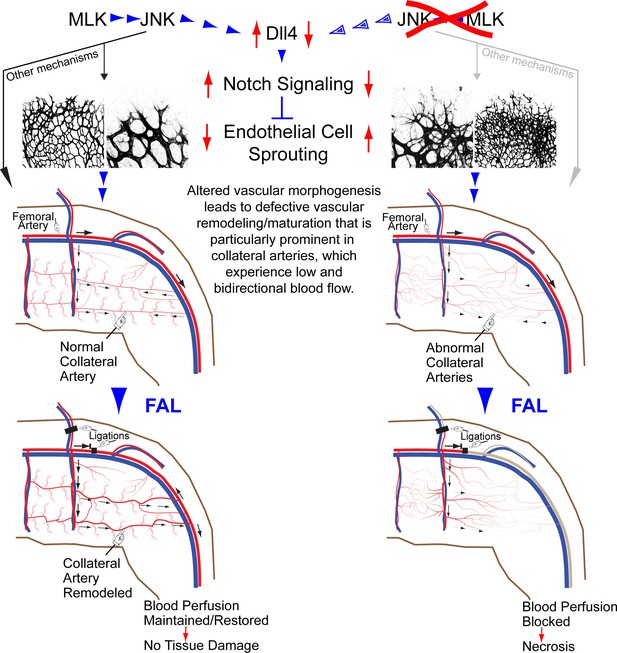
Suppression of ischemia in arterial occlusive disease by JNK-promoted native collateral artery development
Figures
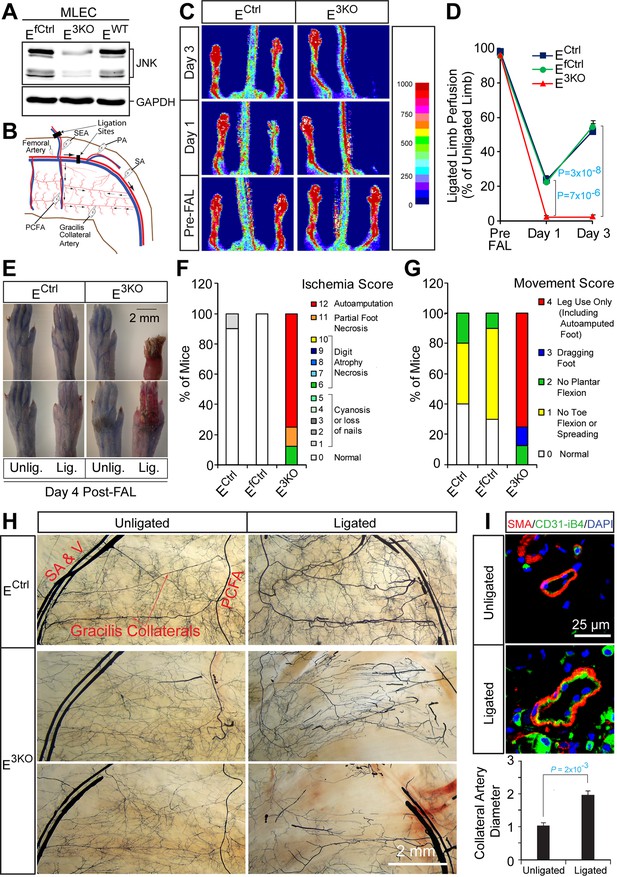
Enhanced blood perfusion blockade and severe ischemic injury in endothelial JNK-deficient mice upon arterial occlusion.
(A) Control and JNK-deficient primary endothelial cells were examined by immunoblot analysis by probing with antibodies to JNK and GAPDH. (B) Simplified diagram of the medial aspect of the mouse hindlimb skeletal muscle vasculature. The common femoral artery (FA) and its main branches (proximal caudal femoral artery [PCFA], popliteal artery [PA] and saphenous artery [SA]) supply blood to the proximal and distal hindlimb. Ligation of the FA plus the superficial epigastric artery (SEA) as indicated, leads to reduced blood flow to the distal hindlimb, while flow through the PCFA and gracilis collaterals is enhanced. (C) Representative laser Doppler images showing blood perfusion (high perfusion red, no perfusion dark blue) in the hindlimbs of control and JNK-deficient mice prior to unilateral FA ligation (Pre-FAL) and on day 1 and 3 post-FAL. (D) Quantitation of hindlimb blood flow shows significantly enhanced blood perfusion blockade following FAL and no recovery 3 days after ligation in JNK-deficient mice compared to control mice (mean ± SEM; n = 7~10). (E) Representative images of mouse paws on Day 4 post-FAL. Lig., ligated; Unlig., contralateral unligated. (F,G) Quantitation of ischemic (F) and movement (G) scores for mice on Day 4 post-FAL (7~10 mice per group). (H) Representative whole mount preparations of the medial surface of Microfil-filled adductor muscle vasculature from day 4 post-FAL hindlimbs and contralateral unligated limbs. (I) The gracilis collateral arteries were stained for SMA and CD31/iB4. The artery diameter was quantitated (mean ± SEM; n = 10~12). Source data are included as Figure 1—source data 1.
-
Figure 1—source data 1
Source data for Figure 1.
This file contains raw source data used to make the graphs presented in Figure 1.
- https://doi.org/10.7554/eLife.18414.004
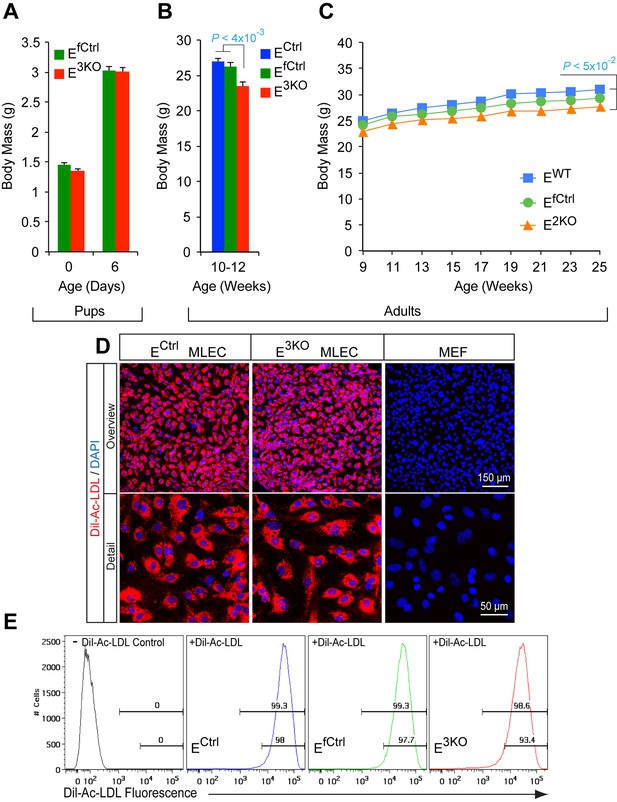
Characterization of endothelial JNK-deficient mice and lung endothelial cells.
(A) The body mass of endothelial JNK-deficient mice and control mice was examined at P0 and P6 (mean ± SEM; n = 12~23). No statistically significant differences in body mass between groups were identified. (B,C) The body mass of adult endothelial JNK-deficient mice and control mice was examined (mean ± SEM; n = 10 (B) and n = 7~10 [C]). The endothelial JNK-deficient mice were found to have a small, but statistically significant, decrease in body mass compared with control mice. (D,E) Primary murine endothelial cells (MLEC) and fibroblasts (MEF) were incubated (4 hr) with Dil-labeled acetylated low density lipoprotein (Dil-Ac-LDL, red), washed, fixed, stained with DAPI (blue), and examined by fluorescence microscopy (D) and flow cytometry (E). Source data are included as Figure 1—figure supplement 1—source data 1.
