nM) did not affect TRPA1-mediated dilation caused by exposure to 4-HNE. N = 5 arteries from three different mice. Arteries used for pressure myography experiments were not incubated with EGTA-AM or CPA.
-
Figure 5—figure supplement 2—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 5—figure supplement 2.
- https://doi.org/10.7554/eLife.35316.038
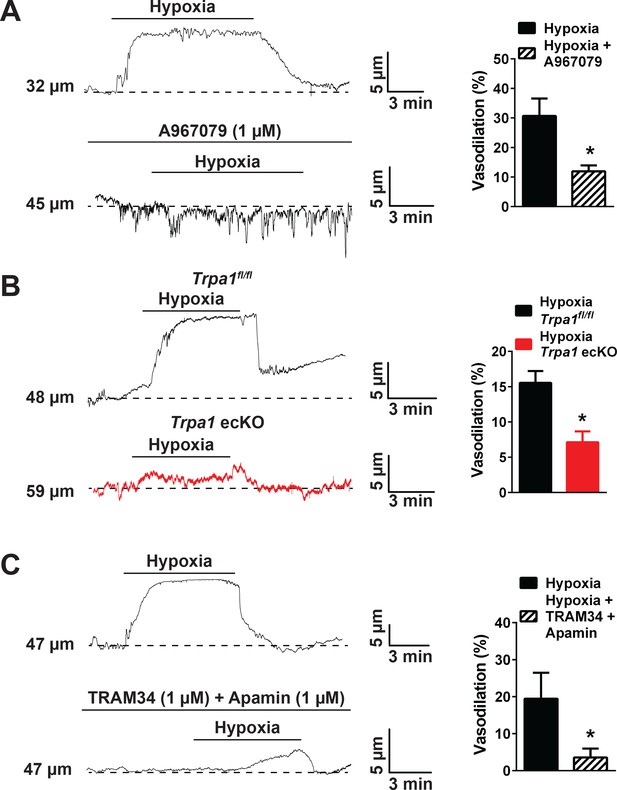
Hypoxia-induced cerebral artery dilation is dependent on endothelial TRPA1, KCa3.1 and KCa2.3 channels.
(A) Representative traces of luminal diameter of pressurized cerebral pial arteries (left) and summary data (right) showing hypoxia-induced dilation in the presence and absence of the selective TRPA1 inhibitor A967079 (*p<0.05, Student’s t-test; n = 5 arteries from three different mice). (B) Representative traces of the luminal diameter of pressurized cerebral pial arteries (left) and summary data (right) showing hypoxia-induced dilation in Trpa1 ecKO mice and wildtype littermates (Trpa1fl/fl) (*p<0.05, Student’s t-test; n = 7–6 arteries from three different mice). (C) Representative traces of the luminal diameter of pressurized cerebral pial arteries (left) and summary data (right) showing hypoxia-induced dilation in the presence of selective inhibitors of KCa3.1 (TRAM34, 1 µM) and KCa2.3 channels (apamin, 1 µM) (*p<0.05, Student’s t-test; n = 5 arteries from three different mice). The arteries used for pressure myography experiments were not treated with EGTA-AM or CPA.
-
Figure 6—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 6.
- https://doi.org/10.7554/eLife.35316.041
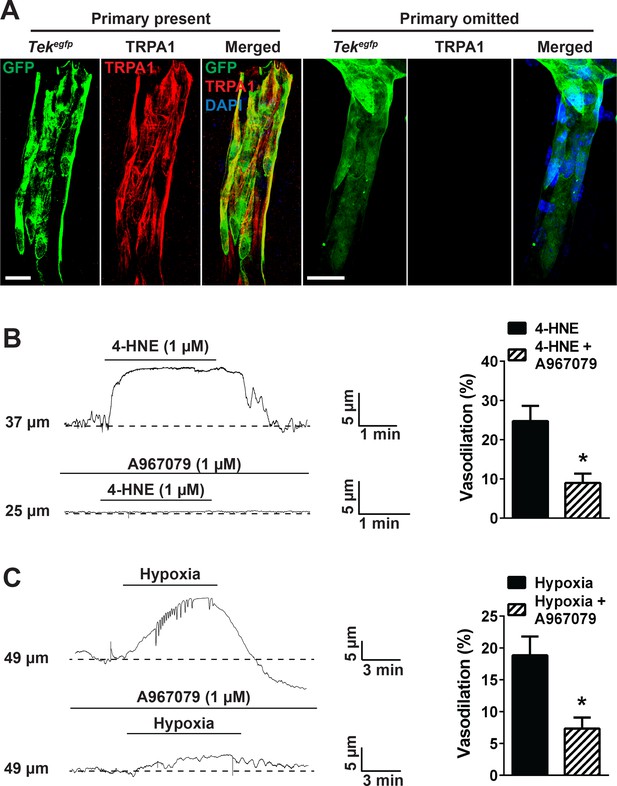
TRPA1 channel activity dilates cerebral penetrating arterioles.