-
Figure 1—figure supplement 1—source data 1
Source data for Figure 1—figure supplement 1.
This file contains raw source data used to make the graphs presented in Figure 1—figure supplement 1.
- https://doi.org/10.7554/eLife.18414.006
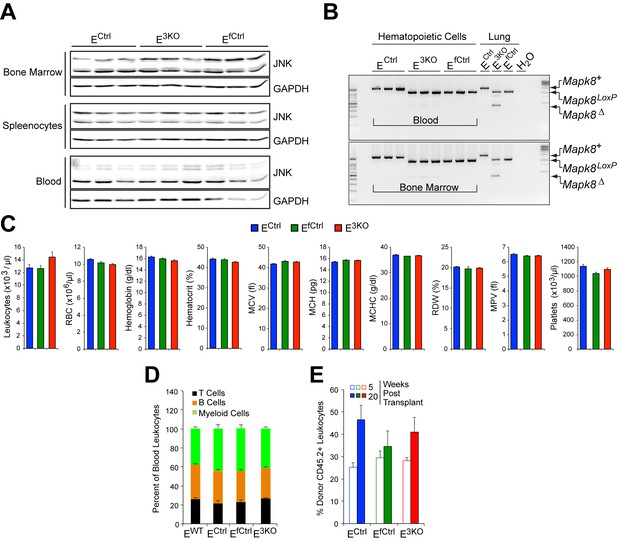
Endothelial JNK-deficient mice have no major perturbations in the hematopoietic system.
(A) Bone marrow, splenocytes, and blood cells isolated from endothelial JNK-deficient mice and control mice were examined by immunoblot analysis by probing with antibodies to JNK and GAPDH. The data presented are representative of 2 independent experiments (n = 5 mice). (B) Genomic DNA isolated from blood, bone marrow, and lung tissue was examined by PCR analysis to detect Cre-mediated recombination of the Mapk8 gene. The data presented are representative of 3 independent experiments (n = 3 mice). (C) Blood cell analysis demonstrated no significant differences (p>0.05) between endothelial JNK-deficient mice and control mice (mean ± SEM; n = 15). RBC, red blood cells; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; RDW, red cell distribution width; MPV, mean platelet volume. (D) Flow cytometry demonstrated no significant differences (p>0.05) in the frequency of myeloid cells (CD11b+), B cells (CD19+) and T cells (CD3e+) in the blood of control and endothelial JNK-deficient mice (mean ± SEM; n = 8~10). (E) Flow cytometry analysis of peripheral blood demonstrated no significant differences in chimerism at 5 and 20 weeks post-transplantation between mice transplanted with bone marrow cells from control and endothelial JNK-deficient mice (mean ± SEM; n = 7~8). Source data are included as Figure 1—figure supplement 2—source data 1.
-
Figure 1—figure supplement 2—source data 1
Source data for Figure 1—figure supplement 2.
This file contains raw source data used to make the graphs presented in Figure 1—figure supplement 2.
- https://doi.org/10.7554/eLife.18414.008
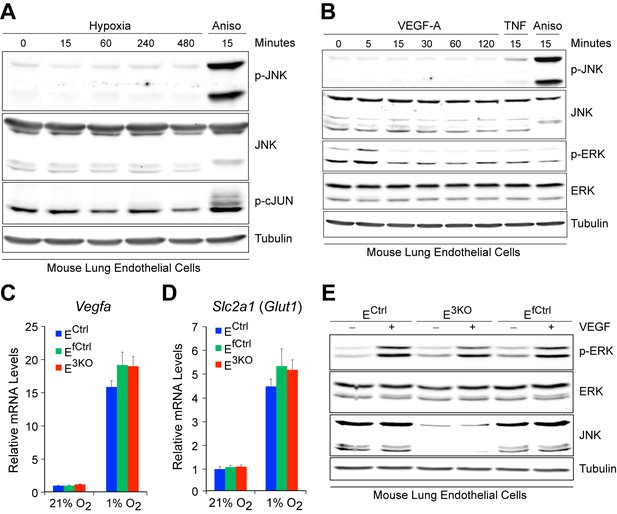
Normal hypoxia responses and VEGF signaling in JNK-deficient endothelial cells.
(A) Primary MLEC incubated overnight in media with 1% FBS were placed under hypoxia (1% O2). The cells were examined by immunoblot analysis with antibodies to pJNK, JNK, pSer63-cJun, and αTubulin. No change in the phosphorylation of JNK or its substrate cJun was detected. In contrast, anisomycin (Aniso, 1 μg/ml) treatment caused phosphorylation of both JNK and cJun. The data presented are representative of two independent experiments. (B) Primary MLEC incubated overnight in media with 1% FBS were treated with VEGFa (100 ng/ml, added directly to existing media). The cells were examined by immunoblot analysis with antibodies to pJNK, JNK, pERK, ERK and αTubulin. VEGF-A treatment caused phosphorylation of ERK at 5 min, but not JNK. In contrast, TNFα (20 ng/ml) and Anisomycin (Aniso, 1 μg/ml) treatment caused JNK phosphorylation. The data presented are representative of two independent experiments. (C,D) Endothelial cells in media containing only 1% FBS were incubated under normoxic (21% O2) or hypoxic (1% O2) conditions for 16 hr. The mRNA expression of the hypoxia responsive genes Vegfa (C) and Slc2a1 (Glut1) (D) was examined by quantitative RT-PCR analysis (mean ± SEM; n = 4). Data presented are from one of at least 2 similar experiments with independent endothelial cell preparations. (E) Endothelial cells incubated overnight in media containing only 1% FBS were treated with VEGF-A (100 ng/ml, added directly to existing media) for 5 min and extracts were examined by immunoblot analysis with antibodies to p-ERK, ERK, JNK and Tubulin. VEGF-A-stimulated ERK phosphorylation was similar in JNK-deficient and control cells. The data presented are representative of two experiments with independent endothelial cell preparations. Source data are included as Figure 1—figure supplement 3—source data 1.
-
Figure 1—figure supplement 3—source data 1
Source data for Figure 1—figure supplement 3.
This file contains raw source data used to make the graphs presented in Figure 1—figure supplement 3.
- https://doi.org/10.7554/eLife.18414.010
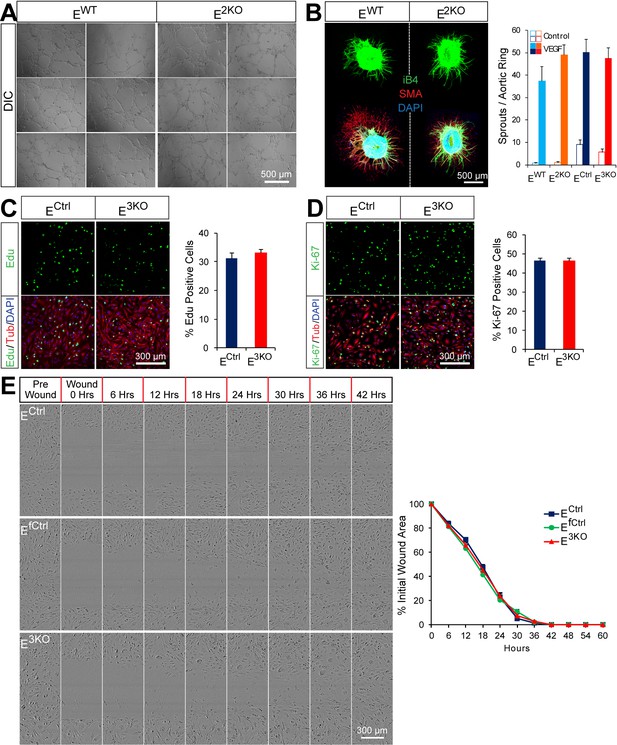
Endothelial JNK is not required for proliferation, migration, and angiogenic responses in vitro.
(A) JNK-deficient and control primary MLEC form similar tubular networks in matrigel. Images are representative of two experiments performed in triplicate with independent primary MLEC preparations. (B) Representative maximum projection confocal images of collagen embedded aortic ring explants. Similar numbers of VEGF-induced iB4 (green) positive microvessels sprouting from aortic rings from control and endothelial JNK-deficient mice were detected. Smooth muscle actin (SMA) immunofluorescence (red) labels supporting cells. DAPI (blue) labels nuclei. Quantitation of microvessel number demonstrated no significant differences between aortic rings from control and JNK-deficient mice (mean ± SEM; n = 8~21 rings per group). The data presented were obtained in one experiment and are representative of three experiments with similar results. Aortas from 2~3 mice per group were used in each experiment. (C,D) Representative confocal images and quantitation of the percentage of endothelial cells staining positive for the incorporation of Edu (green, C) or the proliferation marker Ki-67 (green, D) (mean ± SEM; n = 10 images per group). The data presented were obtained in one experiment and are representative of three experiments with similar results. αTubulin (red) labels cell bodies. DAPI (blue) labels nuclei. (E) Endothelial monolayers were examined in a scratch assay and wound closure was monitored using an IncuCyte ZOOM system. Representative images show similar migratory ability of control and JNK-deficient endothelial cells. Quantification of wound area closure demonstrated no statistically significant differences (p>0.05) between JNK-deficient and control endothelial cells (mean ± SEM; n = 8). The data presented were obtained in one experiment and are representative of three experiments with similar results. Source data are included as Figure 1—figure supplement 4—source data 1.
-
Figure 1—figure supplement 4—source data 1
Source data for Figure 1—figure supplement 4.
This file contains raw source data used to make the graphs presented in Figure 1—figure supplement 4.
- https://doi.org/10.7554/eLife.18414.012
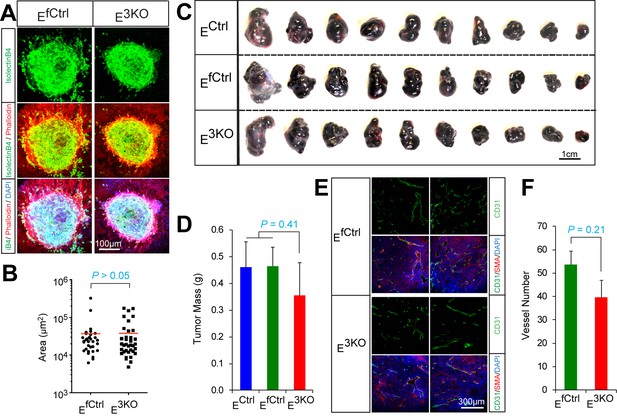
Endothelial JNK is not required for in vivo pathologic angiogenesis.
(A,B) Representative confocal images of laser-induced choroidal neovascular (CNV) tufts in control and JNK-deficient mice at day 7 post-treatment stained with iB4, Phalloidin, and DAPI (A). Quantitation of CNV area (B) demonstrated no statistically significant differences (p>0.05) between groups (mean ± SEM; n = 32~36 CNV tufts; 5 mice). (C,D) Congenic B16F10 melanoma cells were grown (2 wks) as subcutaneous tumors in the flanks of JNK-deficient and control mice (C). Measurement of tumor mass (D) demonstrated no statistically significant differences (p>0.05) between groups (mean ± SEM; n = 10 tumors). The data presented are representative of data obtained from two different experiments. (E,F) B16F10 melanoma tumor angiogenesis was examined by staining frozen tumor sections with antibodies to CD31 (green) and SMA (red). DNA was stained with DAPI (E). Quantitation of vessel number (F) demonstrated no statistically significant differences (p>0.05) between groups (mean ± SEM; n = 10 tumors, 5~6 images per tumor). Source data are included as Figure 1—figure supplement 5—source data 1.
-
Figure 1—figure supplement 5—source data 1
Source data for Figure 1—figure supplement 5.
This file contains raw source data used to make the graphs presented in Figure 1—figure supplement 5.
- https://doi.org/10.7554/eLife.18414.014
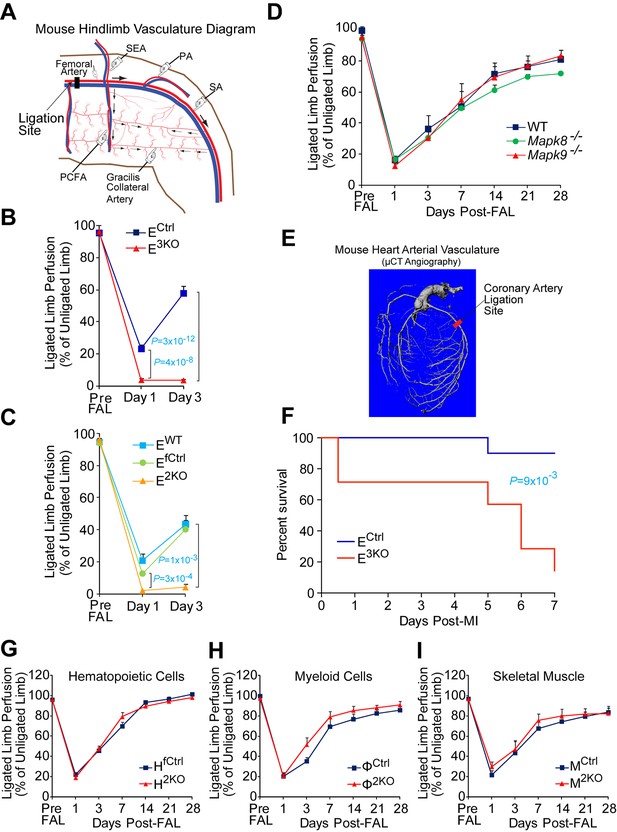
Compound JNK-deficiency in endothelial cells causes defects in the response to arterial occlusion.
(A,B) Simplified diagram of the medial aspect of the mouse hindlimb skeletal muscle vasculature indicating the location of the femoral artery ligation site (proximal to the PCFA) (A). Quantitation of limb blood flow by laser doppler imaging demonstrates increased blood perfusion blockade and no recovery at day 3 post-FAL in compound JNK1/2/3-deficient (E3KO) mice (B) (mean ± SEM; n = 7~14). (C) Post-FAL (as in panel A,B), quantitation of limb blood flow by laser doppler imaging demonstrates increased blood perfusion blockade and no recovery 3 days after ligation in endothelial JNK1/2-deficient (E2KO) mice (mean ± SEM; n = 4). (D) Post-FAL (panel A), quantitation of limb blood flow by laser doppler imaging demonstrates no significant differences in blood perfusion blockade and recovery by Mapk8-/- mice or Mapk9-/- mice compared to WT mice (mean ± SEM; n = 5). (E,F) Simplified diagram of the coronary artery circulation indicating the location of the coronary artery ligation site (E). After coronary artery ligation, endothelial JNK-deficient mice exhibit significantly decreased survival compared with control mice (n = 7~10) (F). (G–I) Limb blood perfusion measured by laser doppler imaging (mean ± SEM (n = 5~10) shows no significant differences (p>0.05) in blood perfusion blockade and restoration post-FAL between control mice and mice that lack JNK1 plus JNK2 in (G) all hematopoietic cells (H2KO), (H) myeloid cells (Φ2KO), or (I) skeletal muscle (M2KO). Source data are included as Figure 1—figure supplement 6—source data 1.