(A) Representative maximum intensity projection laser scanning confocal images showing immunolabeling of TRPA1 channels (red) in EGFP-expressing endothelial cells (green) of penetrating arterioles in the brain (left panels, primary present). Scale bar = 20 µm. TRPA1 immunoreactivity was absent when the primary antibody was omitted (right panels). Nuclei of cells were stained by DAPI (blue). Scale bar = 20 µm. (B) Incubation of ex vivo pressurized penetrating arterioles with the TRPA1 agonist 4-HNE (1 µM) induced arteriolar dilation which was significantly diminished by A967079 (1 µM). Representative traces are shown on the left and the summary graph is shown on the right. (*p<0.05, Student’s t-test; n = 5 arteries from three different mice). (C) Hypoxia induced dilation of penetrating arterioles that was significantly blunted by A967079 (*p<0.05, Student’s t-test; n = 6 arteries from three different mice). The arterioles used for pressure myography experiments were not incubated with EGTA-AM or CPA.
-
Figure 7—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 7.
- https://doi.org/10.7554/eLife.35316.043
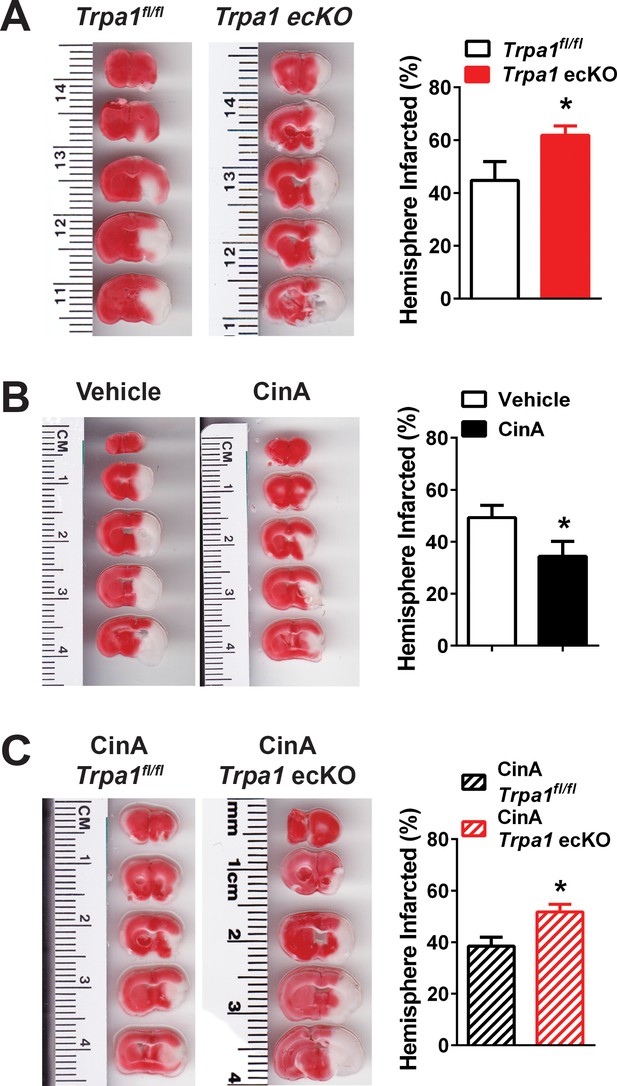
Endothelial cell TRPA1 channel activity protects against ischemic strokes.
(A) Representative photographs of brain slices (left) and summary data (right) showing significantly greater ischemic damage 24 hr after MCAO in Trpa1 ecKO mice compared with Trpa1fl/fl. Brain slices were stained with 2,3,5-triphenyltetrazolium chloride (TTC), which stains metabolically active tissue red, whereas infarcted tissue remains unstained (white). Infarcted areas were quantified and expressed as a percentage of total hemisphere area (*p<0.05, Student’s t-test; n = 5–5 mice). (B) Representative photographs of brain slices (left) and summary data (right) showing reduced cerebral ischemic damage in wildtype C57/bl6 mice treated with the TRPA1 channel activator cinnamaldehyde (CinA, 50 mg/kg i.p.), injected 15 min after MCAO (*p<0.05 for CinA vs. vehicle, Student’s t-test; n = 5–6 mice). (C) Representative photographs of brain slices (left) and summary data (right) showing that the protective effects of CinA were blunted in Trpa1 ecKO mice (*p<0.05 for CinA-treated Trpa1 ecKO mice vs. CinA-treated Trpa1fl/fl mice, Student’s t-test; n = 6–5 mice). Legends for Supplemental Figures.
-
Figure 8—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 8.
- https://doi.org/10.7554/eLife.35316.050
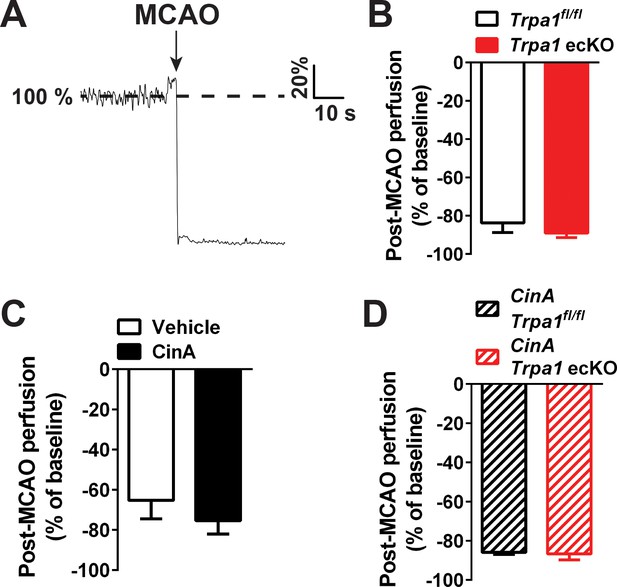
Post-MCAO perfusion is not significantly different between all experimental groups.
(A) Representative laser Doppler flowmetry trace showing a sharp reduction in perfusion of the middle cerebral artery vascular territory after MCAO. Perfusion data are shown as a % of pre-occlusion perfusion. (B–D) Laser Doppler flowmetry assessment of cerebral perfusion to the middle cerebral artery territory showed that the extent of MCA occlusion was not significantly different between mice from all treatments and genetic backgrounds.
-
Figure 8—figure supplement 1—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 8—figure supplement 1.
- https://doi.org/10.7554/eLife.35316.046
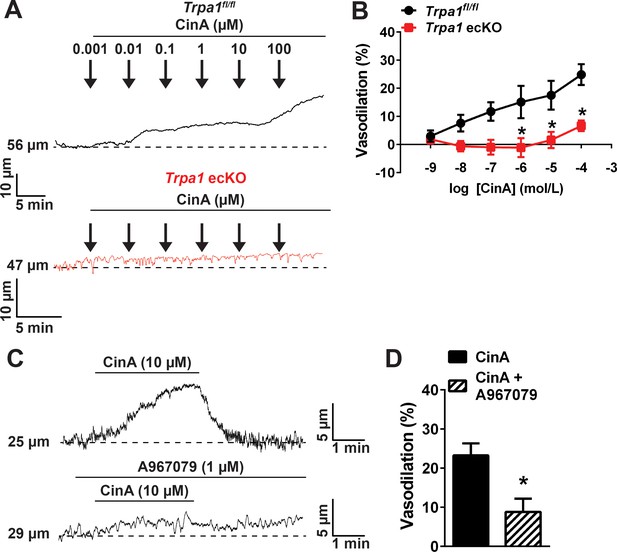
Cinnamaldehyde causes cerebral artery and arteriolar dilation by activating TRPA1 channels in endothelial cells.
(A) Representative traces of the lumen diameter of pressurized cerebral arteries incubated with increasing concentrations of CinA (1 nM to 100 µM) in Trpa1fl/fl (top trace, black) and Trpa1 ecKO mice (bottom trace, red). (B) Summary graph showing that CinA induces a concentration dependent dilation of cerebral arteries via TRPA1 channels located in endothelial cells. *p<0.05, n = 6–6 arteries from three different Trpa1fl/fl and three different Trpa1 ecKO mice. (C–D) CinA (30 µM) induces dilation of pressurized penetrating arterioles via TRPA1 channels, as evidenced by the representative traces (C) and the summary data (D). *p<0.05. n = 5 arterioles from three different mice. Arteries and arterioles used for pressure myography experiments were not incubated with EGTA-AM or CPA.
-
Figure 8—figure supplement 2—source data 1
Excel spreadsheet containing the individual numeric values of the parameters analyzed in Figure 8—figure supplement 2.