-
Figure 1—figure supplement 6—source data 1
Source data for Figure 1—figure supplement 6.
This file contains raw source data used to make the graphs presented in Figure 1—figure supplement 6.
- https://doi.org/10.7554/eLife.18414.016
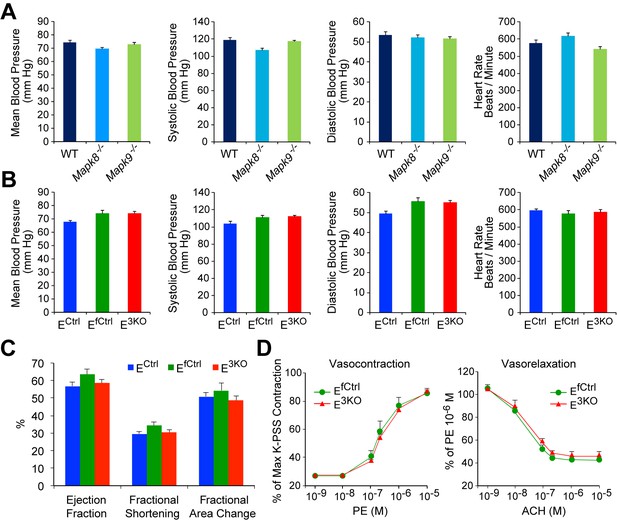
JNK deficient mice show no perturbations in overall cardiovascular function.
(A,B) Analysis of blood pressure and heart rate in WT mice, Mapk8-/- mice, and Mapk9-/- mice (A) and endothelial JNK-deficient mice and control mice (B) demonstrates no statistically significant differences (p>0.05) between groups (mean ± SEM; n = 9~15). (C) Echocardiographic analysis of heart function demonstrates no statistically significant differences (p>0.05) between endothelial JNK-deficient mice and control mice (mean ± SEM; n = 12~15). (D) Segments from thoracic aortas from E3KO and control mice were mounted on a myograph and vasocontraction and vasorelaxation in response to increasing doses of phenylephrine (PE) or acetylcholine (ACH), respectively, were recorded. Contraction in response to PE is expressed as a percentage of maximum aortic contraction obtained in the presence of K+ containing buffer (K-PSS). Vasorelaxation in response to ACH is expressed as a percentage of maximum contraction obtained in the presence of 10 μM PE (mean ± SEM; n = 3). The data presented are representative of two independent experiments. Source data are included as Figure 1—figure supplement 7—source data 1.
-
Figure 1—figure supplement 7—source data 1
Source data for Figure 1—figure supplement 7.
This file contains raw source data used to make the graphs presented in Figure 1—figure supplement 7.
- https://doi.org/10.7554/eLife.18414.018
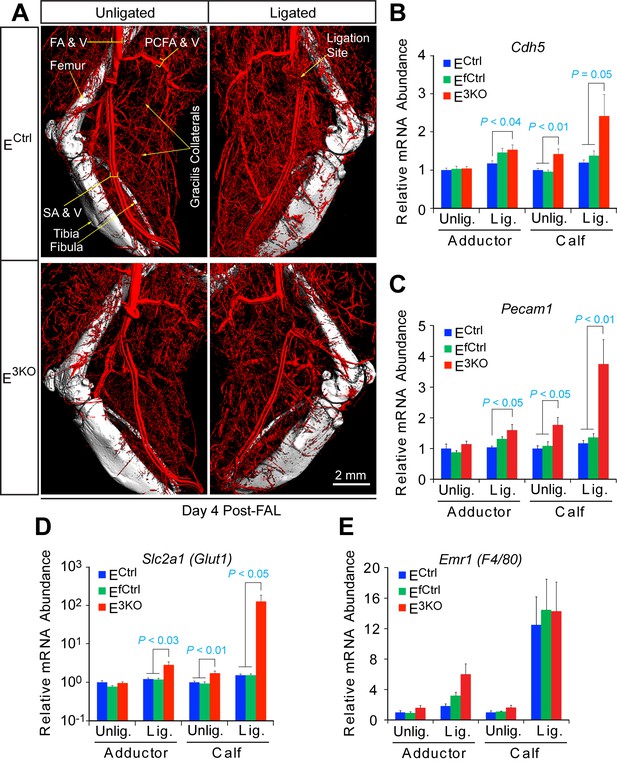
JNK-deficiency causes defects in artery size and connectivity, but not the hypoxia response after femoral artery ligation.
(A) Contrast (Bismuth/gelatin) perfused hindlimb vasculature was examined by µCT analysis at day 4 post-FAL. The images are representative of 7~8 mice analyzed per group. (B–E) Taqman gene expression analysis quantitating the mRNA abundance of the endothelial cell specific markers Cdh5 (B) and Pecam1 (C), the hypoxia responsive gene Slc2a1 (Glut1) (D), and the macrophage marker Emr1 (F4/80) (E) on day 4 post FAL in the adductor and calf muscles (mean ± SEM; n = 7~8 mice per group). Source data are included as Figure 1—figure supplement 8—source data 1.
-
Figure 1—figure supplement 8—source data 1
Source data for Figure 1—figure supplement 8.
This file contains raw source data used to make the graphs presented in Figure 1—figure supplement 8.
- https://doi.org/10.7554/eLife.18414.020
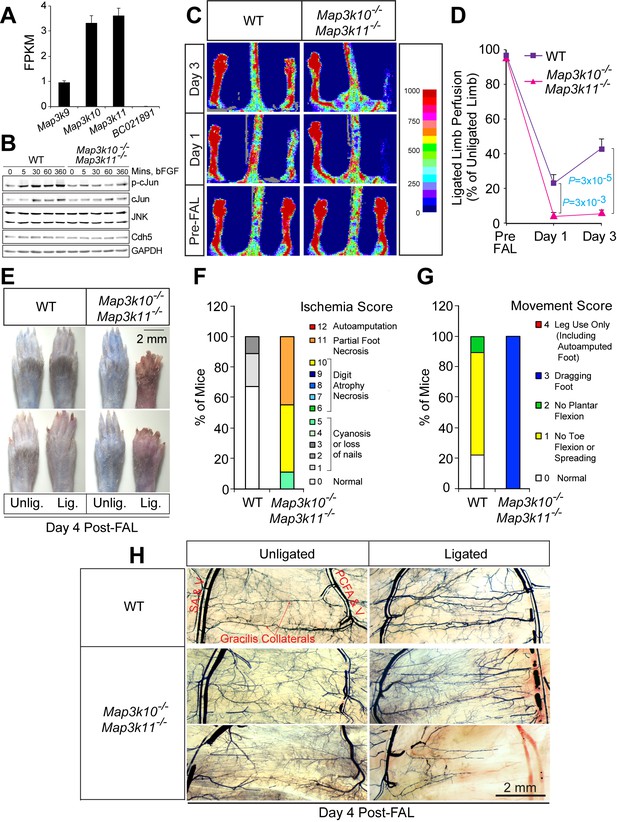
Severe ischemic injury in MLK2/3-deficient mice after femoral artery ligation.
(A) The expression of members of the MLK protein kinase family (MLK1, MLK2, MLK3 & MLK4) in primary MLEC cultures was examined by measurement of Map3k9, Map3k10, Map3k11, and BC021891 mRNA by RNA-seq analysis (mean fragments per kilobase of transcript per million mapped reads (FPKM) ± SEM; n = 6). (B) Primary wild-type (WT) and Map3k10-/- Map3k11-/- MLEC cultures were treated without and with 100 ng/ml bFGF and examined by immunoblot analysis by probing with antibodies to pSer63-cJun, cJun, JNK, Cdh5, and GAPDH. (C) Representative laser doppler images showing blood perfusion (high perfusion red, no perfusion dark blue) in the hindlimbs of WT and Map3k10-/- Map3k11-/- mice mice prior to unilateral FA ligation (Pre-FAL) and post-FAL. (D) Quantitation of hindlimb blood flow demonstrated that MLK2/3-deficient mice exhibited significantly increased blood perfusion blockade and no recovery by day 3 post-FAL compared with control mice (mean ± SEM; n = 7). (E) Representative images of mouse paws on Day 4 post-FAL. Lig., ligated; Unlig., contralateral unligated. (F,G) Quantitation of ischemic (F) and movement (G) scores for mice on Day 4 post-FAL (n = 9). (H) Representative whole mount preparations of the medial surface of Microfil-filled adductor muscle vasculature isolated from day 4 post-FAL hindlimbs and contralateral unligated limbs. Source data are included as Figure 2—source data 1.
-
Figure 2—source data 1
Source data for Figure 2.
This file contains raw source data used to make the graphs presented in Figure 2.
- https://doi.org/10.7554/eLife.18414.022
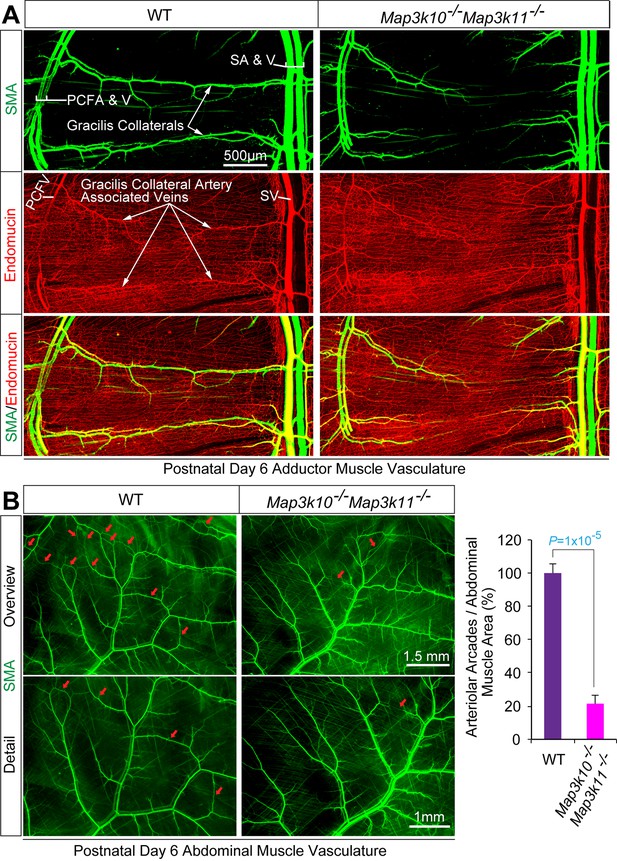
MLK2/3-deficient mice exhibit defects in native collateral artery formation.
(A) Representative confocal images (n = 5 mice) of control and MLK2/3-deficient whole mount P6 adductor muscle vasculature stained with antibodies to endomucin (capillary and venous vasculature, red) and SMA (arterial and venous smooth muscle, green). Gracilis collateral arteries in WT mice, but not MLK2/3-deficient mice, interconnect the PCFA to the SA. (B) Representative stereomicroscope images of P6 whole mount abdominal muscle stained with an antibody to SMA (green). Arteriole-to-arteriole arcades are indicated (red arrows). The abdominal muscle vasculature of Map3k10-/-Map3k11-/- mice shows very few arteriole-to-arteriole interconnections. Quantitation reveals significantly reduced arteriolar arcade numbers in Map3k10-/-Map3k11-/- mice compared to WT mice (mean ± SEM; n = 5 mice). Source data are included as Figure 3—source data 1.
-
Figure 3—source data 1
Source data for Figure 3.
This file contains raw source data used to make the graphs presented in Figure 3.
- https://doi.org/10.7554/eLife.18414.024
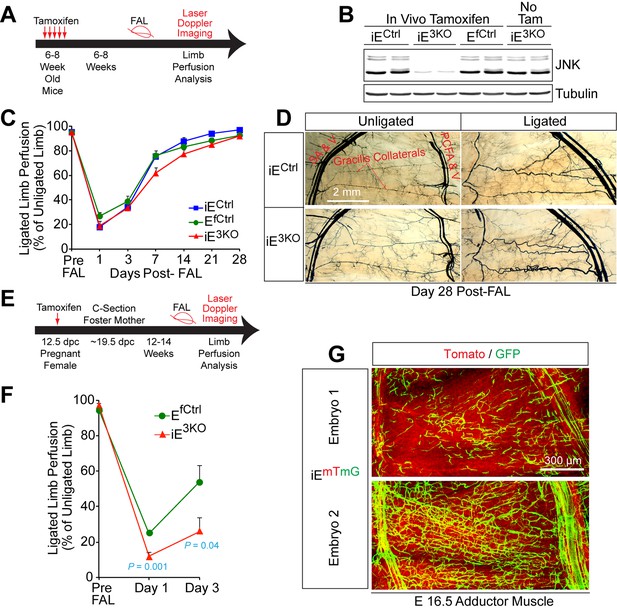
Endothelial JNK is not required for the arteriogenic response of gracilis collaterals in adult mice.
(A) Timeline of tamoxifen administration to induce disruption of Mapk8LoxP and Mapk9LoxP alleles in the vascular endothelium of adult mice prior to FAL and analysis of blood flow by laser doppler imaging. (B) Primary MLEC cultures prepared from mice treated without and with tamoxifen were examined by immunoblot analysis by probing with antibodies to JNK and αTubulin. The data are representative of two independent MLEC isolations per group (2~3 mice used per cell preparation). (C) Quantitation of laser doppler analysis of limb blood flow demonstrated no significant differences (p>0.05) in blood perfusion blockade and recovery over 28 days post-FAL between tamoxifen-treated endothelial JNK-deficient mice and tamoxifen-treated control mice (mean ± SEM; n = 5~10). (D) Microfil perfusion of adductor muscle vasculature demonstrated the presence of similar gracilis collateral arteries in JNK-deficient and control mice and similar collateral artery remodeling at day 28 post-FAL. The images are representative of 5~8 mice per group. (E) Timeline of tamoxifen administration to induce disruption of Mapk8LoxP and Mapk9LoxP alleles in the vascular endothelium during embryonic development prior to analysis of FAL in adults and examination of blood flow by laser doppler imaging. (F) Quantitation of laser doppler analysis of limb blood flow demonstrated significantly enhanced blood perfusion blockade in adult mice with embryonic endothelial JNK-deficiency compared with control mice (mean ± SEM; n = 5~6 mice per group). (G) The adductor muscle vasculature of two E16.5 embryos obtained from a pregnant female mouse that was treated with tamoxifen at 12.5 dpc was examined by confocal microscopy. The Rosa26mTmG genetic background allows detection of Cre-mdiated recombination in vascular endothelial cells (green). The data presented are representative of six mice examined. Source data are included as Figure 4—source data 1.