- https://doi.org/10.7554/eLife.35316.048
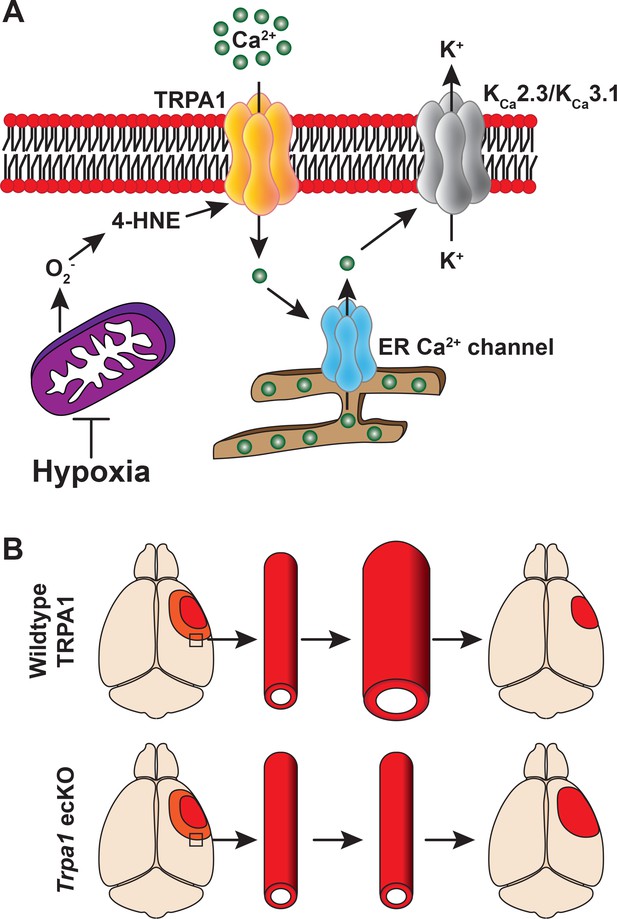
Mechanism of endothelial TRPA1 channel-mediated neuroprotection.
(A) Acute hypoxia induces a mitochondria-dependent increase in superoxide production, which leads to accumulation of the lipid peroxide 4-hydroxynonenal (4-HNE) in endothelial cells. 4-HNE is an endogenous activator of the TRPA1 channel, increasing the frequency of Ca2+ influx events – TRPA1 sparklets, which is amplified by Ca2+-induced Ca2+-release from the endoplasmic reticulum. The subcellular increase in intracellular Ca2+ activates KCa2.3 and KCa3.1, resulting in K+ efflux and vasodilation. (B) In vivo, endothelial cell TRPA1 channel activity may act as an adaptive mechanism to prevent infarct expansion (red) by promoting vasodilation in the ischemic penumbra (orange). Endothelial-cell specific deletion of Trpa1 removes this protection, ultimately leading to an increase in infarct volume following an ischemic stroke.
Videos
Spontaneous Ca2+transients in the endothelium of a cerebral artery.
Representative movie of a cerebral artery from a Tek:Gcamp6f mouse mounted en face and superfused with normoxic (21% O2, 6% CO2, 73% N2), warm (37°C) physiological saline solution (PSS) without EGTA-AM or CPA.
Ca2+events in the endothelium of a mesenteric artery from a Tek:Gcamp6f mouse.
Representative movie of a mesenteric artery from a Tek:Gcamp6f mouse mounted en face and superfused with normoxic (21% O2, 6% CO2, 73% N2), warm (37°C) physiological saline solution (PSS) without EGTA-AM or CPA.
Ca2+events in the endothelium of a skeletal muscle artery from a Tek:Gcamp6f mouse.
Representative movie of an skeletal muscle artery from a Tek:Gcamp6f mouse mounted en face and superfused with normoxic (21% O2, 6% CO2, 73% N2), warm (37°C) physiological saline solution (PSS) without EGTA-AM or CPA.
Ca2+events in the pulmonary endothelium from a Tek:Gcamp6f mouse.
Representative movie of a pulmonary artery from a Tek:Gcamp6f mouse mounted en face and superfused with normoxic (21% O2, 6% CO2, 73% N2), warm (37°C) physiological saline solution (PSS) without EGTA-AM or CPA.
Spontaneous Ca2+influx events the cerebral endothelium.
Representative movie of a cerebral artery isolated from a Tek:Gcamp6f mouse mounted en face and superfused with normoxic (21% O2, 6% CO2, 73% N2), warm (37°C) physiological saline solution (PSS) in the presence of EGTA-AM (10 µM) and CPA (30 µM).
4-HNE stimulates TRPA1 sparklets the cerebral artery endothelium.