-
Figure 4—source data 1
Source data for Figure 4.
This file contains raw source data used to make the graphs presented in Figure 4.
- https://doi.org/10.7554/eLife.18414.026
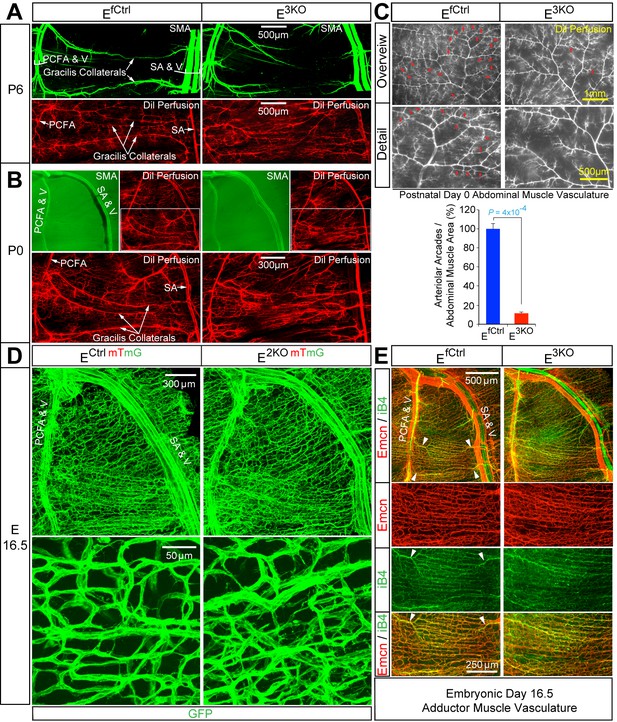
Endothelial JNK-deficient mice display abnormal native collateral arteries.
(A,B) Representative confocal images (n = 7 mice) of whole mount adductor muscle vasculature reveals SMA-covered gracilis collateral arteries in P6 control mice, but not JNK-deficient mice (A). Confocal imaging of Dil perfused P6 adductor muscle vasculature (n = 5 mice) demonstrates distinct gracilis collaterals interconnecting the PCFA to the SA in control mice. Vessels emerging from the PCFA and the SA in E3KO mice do not fully interconnect, but branch into smaller vessels. At P0, gracilis collaterals were not SMA-covered, but were perfused with Dil in control mice (B). The analogous vessels in E3KO mice did not interconnect, but branched extensively into smaller vessels (B) (n = 5 mice). (C) Representative stereomicroscope images of Dil-perfused abdominal muscle arterial vasculature of control and JNK-deficient P0 mice. Arteriole-to-arteriole arcades (indicated by red arrows) were quantitated (mean ± SEM; n = 3 mice). (D) Representative confocal images (n = 3~4 mice) of whole mount adductor muscles of control and JNK-deficient E16.5 embryos showing GFP-labeled vascular endothelial cells. (E) Representative confocal images (n = 3~5 mice) of control and JNK-deficient E16.5 embryo adductor muscle vasculature immunostained for Endomucin (Emcn, red) and isolectinB4 (iB4, green). Prominent vessels emerging from the PCFA and SA are indicated with white arrowheads. Source data are included as Figure 5—source data 1.
-
Figure 5—source data 1
Source data for Figure 5.
This file contains raw source data used to make the graphs presented in Figure 5.
- https://doi.org/10.7554/eLife.18414.028
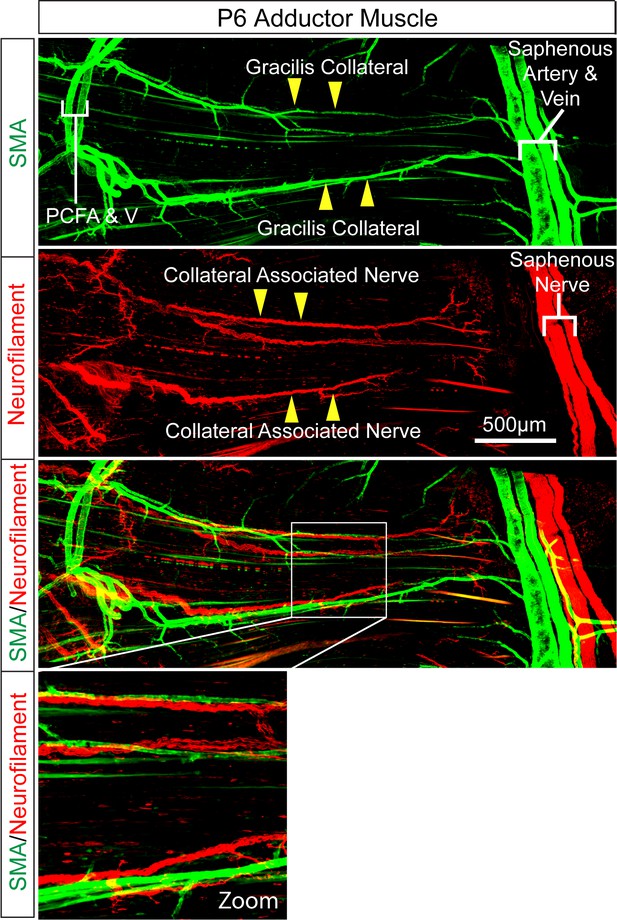
Intimate association of gracilis collaterals and peripheral nerves in adductor muscles.
Confocal microscopy of a whole mount adductor muscle stained with antibodies to SMA (green) and Neurofilament-M (red) illustrates the close association of gracilis collateral arteries with peripheral nerves.
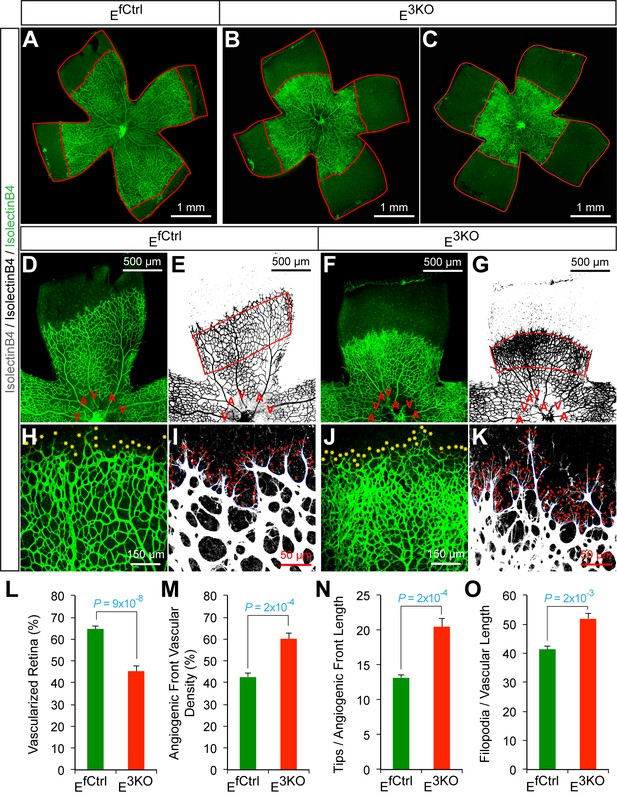
Abnormal retinal vascular development associated with excessive sprouting of tip cells in endothelial JNK-deficient mice.
(A–C) Collages of confocal images of P6 whole mount retinas stained with isolectinB4 (iB4) show reduced vascular extension in JNK-deficient retinas (B,C) compared with littermate control retinas (A). The images are representative of 17~31 retinas examined for each genotype. (D–K) Higher magnification reveals increased vascular density (D–G, H, J), increased numbers of tip cells (yellow asterisks, H & J) and increased numbers of filopodia (red dots, I and K) at the vascular front region of JNK-deficient retinas compared to littermate control retinas. A, artery; V, vein. (L–O) The vascularized retinal area (L), vascular density within angiogenic front regions outlined in panels E & G (M), tip cell number (N), and filopodia number (O) is presented (mean ± SEM; n = 17~31 (panel l); n = 6~10 (panels M–O). Source data are included as Figure 6—source data 1.
-
Figure 6—source data 1
Source data for Figure 6.
This file contains raw source data used to make the graphs presented in Figure 6.
- https://doi.org/10.7554/eLife.18414.031
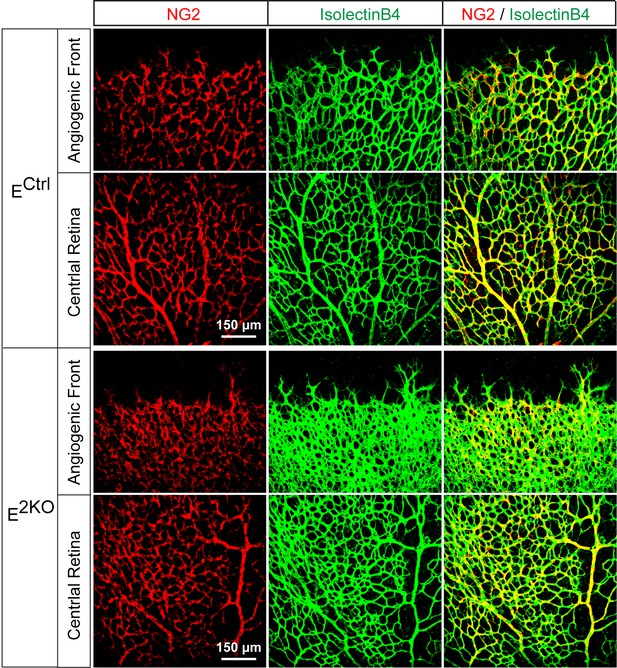
NG2+ pericyte coverage of the P6 retinal vasculature.
Confocal microscopy of whole mount retinal vasculature stained with an antibody to the pericyte marker NG2 (red) and isolectinB4 (green) demonstrated no obvious differences in vessel pericyte coverage in retinas from endothelial JNK-deficient mice compared to littermate control mice. The data presented are representative of images obtained from 4 mice per group.
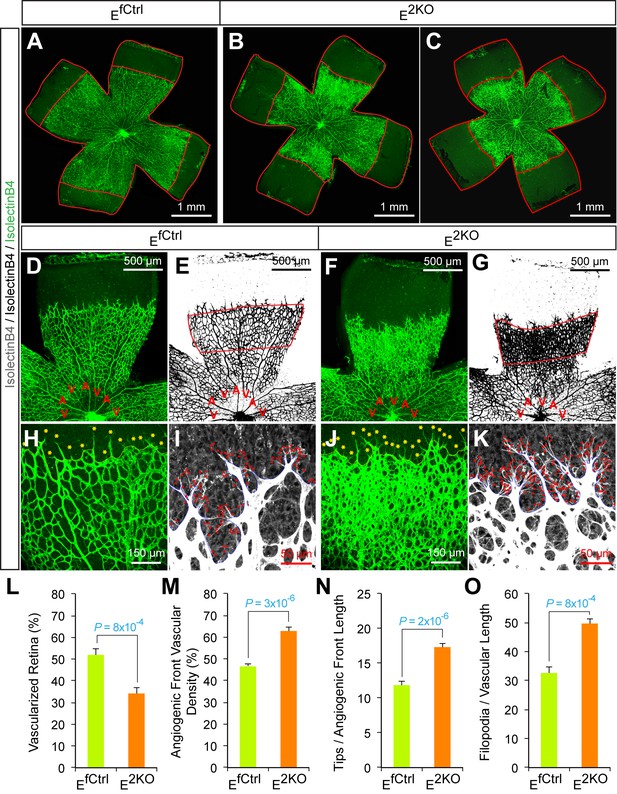
Abnormal retinal vascular development associated with excessive sprouting in endothelial JNK1/2-deficient mice.
(A–C) Collages of confocal images of P6 whole mount retinas stained with iB4 show reduced vascular extension in JNK1/2-deficient retinas (B,C) compared with littermate control retinas (A). The images are representative of 4~9 retinas examined for each genotype (≥4 mice per group). (D–K) Higher magnification reveals increased vascular density (D–G, H, J), increased numbers of tip cells (yellow asterisks, H & J) and increased numbers of filopodia (red dots, I and K) at the vascular front region of JNK1/2-deficient retinas compared to littermate control retinas. A, artery; V, vein. (L–O) The vascularized retinal area (L), vascular density within angiogenic front regions outlined in panels e & g (M), tip cell number (N), and filopodia number (O) is presented (mean ± SEM; n = 4~9 (panel l); n = 4~8 (panels M–O) (≥4 mice per group). Source data are included as Figure 6—figure supplement 2—source data 1.
-
Figure 6—figure supplement 2—source data 1
Source data for Figure 6—figure supplement 2.
This file contains raw source data used to make the graphs presented in Figure 6—figure supplement 2.
- https://doi.org/10.7554/eLife.18414.034
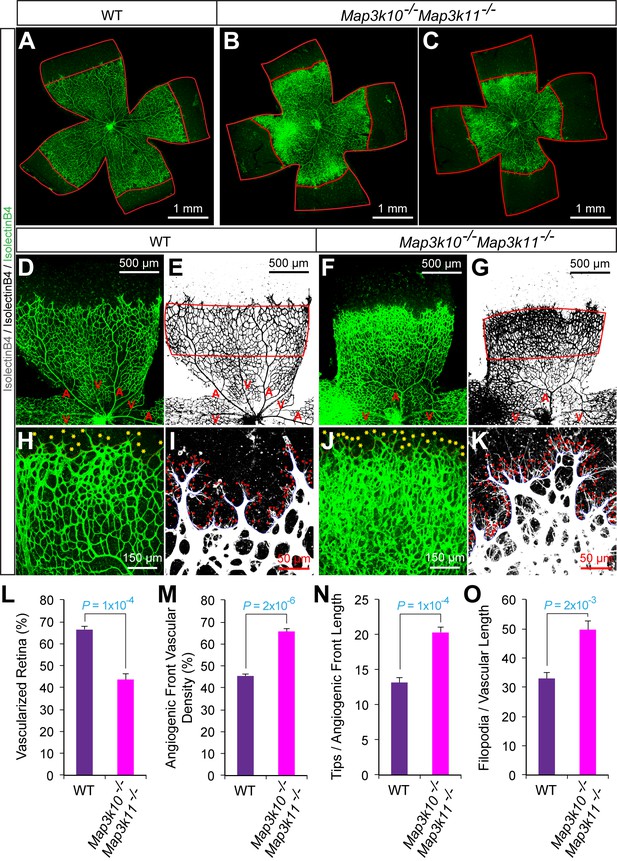
Abnormal retinal vascular development associated with excessive sprouting in Map3k10-/- Map3k11-/- mice.