Representative movie of the same field of view as in Video 5 showing that application of 4-HNE (1 μM) significantly increased the frequency of TRPA1 sparklets in the cerebral endothelium. TRPA1 sparklets were recorded in the presence of EGTA-AM (10 µM) and CPA (30 µM).
Spontaneous large Ca2+transients in the endothelium of a cerebral artery.
Representative movie of a cerebral artery from a Tek:Gcamp6f mouse mounted en face and superfused with normoxic (21% O2, 6% CO2, 73% N2), warm (37°C) physiological saline solution (PSS) without EGTA-AM or CPA.
4-HNE increases the frequency of Ca2+transients in the cerebral artery endothelium.
Representative movie of the same field of view as in Video 7 showing that 4-HNE (1 µM) greatly increases the frequency of Ca2+ transients in the endothelium. Events were recorded in the absence of EGTA-AM or CPA (30 µM).
TRPA1 channels mediate the increase in Ca2+transients caused by 4-HNE.
Representative movie of the same field of view as in Videos 7 and 8 showing that TRPA1 inhibition with A967079 (1 µM) prevents the increase in Ca2+ transients frequency elicited by 4-HNE (1 µM) in the endothelium. TRPA1 sparklets were recorded in the presence of EGTA-AM (10 µM) and CPA (30 µM).
Spontaneous Ca2+influx events the cerebral endothelium during normoxia.
Representative movie of a cerebral artery isolated from a Tek:Gcamp6f mouse mounted en face and superfused with normoxic (21% O2, 6% CO2, 73% N2), warm (37°C) physiological saline solution (PSS) in the presence of EGTA-AM (10 µM) and CPA (30 µM).
Hypoxia stimulates TRPA1 sparklet frequency in the cerebral endothelium.
Representative movie of the same field of view from Video 10 showing that superfusing the preparation with hypoxic (5% O2, 6% CO2, 89% N2), warm (37°C) physiological saline solution (PSS) increases the frequency and number of active sites of TRPA1 sparklets. TRPA1 sparklets were recorded in the presence of EGTA-AM (10 µM) and CPA (30 µM).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| strain, strain background (Mus musculus), C57bl6 | Tekcre | Jackson Laboratories; PMID: 11161575 | stock # 008863 | |
| strain, strain background (Mus musculus), C57bl6 | mT/mG | Jackson Laboratories; PMID: 17868096 | stock # 007676 | |
| strain, strain background (Mus musculus), C57bl6 | Tekgfp | Jackson Laboratories; PMID: 11064424 | stock # 003658 | |
| strain, strain background (Mus musculus), C57bl6 | Gcamp6f | Jackson Laboratories; PMID: 25741722 | stock # 028865 | |
| strain, strain background (Mus musculus), C57bl6 | Trpa1 ecKO | Other; PMID: 25564678 | NA | Mice with endothelium-specific deletion of Trpa1. We have characterized these mice in a previous publication (PMID: 25564678). |
| strain, strain background (Mus musculus), C57bl6 | Tek:Gcamp6f | This paper | NA | Mice expressing the fast kinetics, genetically encoded Ca2+ biosensor Gcamp6f exclusively in endothelial cells. |
| antibody | anti-4-HNE (rabbit polyclonal) | Abcam | ab46545 | 1:1000 dilution |
| antibody | anti-TRPA1 (rabbit polyclonal) | Alomone Labs; UniProtKB - O75762 (TRPA1_HUMAN) | ACC-037 | 1:1000 dilution |
| antibody | anti-GFP (goat polyclonal) | Abcam; UniProtKB - P42212 (GFP_AEQVI) | ab5450 | 1:500 dilution |
| antibody | Alexa 488 or 594 secondaries | ThermoFisher Scientific | A11055 (Alexa 488); A21207 (Alexa 594) | 1:1000 (Alexa 488); 1:2000 (Alexa 594) |
| other | Isolectin GS-IB4 conjugated to Alexa 488 | ThermoFisher Scientific | I21411 | 1:1000 dilution |
| other | dyhydroethidium | ThermoFisher Scientific | D11347 | |
| other | Fluoroshied mounting medium with DAPI | Abcam | ab104139 | |
| software, algorithm | SparkAn | Dr. Adrian Bonev and Dr. Mark Nelson; PMID: 22095728 | NA | Software to analyze Ca2+ events. Kindly provided by Dr. Adrian Bonev and Dr. Mark Nelson from the University of Vermont. |
Additional files
-
Supplementary file 1
Table 1.
- https://doi.org/10.7554/eLife.35316.051
-
Supplementary file 2
Table 2.
- https://doi.org/10.7554/eLife.35316.052
-
Transparent reporting form
- https://doi.org/10.7554/eLife.35316.053