(A–C) Collages of confocal images of P6 whole mount retinas stained with isolectinB4 show reduced vascular extension in MLK2/3-deficient retinas (B,C) compared with littermate control retinas (A). The images are representative of 5 mice examined for each genotype. (D–K) Higher magnification reveals increased vascular density (D–G, H, J), increased numbers of tip cells (yellow asterisks, H & J) and increased numbers of filopodia (red dots, I and K) at the vascular front region of MLK2/3-deficient retinas compared to littermate control retinas. A, artery; V, vein. (L–O) The vascularized retinal area (L), vascular density within angiogenic front regions outlined in panels E & G (M), tip cell number (N), and filopodia number (O) is presented (mean ± SEM; n = 5 mice per group). Source data are included as Figure 6—figure supplement 3—source data 1.
-
Figure 6—figure supplement 3—source data 1
Source data for Figure 6—figure supplement 3.
This file contains raw source data used to make the graphs presented in Figure 6—figure supplement 3.
- https://doi.org/10.7554/eLife.18414.036
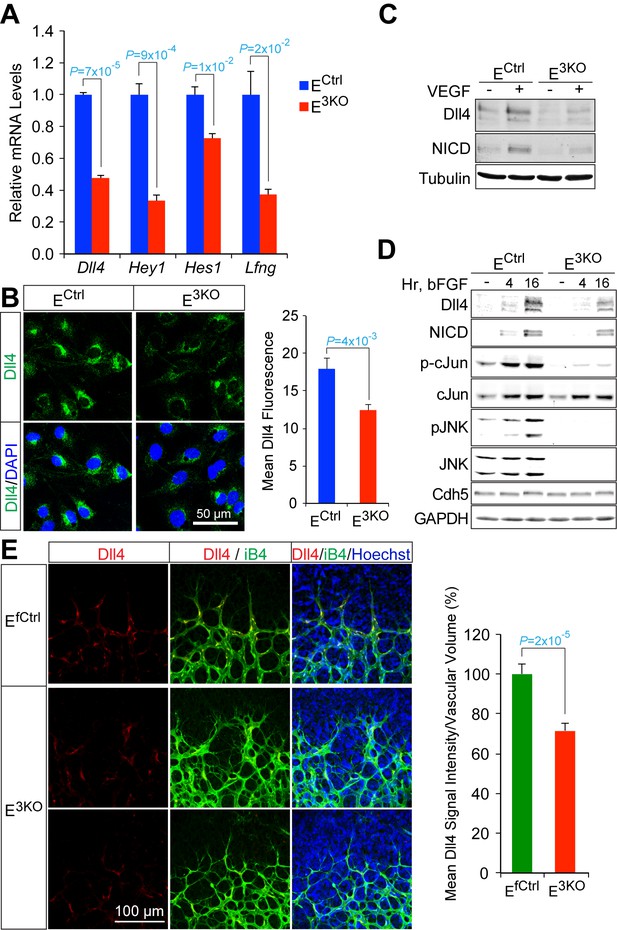
Reduced Dll4 / Notch signaling in the JNK-deficient vascular endothelium.
(A) Quantitative RT-PCR analysis of Notch pathway genes revealing reduced expression in JNK-deficient primary endothelial cells compared with control cells (mean ± SEM; n = 4). The data shown are representative of the results obtained with three independent primary endothelial cell preparations. (B) Dll4 expression by control and JNK-deficient primary endothelial cells was examined by Immunofluorescence analysis (mean ± SEM; n = 10). (C) Control and JNK-deficient primary endothelial cells treated without and with 100 ng/ml VEGF (16 hr) were examined by immunoblot analysis by probing with antibodies to Dll4, Notch intracellular domain (NICD), and αTubulin. The data are representative of experiments performed using two independent endothelial cell preparations. (D) Endothelial cells treated without and with 100 ng/ml bFGF were examined by immunoblot analysis by probing with antibodies to Dll4, NICD, pSer63-cJun, cJun, pJNK, JNK, Cdh5 and GAPDH. The data are representative of experiments performed using two independent endothelial cell preparations. (E) Confocal immunofluorescence analysis of P6 whole mount retinas immunostained for Dll4 (red), isolectinB4 (iB4, green), and Hoechst (DNA, blue) demonstrates that JNK-deficiency causes reduced expression of Dll4 at the angiogenic vascular front compared with retinas from littermate control mice (mean ± SEM; n = 42~44). Source data are included as Figure 7—source data 1.
-
Figure 7—source data 1
Source data for Figure 7.
This file contains raw source data used to make the graphs presented in Figure 7—figure supplement 1.
- https://doi.org/10.7554/eLife.18414.038
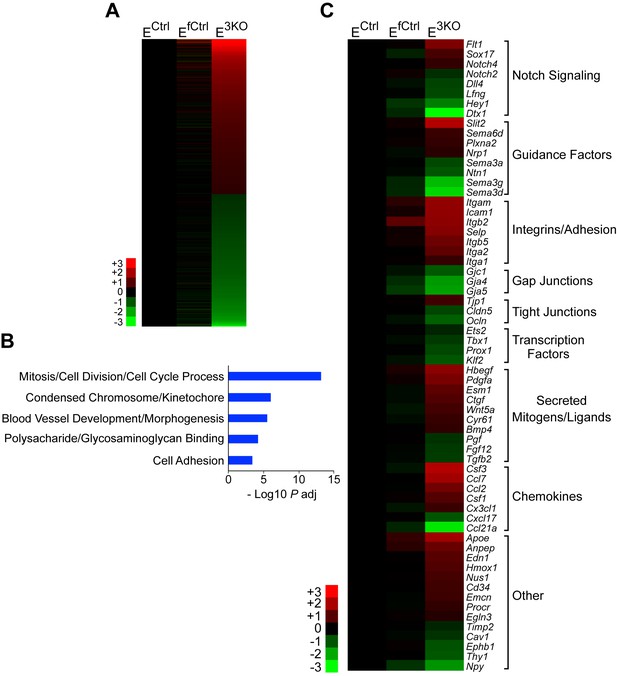
RNA-Seq analysis of differentially expressed genes between control and JNK-deficient endothelial cells.
(A) Heatmap of the 781 differentially expressed genes (FPKM > 2; absolute log2 fold change > 0.5; n = 3; q < 0.05) between MLEC cultures isolated from endothelial JNK-deficient mice and control MLEC cultures. JNK-deficiency caused similar numbers of upregulated and downregulated genes. (B) Gene ontology analysis of the differentially expressed genes demonstrated significant enrichment for several biological processes. (C) Differentially expressed genes related to vascular development, morphogenesis, and function were grouped in several categories and are presented as a heatmap. Genes are displayed with highest upregulation top and highest downregulation bottom within each category. Source data are included as Figure 7—figure supplement 1—source data 1.
-
Figure 7—figure supplement 1—source data 1
Source data for Figure 7—figure supplement 1.
This file contains raw source data used to make the graphs presented in Figure 7—figure supplement 1.
- https://doi.org/10.7554/eLife.18414.040